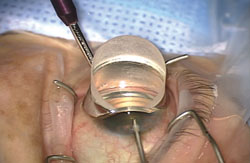
Now that is changing, thanks to the development of new surgical procedures that involve far less risk: the microincision glaucoma surgeries, or MIGS. Current options in this category include the iStent, the Trabectome and endoscopic cyclophotocoagulation, or ECP (although not everyone considers ECP a MIGS procedure). In addition, several more MIGS options are in the pipeline. Suddenly, because of the low risk associated with these procedures, using surgery to address mild, early cases of glaucoma is becoming a valid option. Furthermore, because they can be performed through a cataract incision, surgeons are using these procedures as adjuncts to cataract surgery. In many ways, it’s a revolution in glaucoma treatment—one being felt by surgeons who would seldom have attempted to treat glaucoma with surgery in the past.
Brian Francis, MD, MS, professor of ophthalmology in the Glaucoma Service at the Doheny Eye Institute, Geffen School of Medicine, University of California, Los Angeles, has used all of the current MIGS options. “A good working definition of MIGS would be glaucoma surgeries that are appropriate for mild to moderate glaucoma, done through a small incision, that combine well with cataract surgery and typically are extremely safe,” he says. “Their IOP-lowering ability is augmented when they’re combined with cataract surgery. And, they generally don’t limit your future options, in terms of traditional filtering surgery, because they don’t scar the conjunctiva. In fact, some people include the lack of a bleb in their definition of MIGS.” (He notes that most doctors do not consider canaloplasty to be a MIGS procedure because of the need for external dissection.)
Here, Dr. Francis and three other surgeons with extensive MIGS experience offer their thoughts on how these procedures compare to each other, why they’re worth adopting, and what the future may hold.
Working With the iStent
|
“There are many advantages to this approach, but the most significant advantage by far is safety,” he continues. “In the United States premarket approval trial, patients were randomized to cataract surgery alone or cataract surgery plus iStent. There was no difference in adverse events between the groups. So, the addition of the iStent did not adversely affect visual outcomes or subject the patient to a greater frequency of side effects as compared to phacoemulsification alone. The safety of the iStent was clearly established.
“In terms of efficacy, both groups in that trial had improved IOP following the surgery, but the patients receiving an iStent were statistically more likely to either be off medications completely or require fewer of them,” he notes. “So that was the benefit: Patients achieved a modest reduction in pressure and medication use without being subjected to significant risk. That’s an option we didn’t have in the past, prior to the MIGS procedures. It’s an excellent adjunct to our surgical offerings.” (For advice on using the iStent in surgery, see the sidebar on p. 28.)
One issue that has been raised in connection to the iStent is whether implanting more than one will produce a better result. Some work by surgeons such as Ike Ahmed, MD, in Toronto, suggests that this may be the case. “Many surgeons believe that placing two iStents will cause a greater pressure reduction because a greater portion of the outflow system is influenced,” says Dr. Samuelson. “Likewise, that’s part of the reason for the development of the Hydrus device [still in the pipeline]; it’s an 8-mm stent, so it covers a much broader segment of the trabecular meshwork and Schlemm’s canal. It should bring more collector channels into play, hopefully providing greater efficacy.” Dr. Samuelson notes, however, that the potential superiority of using multiple iStents hasn’t been clinically proven in a randomized trial. “It makes sense intuitively, but we need more data to support and further delineate that approach, especially to help guide us on the ideal location to implant,” he says.
| ||||||
Using the Trabectome
In the Trabectome procedure, the surgeon uses a disposable handpiece equipped with irrigation and aspiration ports to perform bipolar cautery on the trabecular meshwork, ablating a portion (or all) of the meshwork as a means to increase aqueous outflow. “The Trabectome is definitely a MIGS procedure,” says Dr. Francis. “It’s angle-based; it has an extremely good safety profile; it’s very effective in mild-to-moderate glaucoma and when combined with cataract surgery. The fact that it doesn’t require placing a foreign body such as a stent inside the eye doesn’t disqualify it; in fact, that may be an advantage.”
Dr. Francis offers advice for surgeons considering adopting the Trabectome:
• Learn the goniosurgical approach. “This is important for both the Trabectome and the iStent,” says Dr. Francis. “This is a different type of surgery. I’d recommend increasing your use of gonioscopy in the clinic so you become very familiar with the trabecular meshwork and angle landmarks. Also, practice using a goniosurgical lens with your cataract patients. When you’re finishing up a standard cataract surgery, take a couple of minutes to tilt the head and microscope in the manner you would if performing gonio surgery; place the goniolens on the eye and practice looking at the angle. I think that’s probably the most helpful thing you can do; it helps you get used to that view. When you’re ready to do the surgery, the fewer things that are novel, the better.”
• Pick appropriate patients. “When selecting patients for this procedure, be sure they have mild to moderate glaucoma, have a target pressure in the mid-teens and can tolerate medications after surgery, should they be needed,” he says.
• Do careful goniotomy on each Trabectome patient before surgery. “You need to know what the patient’s angle looks like before you go in there,” he says.
• Make sure you’re in the trabecular meshwork. “The main thing is that you need to hit the target tissue,” says Dr. Francis. “In most cases you can do this because the angle landmarks are usually clear and the trabecular meshwork usually has some degree of pigment.
“There are a couple of tricks you can use if the trabecular meshwork is not readily visible,” he continues. “One is to actually decompress the eye, letting it become hypotonous for about 10 seconds. That allows blood to reflux into Schlemm’s canal. Then, even after you repressurize the eye, there’s a nice red stripe defining the trabecular meshwork. It makes it very easy to see the target. Also, if you’re confused about the angle landmarks, you can look at other parts of the eye. Usually the inferior segment of the angle is the easiest to identify because it has some pigment in the trabecular meshwork. So in a pinch, look inferiorly, locate the trabecular meshwork and follow the anatomy around to the nasal quadrant.”
• Learn to know when you’ve gone too deep. “It’s easy to go too deep,” says Dr. Francis. “If this happens, you’ll feel the resistance and see the eye rotating because the tip is probably digging into the outer wall of Schlemm’s canal. When you’re correctly ablating the trabecular meshwork, there’s almost no resistance; the device moves very easily through the tissue. That’s one reason the company requires wet-lab training; you learn to recognize the feeling that lets you know you’re in the right area.”
• Flare the internal opening of the corneal incision. “This means instead of going straight in with the keratome blade, once you’re inside, you turn the blade side to side,” Dr. Francis explains. “It makes the internal opening larger than the external opening. That allows you to move the instrument side to side inside the eye without tugging on the corneal tissue. That, in turn, allows you to do more ablation.
“Typically, a beginning surgeon can do about three clock hours, or 90 degrees, fairly easily,” he continues. “But when you become more comfortable using the device and you have a flared incision, you can ablate as much as five or six clock hours, treating more of the angle. Treating more of the angle does seem to correlate with better lowering of IOP because you’re accessing more of the collector channels. However, I don’t recommend that surgeons new to the procedure try to ablate five or six clock hours. You’ll end up slipping out of Schlemm’s canal and causing iris trauma or corneal trauma. Start with what’s comfortable and work up to doing larger segments of the angle.”
ECP as MIGS
As previously noted, not every surgeon considers ECP—in which fiber optics allow the surgeon to partially coagulate ciliary process tissue from inside the eye, reducing the production of aqueous—one of the MIGS procedures. “I don’t know whether I’d classify ECP as a classic MIGS procedure, but it’s conceptually similar,” says Dr. Samuelson. “It’s a less-invasive approach to pressure reduction, and it has considerably less risk than trabeculectomy or aqueous drainage devices. So I think it’s reasonable to talk about ECP in the same conversation as MIGS procedures. There are patients in whom ciliary body ablative procedures may be beneficial, so I do believe there’s a role for ECP in select patients.”
| ||||||
One factor working against more widespread use of ECP, says Dr. Noecker, is the capital cost involved in purchasing the equipment. “Also, some doctors may still equate using ECP with the issues surrounding transscleral cyclophotocoagulation,” he says. “Of course, TCP is a lot more traumatic to the eye than ECP, in terms of causing inflammation and affecting structures we don’t necessarily want to affect.”
Dr. Francis does see ECP as more versatile than the other MIGS procedures. “ECP can be used in open-angle glaucoma, narrow-angle glaucoma, even neovascular glaucoma,” he notes. “And it can be used after another procedure has failed. In other words, if a trabeculectomy or tube shunt has failed, you can go in with ECP and get additional pressure lowering. The other MIGS procedures are generally reserved for first-line surgery.”
Dr. Francis adds that the endoscope can be put to other uses as well. “You can use it to view the lens if you have problems with a lens implant,” he points out. “You can look at zonular stability if you’re trying to decide whether or not to put in a sulcus lens. You can even use it for retinal procedures when you have a corneal opacity and can’t see the retina, or when you want to work inside the eye very anteriorly, even anterior to the equator.”
Dr. Noecker offers these suggestions when first attempting ECP:
• Pick easy cases. “Start with a normal-sized eye, not a small eye, and avoid eyes that have other problems,” says Dr. Noecker. “It’s all about recognizing the anatomy, so pick eyes in which the anatomy is easy to work with and you have a good view. If you can’t see the anatomy, you don’t want to treat.”
• Be thorough in your treatment. “The average eye has about 70 ciliary processes,” he says. “If you treat all 70 you’ll get the best result. If you only treat 30 of them you won’t lower pressure quite as well because there are still a lot of processes making aqueous.”
• Treat inflammation aggressively. “I’m very proactive about controlling inflammation,” he says. “I give IV steroids at the time of surgery; I put them inside the eye; and I use intensive eye drop therapy right out of the gate. For the first few days postop I use a lot of steroids, and the eyes quiet down very quickly.”
Comparing Efficacy
One issue facing surgeons who are interested in adopting one of the MIGS procedures is deciding which one to adopt. Of course, there are a number of ways to compare procedures; one of the most obvious ways is to compare their efficacy and risk of complications. This is difficult to do definitively, because so far there’s no clinical data from head-to-head, randomized, controlled comparisons of the MIGS procedures. (Dr. Noecker also believes surgical results of the different MIGS procedures are difficult to compare because the results are somewhat surgeon-dependent.)
| ||||||
“Trabectome also comes with a risk of bleeding, which can mess up the patient’s vision in the short term,” he continues. “I wouldn’t use the Trabectome with a monocular patient because there’s a chance you’ll get blood in the eye that will interfere with his vision for a few weeks. With iStent there’s a very low occurrence of that, although you still have to be careful with people who are on blood thinners; even iStent patients can get hyphemas postoperatively. If you look at morbidity or visual downside, I’d say that ECP is probably the most favorable of the three.
“In terms of efficacy, my impression after having done all three procedures is that ECP is the most efficacious thing I do, followed by Trabectome and the iStent,” Dr. Noecker says. “The short-term efficacy achieved with the Trabectome may be comparable to what you get with ECP, but I see more failures with the Trabectome long term. That’s probably a result of the mechanism by which it works. You’re cauterizing away a strip of the trabecular meshwork, which eliminates resistance at the angle. That can be very effective in the short term, but anytime you’re cauterizing something, at a cellular level you’re going to get chronic inflammation. You’re creating a cleft, and the body doesn’t like to leave clefts alone. If the body can figure out a way to close it, it will. I think that’s why the Trabectome sometimes has late-term failures; over time the healing response catches up in many patients. Of course, if a procedure controls IOP for a few years and minimizes the need for medications, I’d say that’s not time badly spent, even if the problem is not resolved forever—as long as the cost isn’t too great.”
Dr. Noecker admits that ECP and iStent can also fail in some patients. “I’ve been doing ECP for a long time,” he says. “I’ve had patients in whom one treatment lasted forever; they’ve come back time and time again and never needed any further treatment. But I’ve also had patients who needed another therapy after a couple of years. Failures will happen with any procedure, so I think it’s the rate of failure that’s important. Right now we don’t know what the rate of failure will be for the iStent; it’s only been commercially available for about a year. In my hands, it seems to be pretty stable in the first year. Beyond that we have some clinical trial information, but not much real-world information.”
In terms of efficacy, Dr. Samuelson agrees that Trabectome and iStent probably have similar pressure-reducing capabilities, at least in the short term. “Long term, I prefer the minimally invasive approach of the iStent,” he says. “The Trabectome procedure involves more tissue destruction; you’re ablating and removing tissue, so it’s doing more to the eye. In contrast, an iStent does very little to the eye. Of course, that doesn’t mean it’s necessarily a better procedure; those who advocate the Trabectome feel that procedure offers some advantages as well.
“In terms of the efficacy of ECP, I’ve often used ECP as a means to help manage pressures that aren’t responding enough to other approaches,” he continues. “If I place an aqueous drainage device and the pressure is too high, sometimes I’ll use ECP to lower the pressure further instead of putting a second tube shunt in. Brian Francis from USC has published on this approach.”
Dr. Samuelson says he suspects the iStent is the safest of the MIGS options (including ECP). “Without a comparative trial, of course, that’s only conjecture,” he says. “However, the iStent is the only one that’s gone through a United States PMA trial. No other glaucoma device in history has gone through the scrutiny that the iStent had to go through as part of its FDA approval process. So we can say that we know with confidence, based on a very well-controlled, PMA trial, that there are few if any serious adverse effects from the iStent. We don’t have that same kind of data for ECP or Trabectome—although in my experience they are safe, and the data that’s available suggests they are safe.”
Comparing Skills Sets
|
“For example, you wouldn’t want to do your first iStent case on a day when your schedule is overbooked and time is tight,” he says. “You want to start when you can take your time. I usually tell new surgeons that whatever their normal cataract time is, they should allow two or three times that much for their first phaco-iStent procedure. Later on it will go much faster, but at the outset it’s better to not feel stressed during the procedure.”
Dr. Samuelson believes the first few cases with the Trabectome are a little bit easier than the first few with the iStent. “Both the Trabectome and
iStent do require very skilled intraoperative gonioscopy,” he notes. “In general, they’re comparable, but because the iStent requires placement of a device in a very specific location and orientation, I think it’s a little bit more challenging. I’d say that ECP is probably the easiest of the three, although not by a great amount.”
Dr. Noecker notes that the iStent, Trabectome and ECP all require some special skills. “To use the Trabectome or iStent you have to use intraoperative gonioscopy,” he explains. “That’s a separate skill that does require some practice, even though some of us use gonioscopy in the office on every single patient every single day. The reality is that doing it in the OR is a little bit different; it takes a while to learn to do it reproducibly, and the first time you try it can be very unnerving. ECP also involves a different skill set; you have to get used to how it looks on the TV monitor when you move your hand and use the tools inside the eye, but you can learn that quite readily.”
Dr. Francis agrees. “Performing ECP is a bit different from what we’re used to in ophthalmology,” he says. “With the microscope we have a 3-D view, limited to what we can see from the front of the eye. With ECP, you can go anywhere inside the eye that the endoscope can go, but instead of looking through the microscope you’re looking at a 2-D screen.”
“All of the MIGS procedures require some special skills,” adds Dr. Noecker, “but they’re all learnable.”
Financial Pros and Cons
“I’d say the Trabectome is probably the most expensive in terms of capital investment, at least in the short term,” says Dr. Noecker. “You have to buy the device and you have reusable costs, too. With ECP you have to buy the laser and endoscope, but you can use the endoscope for different applications such as retina and glaucoma procedures or manipulating inside the eye. The iStent has the smallest upfront costs; you need a goniolens and the device itself. You can order one and get reimbursed for it relatively quickly.”
Dr. Francis concurs. “In terms of costs, the iStent is on a use-by-use basis; you pay for it, you use it,” he says. “The Trabectome involves a capital expense for the unit itself and a per-use cost for the disposable handpiece. ECP also has an initial cost for the unit, and there are handpieces to purchase, but the handpieces are not disposable; you can generally get 15 to 20 uses out of each one.
“From the patient’s perspective, the MIGS procedures are similar in cost,” he adds. “The only significant difference occurs if the surgeon uses more than one iStent during cataract surgery, or if a patient wants an iStent when cataract surgery isn’t involved. The iStent is only approved for use with cataract surgery, with a single iStent. Outside of those parameters, the patient has to cover the additional cost.”
Dr. Noecker notes that in terms of reimbursement, ECP and the Trabectome have been around for a while. “Claims involving them are rarely rejected,” he points out. “They’re pretty much covered by commercial insurance and Medicare and Medicaid, although the Trabectome may be a little less likely to be universally reimbursed. The drawback with the iStent is that although it’s Medicare-reimbursable right now, it’s limited in terms of commercial coverage, and in my experience, it’s not covered by Medicaid. So you can’t do it on every patient you might want to do it on. The other restriction with the iStent is that it’s only approved and reimbursed in combination with cataract surgery, so you won’t get reimbursed for implanting it in a patient who has already had cataract surgery. You also may not be reimbursed if the patient has a type of glaucoma other than open-angle glaucoma.”
Taking the Plunge
Once you’ve decided to add MIGS to your armamentarium, surgeons offer these suggestions:
• Don’t under-prepare. “Make sure you do all of the certifying courses that are necessary,” says Dr. Samuelson. “Watch lots of videos of the procedure in question.”
• Practice visualizing before attempting the procedures. “With any of these new techniques it’s helpful to try pretending that you’re going to take the next step without actually doing so,” says Dr. Noecker. “With ECP, during a routine cataract surgery, inflate the eye and practice just looking at the ciliary processes with the endoscope. If you’ll be using the Trabectome or iStent, practice getting a pristine gonioscopic view so that won’t be an issue when you actually begin doing the procedure.”
• Practice gonioscopy following routine cataract surgery. Dr. Samuelson says it’s important to master intraoperative gonioscopy before taking on your first case. “Intraoperative gonioscopy is very different from gonioscopy in the office,” he points out. “They’re not even comparable—there’s more difference than similarity. Patient orientation is different; you have to change the orientation of the microscope; and you have to move the patient’s head. In the OR the patient is lying down and you’re doing something technical with the other hand while doing the gonioscopy. They really are different procedures.”
• A clear view is the top priority. Dr. Noecker notes that when using the Trabectome or iStent, mastering intraoperative gonioscopy and being able to clearly see the target tissue is crucial. “The biggest thing with the Trabectome or iStent is visualization,” he says. “In terms of mastering the technique, it really comes down to making sure you’re in the right spot. In some cases I’ll spend a few minutes making sure I can see what I want to see before commencing the MIGS procedure. Then, placing the stent or doing the Trabectome is relatively straightforward.
“Make sure you turn the patient’s head at least 45 degrees,” he continues. “You have to tilt the microscope, and you have to have the mirror on the eye and a really good view without striae. Basically, if you have a crystal-clear view, it’s going to be an easy procedure. If the view is marginal, it’s going to become a big struggle. There’s no point in hoping the view will improve during the procedure; visualization can only get worse.”
• Record your own cases. “That will allow you to watch and learn from your own mistakes and successes,” notes Dr. Samuelson.
Other Questions
MIGS procedures obviously don’t have to be performed at the same time as cataract surgery. What about using them as stand-alone procedures?
Dr. Samuelson notes that to some degree this is uncharted territory, especially with the iStent, although data is quickly being acquired from work completed in Armenia and elsewhere demonstrating the efficacy of the iStent as a stand-alone procedure. “I think there will be a role for these procedures outside of cataract surgery, but we need more data to help guide us with that,” he says. “Currently you can do ECP and Trabectome separately from cataract surgery, but the iStent is not approved for that.”
Another question is whether it makes sense to add two MIGS procedures to your repertoire instead of just one. Dr. Noecker sees the different modes of action used by different devices as an advantage in this area. “We’ve always done a little bit of combining procedures,” he notes. “There are times we put two glaucoma drainage devices such as an Ahmed and a Molteno in an eye, for example, because the patient has a really high pressure. MIGS procedures only lower pressure on average by maybe five points—but it’s a safe five points. Because the MIGS procedures use different approaches, we can always add a second MIGS procedure that lowers pressure via a different mechanism.
“For example, I often combine ECP and the iStent,” he says. “One works via the outflow system, the other works on the inflow system. That addition can get us another five points. This may get us close to the amount of pressure reduction we’d achieve with the riskier traditional procedures that would have caused a lot more bleeding and inflammation, swelling and temporary bad vision.”
Dr. Francis believes that all ophthalmologists who treat glaucoma, including general ophthalmologists, should have at least one MIGS procedure in their armamentarium, while glaucoma subspecialists should be versed in two, spanning two different modes of action. “For example, rather than being able to perform both Trabectome and iStent, which work through the same pathway, you might learn to use the Trabectome and ECP, or the iStent and one of the suprachoroidal shunts once they’re approved,” he says.
Dr. Noecker notes that although having more than one of these procedures in your armamentarium is an advantage, it isn’t necessary. “I have a lot of use for these procedures because all of my patients have glaucoma,” he says. “I do a half dozen of these cases every surgery day. I think a general surgeon would be happy mastering one, and that’s great. It’s much better to do one surgery really well than to take on three different surgeries and do them mediocrely—which is more likely to happen if you only get to do the surgery once or twice a month.”
The Future Looks Bright
There’s no question that the MIGS options bode well for the future of glaucoma treatment. “Many cataract surgeons treat a fair number of patients with glaucoma, but until now surgeons were resistant to combining cataract and glaucoma surgery because the glaucoma surgery could cause problems with the cataract surgery,” says Dr. Noecker. “Because the MIGS procedures increase the safety profile of doing combination surgery, I think it will increase the number of glaucoma surgeries that are done, which will help to lower people’s eye pressures safely and get them off of eye drops. In many cases, intervention will happen earlier in the disease. I think it will become the norm for any glaucoma patient to have something done at the time of cataract surgery, and that’s a good thing.”
Dr. Noecker adds that he anticipates a day when better information about the reason for high pressure inside a given eye will allow surgeons to decide which MIGS approach will be most helpful. “We’ll be able to tell that one patient has a problem with trabecular resistance, so we’ll use a device that bypasses the trabecular meshwork,” he says. “Or we may discover that the resistance to outflow is farther back, so the more efficacious solution is a suprachoroidal shunt.”
Dr. Francis, for one, sees little value in waiting any longer for such refinements. “Having one or more of the current procedures in your armamentarium will help you no matter what other things come down the pike in the future,” he says. “And in terms of waiting, how long are you going to wait? The iStent was just approved a year or two ago, but the Trabectome has been available for six years. At some point you have to adopt one of these procedures or you’ll just keep doing trabeculectomies and tube shunts as your primary surgeries.
“I think the time is now to adopt at least one of these procedures in your practice,” he concludes. “MIGS is not going away. Any given device or procedure may evolve over time, but this category and the theory behind it are here to stay. That’s why I recommend that ophthalmologists adopt at least one of the MIGS procedures.”
“This is a very exciting time in glaucoma, to say the least,” adds Dr. Noecker. “We had our medication revolution about a decade ago, going from two choices to multiple choices. That allowed us to customize medical therapy according to the patient’s risk profile. Now we’re seeing a similar revolution in glaucoma surgery. We can weigh the risk-benefit profile of each of the procedures, and just as we do with medical therapy, we can decide which procedure is most appropriate. We can also combine multiple procedures, if necessary. Now, saying a patient needs glaucoma surgery won’t just mean one thing.”
Dr. Samuelson agrees. “I think the MIGS options are going to keep improving,” he says. “It’s only going to get better from here.” REVIEW
Dr. Francis has consulted for Neomedix and Endo Optiks. Dr. Noecker is a consultant for Endo Optiks and has received research support from Glaukos. Dr. Samuelson has consulted for Glaukos and Endo Optiks. Dr. Berdahl is a consultant for Glaukos.