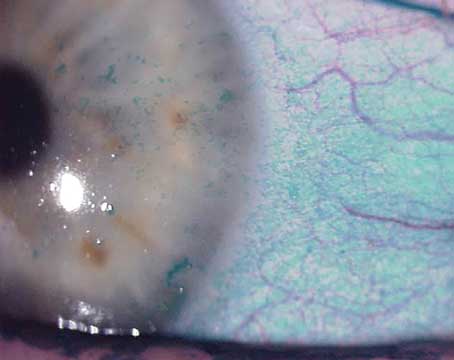
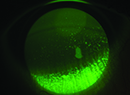
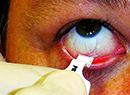
Dry-eye disease affects more than 20 million people in the United States alone, according to recent studies, with estimates ranging as high as 60 million worldwide. It can cause poor vision and chronic pain, and its incidence increases with age. Fortunately, researchers here and internationally are searching for new ways to treat dry eye.
This article will look at some of the potential dry-eye treatments that are currently in various stages of Phase II and III Food and Drug Administration trials, based on data from the FDA’s clinical trials website, and other non-pharmaceutical approaches.
SAR 1118
 |
In September, the company initiated enrollment in a Phase III study (OPUS-1). This study will assess the safety and efficacy of SAR 1118 for the treatment of dry-eye disease. Approximately 588 patients will be randomized to receive SAR 1118 5.0% ophthalmic solution or placebo twice daily for 12 weeks. The primary outcome measures include inferior corneal fluorescein staining, vision-related function subscale of the Ocular Surface Disease Index, and safety and tolerability. The company plans to complete the study in the first half of 2012.
The Phase II trial was a randomized, placebo-controlled, multicen-ter trial that included 230 patients with dry eye. In this study, SAR 1118 demonstrated dose-dependent significant improvements in inferior corneal staining over 12 weeks. A statistically significant increase in tear production and improvement in vision-related functions were seen as early as two weeks after initiation of treatment. SAR 1118 was well-tolerated, and no serious ocular adverse events were reported.
CF101
CF101 from Can-Fite BioPharma Ltd. (Petah-Tikva, Israel) is an A3 adenosine receptor agonist; the company is currently recruiting for a Phase III clinical trial. The randomized, double-masked trial will compare two doses of CF101 to placebo. The study will enroll approximately 240 patients at multiple centers, and patients will be treated for 24 weeks. Primary outcome measures are complete clearing of corneal staining by fluorescein and the nature and frequency of adverse events.
In an earlier Phase II study, CF101 was taken orally as monotherapy for 12 weeks.1 The study included 68 patients (35 in the placebo group and 33 in the treatment group) with moderate to severe dry eye. Patients were treated orally with either 1 mg of CF101 or matching vehicle-filled placebo twice daily for 12 weeks and were followed for two weeks after treatment ended.
There was a statistically significant increase in the proportion of patients who achieved more than a 25-percent improvement in corneal staining and in the clearance of corneal staining between the CF101-treated group and the placebo group. Patients treated with CF101 experienced a statistically significant improvement in the mean change from baseline at week 12 of corneal staining, tear breakup time and tear meniscus height. Additionally, CF101 was well-tolerated, and no serious adverse events were reported.
Sodium Hyaluronate
River Plate Biotechnology (Chapel Hill, N.C.), is currently recruiting for a Phase III clinical trial of sodium hyaluronate ophthalmic solution, 0.18% (Rejena, formerly Vismed). Alcon Laboratories has licensed the U.S. marketing rights to this product. The estimated enrollment in this study is 1,400 patients. Patients will receive either one or two drops of sodium hyaluronate ophthalmic solution 0.18% or vehicle in each eye three to six times daily as needed. The study began in July and is expected to be completed in June 2012.
Under the name Vismed, this product has been used in Europe as a viscoelastic lubricant eye drop. A 2010 study demonstrated the clinical efficacy of Rejena in the treatment of dry eye.2 This placebo-controlled study included 444 patients: 221 were randomized to the treatment group and 223 were randomized to the control group. Patients instilled one or two drops three to six times daily for 14 days. At day seven, the differences between the two groups in change from baseline for lissamine green staining score and global symptom frequency score were both statistically significant.
CMC, Glycerin & Polysorbate 80
Optometric Technology Group Ltd. (London), in collaboration with Allergan, is currently recruiting participants for a Phase III clinical trial of this formulation. The study began in March and has an estimated enrollment of 40 patients. Patients will receive one drop of the formulation in each eye three times daily for 30 days. The primary outcome measure of this study is tear-film evaporation rate.
Eye Drops New Platform (EDNP)
Another Phase III clinical trial sponsored by Allergan will compare two experimental formulations (EDNP-1 and EDNP-2) with a CMC sodium-based artificial tear (Refresh Tears). The study began in May, and its estimated enrollment is 297. Patients will be randomized to receive one of the three formulations at least twice daily for 90 days. Results are expected early in 2012.
Hydroxychloroquine
A study sponsored by the Assistance Publique-Hôpitaux de Paris in collaboration with Sanofi-Aventis (both located in Paris) is comparing hydroxychloroquine (Plaquenil) with placebo for the treatment of dry eye associated with Sjögren’s syndrome. This study is currently recruiting participants. The estimated enrollment for this study is 120. One group of patients will receive 200 mg/day of hydroxychloroquine for 24 weeks, and the other group will receive a placebo for 24 weeks. Response is defined by the improvement of 30 percent or more on at least two out of three of the following visual analog scales: most disabling dryness; fatigue; and pain.
EGP-437
EyeGate Pharmaceuticals (Wal-tham, Mass.) recently completed a Phase III study to assess the safety and efficacy of EGP-437 (dexamethasone phosphate formulated for ocular iontophoresis) using the EyeGate II iontophoresis system in patients with dry eye. The study included 198 patients. One group of patients was given a high dose of EGP-437 (6.5 mA-min at 2.5 mA), one group received a low dose of EGP-437 (4.0 mA-min at 1.5 mA), and one group received placebo (100 mM of sodium citrate buffer solution). This study was conducted to confirm and extend the results of the Phase II study. Phase III study results have not yet been reported.
The Phase II study was single-center, randomized, double-masked, and placebo-controlled; it included 105 patients with mild to moderate dry eye using the CAE model, which allows for standardized measurement of dry-eye signs and symptoms in the investigation of therapeutic agents. This study showed that, compared to placebo, ocular iontophoresis treatments with EGP-437 had both a rapid onset of action and long-term effectiveness.
FID 114657
Alcon has completed a Phase III trial evaluating FID 114657 for dry-eye symptom relief in patients with Sjögren’s syndrome. The study included 49 patients. All were given FID 114657 (ORB Preserved Ocular Emulsion) as needed throughout the day for four weeks. Study results have not yet been reported.
Diquafosol
Merck (Whitehouse Station, NJ) sponsored a Phase III trial to confirm the safety and efficacy of this solution, compared with placebo, for the treatment of dry-eye disease. The study included 490 patients. One group of patients received diquafosol tetrasodium ophthalmic solution, 2%, one to two drops in each eye four times daily for six weeks. A second group of patients received a placebo four times daily for six weeks. Study results have not yet been reported.
OPC-12759
Otsuka Pharmaceutical (Tokyo) sponsored a Phase III trial to compare OPC-12759 ophthalmic suspension with placebo for the treatment of dry eye. The study included 180 patients. One group of patients received OPC-12759 four times a day for four weeks, while a second group of patients received hyalein mini ophthalmic solution 0.1% six times a day for four weeks. Study results have not yet been reported.
Another Phase III trial has been conducted on the long-term use of this product. This study ended in January and included 153 patients who instilled OPC-12759 ophthalmic suspension four times a day for 52 weeks. Results for this study have also not been reported.
Emulsion-Type Artificial Tears
Milton Hom, OD, in collaboration with Allergan, is recruiting (by invitation only) for a Phase III trial of two emulsion-type artificial tears. The estimated enrollment in the study is 48, and the study is expected to wrap up in the next month or two.
Rituximab
University Hospital in Brest and the Ministry of Health in France are collaborating to study Rituximab. The study is ongoing, but is no longer recruiting participants. This multicenter, randomized, double-blind, placebo-controlled trial has 120 participants. Patients are receiving either Rituximab (mabthera) injection or NaCl 0.9% or glucose 5% (both placebos). The study is expected to conclude early next year.
Bromfenac
Ista Pharmaceuticals is conducting a dose-ranging study to evaluate the safety and efficacy of bromfenac ophthalmic solution in dry-eye disease. The study is ongoing, but is not recruiting participants. The estimated enrollment is 990 patients, and the study is expected to be completed by the end of the year. One group of patients is receiving bromfenac ophthalmic solution A, another group is receiving bromfenac ophthalmic solution B, and a third group is receiving a placebo comparator.
Omega-3 Essential Fatty Acid
The Dry Eye Assessment and Management: Feasibility Study, sponsored by Penny Asbell, MD, in collaboration with the National Eye Institute, is currently recruiting patients. The study began in January 2010, and the estimated enrollment is 35. Patients in the treatment group will take five capsules daily of omega-3 essential fatty acid for a total dose of 3 g, while patients in the placebo group will take five capsules containing 3 g of olive oil. The primary outcome measure is change on the OSDI during 90 ±14 days after initiation of the treatment regimen.
Acupuncture
The Korea Institute of Oriental Medicine recently completed a Phase III study to assess whether acupuncture is more effective than an artificial tear drop for the treatment of dry eye. The study included 150 patients. Patients in one group received 12 acupuncture treatments in four weeks. Seventeen acupuncture points were selected, and disposable acupuncture needles were inserted on these points. Acupuncture needles were twisted and retained for 20 minutes before being removed. Patients in the other group were given preservative-free, single-use Refresh Plus (CMC sodium, Allergan), and they used the artificial tear in both eyes at least once daily for four weeks. The primary outcome measure was OSDI. Results have not yet been posted.
In a previous study by this group, standardized acupuncture was compared to non-acupoint shallow penetration acupuncture for the treatment of dry eye. The study included 42 patients (21 in each group). Patients in the standardized acupuncture group underwent nine acupuncture treatment sessions (three times a week for three weeks). Seventeen acupuncture points were selected, and acupuncture needles were retained for 20 minutes. In the shallow penetration group, 17 sham points were selected, and acupuncture needles were also retained for 20 minutes. After three weeks of treatment, patients in the standardized acupuncture group had an OSDI of -17.61 ±15.61 compared to an OSDI of -17.20 ±18.81 in the non-acupoint shallow penetration group.
Many of these products in the pipeline have shown promising results in Phase II and III studies. Which, if any, will provide dry-eye patients with new treatment alternatives should be known in near future.
1. Avni I, Garzozi HJ, Barequet IS, et al. Treatment of dry eye syndrome with orally administered CF101: Data from a Phase II clinical trial. Ophthalmology 2010;117:1287-1293.
2. Vogel R, Crockett RS, Oden N, Laliberte TW, Molina L; Sodium Hyaluronate Ophthalmic Solution Study Group. Demonstration of efficacy in the treatment of dry eye disease with 0.18% sodium hyaluronate ophthalmic solution (Vismed, Rejena). Am J Ophthalmol. 2010;149:594-601.