The amount of time spent worrying about endophthalmitis prevention is very much out of proportion to its actual incidence—but for good reason: Miss a case of intraocular infection and vision could be lost within a day. Because of this, cataract surgeons have developed a number of methods that they agree help to avoid this dreaded complication. They include fastidious wound construction and iodine irrigation, as well as methods that are still in contention, such as intracameral antibiotics vs. frequent topical antibiotics. In this article, several surgeons from the
Prep and the Incision
Since the source of infection is often the patient's ocular surface, the lids, lashes and tear film,
"Make sure they don't have an eyelid condition that would predispose them toward having endophthalmitis," he says. "Check for underlying blepharitis, chronic infections or underlying surface disease. These need to be taken care of before surgery, either with lid hygiene or topical or oral antibiotics."
Samuel Masket, MD, president of the American Society of Cataract and Refractive Surgery, says that after underlying disease is taken care of, the first line of defense against infection is chemoprophylaxis: the use of Betadine or povidone iodine in patient preparation as well as the use of topical antibiotics prior to surgery. "Interestingly," he says, "while we have clear literature evidence that you can reduce the microbe counts on the surface with topical antibiotics,1 we don't yet have clear evidence that the use of topical antibiotics reduces the incidence of infection. But, it's logical that if you combine antibiotics as well as antiseptics, and you can reduce the microbial count, then you're going to greatly reduce the likelihood of infection."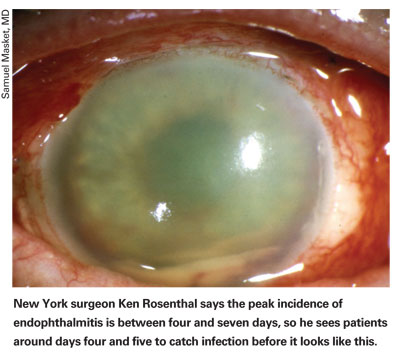
Though povidone iodine is an accepted preop mainstay, there isn't a consensus on which antibiotic to use. "But I think the majority of surgeons believe we should be using fourth-generation fluoroquinolones," says Dr. Masket.
Dr. Ta says that although the use of preop antibiotics is still controversial, as Dr. Masket points out, "I think it's a reasonable approach to use an antibiotic a few days before surgery," he avers, "because it's been demonstrated that use of an antibiotic three or four days prior to surgery is more effective at eliminating bacteria than just using the agent immediately before surgery."2
Surgeons say proper patient draping is crucial, though it may, unfortunately, receive short shrift sometimes.
He thinks that, theoretically, in the act of placing a drape, the surgeon may wipe his fingers along the edge of the unprotected lid margin and actually pick up bacteria on a glove. "So, for draping," he says, "I place the drape down with a slight overlap on the lids, without touching the lids or lashes with the fingers, and then place the speculum so it pulls the drape underneath, around the edge of the lid margin."
Dr. Masket offers additional ideas: "Typical strategies that work are to turn the patient's head toward the operative side," he says. "Some surgeons use a speculum that has an aspirating function, so there's actually suction on the field, and some surgeons prefer the use of drains or sponge drains placed in the cul-de-sac to clear fluid from the ocular surface."
Once the patient is prepped and draped, there is an ongoing debate about the next step: the incision.
In 2004, researchers at the
The researchers created self-sealing clear corneal wounds in cadaveric human and rabbit eyes, and controlled intraocular pressure with an infusion cannula. They used optical coherence tomography to image the incisions in real time as the researchers varied the IOP. Using an artificial anterior chamber, the surgeons applied India ink to the surface of the cadaver eyes with clear corneal incisions to detect the flow of surface fluid along the wound.
The researchers found that the wounds would change depending on the IOP. When pressures were high, the wound edges were apposed, and didn't tend to leak. But when IOPs were low, the edges tended to gape, starting at the internal aspect of the wound. Histological exam revealed ink particles in all the incisions for up the 75 percent of the length of the wound. The researchers concluded that transient reductions in IOP could result in poor wound apposition with clear corneal incisions, with the potential for fluid flow across the cornea and into the anterior chamber, with the corresponding risk of endophthalmitis.3
Randall Olson, MD, director of the
"It appears that cornea-scleral wounds are much more forgiving than corneal wounds," he says. "But forgiving is the operative word: Doing clear corneal incisions doesn't mean there has to be an increased risk of endophthalmitis. Some have seen it with these incisions, large groups have seen it, nationally we've seen it, Europe has seen it, but a lot of individuals haven't seen it … So, it's clear that the risk focuses on how forgiving the incision is. A sutured wound is more forgiving, as is cornea-scleral."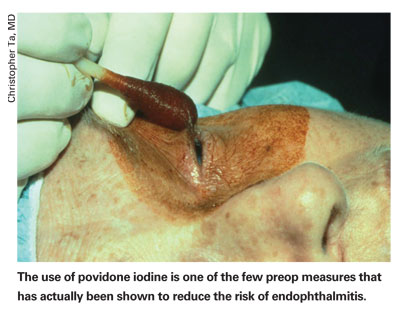
Dr. Olson says the key to ensuring that your incision isn't the weak link in the prophylaxis chain is having a keen eye for a marginal, rather than ideal, incision. "There are a few core principles you follow when looking for them," he explains. "First, if the incision isn't at least 2 mm long [in terms of depth into the eye] at any point along its length, you've got a marginal incision. Two millimeters is a pretty good figure. Often, people have a pretty good incision, but it's torn on one edge, where it's maybe only 1 or 1.5 mm. Sam Masket reported on a study at the American Academy of Ophthalmology in New Orleans that showed exactly that: With 1-mm incisions, they leak at virtually any IOP; at 1.5 mm, they would hold a bit; but at 2 mm, an incision isn't leaking. So, if you look at your incision and say, 'It's a little short on one side,' then you've got a marginal wound and should close it with a single 10-0 nylon suture.
"Another principle is if you look at the incision and it looks as if you stretched it and it looks unusual, or if it fishmouths—where it literally just pops open and stares at you—it's marginal and should get a suture. Another wound that deserves a suture is if, as you're trying to go through the stab incision, you have a Descemet's detachment and therefore don't have the endothelial pump to hold the wound together."
Dr. Olson says that, although he has a very low threshold for suturing a wound, he actually doesn't put a lot of sutures in. "I'll probably use a suture in 3 to 5 percent of cases," he says. "I'll usually take it out the next day if everything looks good, or will wait a week if I'm more concerned. Placing a suture turns a risky situation into a no-risk one." As evidence of the importance of a watertight wound, Dr. Olson recounts a study he took part in that was published in 2005. "In it, we looked at over 15,000 cases and were able to have enough cases of endophthalmitis, 27, to do a good analysis," he says. "We found that if you have a leaky wound the day after surgery, your odds of getting endophthalmitis go up 44 times."4
Dr. Olson says that study also taught him and his colleagues that, when you have a complication, either torn zonules or a broken capsule, the risk for endophthalmitis goes up 15 times. "For me, if I break a zonule or a capsule, that deserves a suture in order to not risk contamination," he says.
Dr. Masket echoes the need to make an appropriate seal and avoid stretching the tissue. "I think it's also important to establish eye pressure at normal levels," he says. "I measure it for every case with a tonometer, and then do an intraoperative Seidel test—fluorescein dye application to the incision area—to ensure it's not leaking." He recently completed a study that is to be published in the March issue of the Journal of Cataract and Refractive Surgery that indicates when the details of wound maintenance are followed, there's no evidence of wound leakage or hypotony in the first two to six hours after surgery, indicating the risk of infection is reduced.
Intracameral Antibiotics and the ESCRS Study
In the realm of infection prevention, you need only say "The ESCRS Study" and everyone will know what you're referring to. This controversial, landmark 2006 study of endophthalmitis prophylaxis from the European Society of Cataract and Refractive Surgery set out to compare perioperative levofloxacin drops to intracameral injection of the antibiotic cefuroxime for endophthalmitis prevention. The study began in 2003, and was partially masked, randomized, placebo-controlled and involved 23 surgery centers throughout
By the end of 2005, the study had follow-up on 13,698 patients, and showed such a strong beneficial effect from the use of the cefuroxime injections that the researchers agreed it would actually be unethical to continue the study and wait for all the follow-up before reporting their results.5 The incidence of endophthalmitis in the treatment groups not receiving the intracameral antibiotics was almost five times higher than those getting the injections. The use of perioperative levofloxacin drops was associated with a reduction in the observed incidence of endophthalmitis, but the researchers say the effect was smaller and not statistically significant. The preliminary results were published last year. The study's final results are still awaiting publication.
Many are impressed by the study's findings.
"Randomized, prospective studies that are large enough to prove a benefit are very difficult to conduct," says
The study has its critics, though, who question it on several points.
"I was struck in the preliminary report by what is not a very strong statistical difference between the study groups that would require the stopping of a study early," says Wiley Chambers, MD, deputy director of the U.S. Food and Drug Administration's Division of Anesthesia, Analgesic and Rheumatology Products. "We stop studies all the time but they have to reach a certain level of statistical significance before we do that; on rare occasions this is a p-value of 0.001 or less, or, more commonly, 0.0001 or less." The ESCRS study stopped at p < 0.002.
Peter Barry, FRSC, one of the ESCRS Endophthalmitis Study's administrators, says there was nothing wrong with how the study was conducted or terminated.
"I've heard a variety of inaccurate theories as to why we terminated recruitment early," says Dr. Barry. "But the basic reason it was terminated was the result that we found with our extraordinarily sophisticated information technology and statistical analysis system. The study's forms for participating patients were downloaded within days of completing a surgery from the Internet to the central office that was keeping all the records, so there was no interim delay. The data monitoring committee met every three months to look for trends, and in January 2006 discovered a trend that showed a clear beneficial treatment effect, but they were masked as to what treatment it was. The effect was determined to be of a magnitude that would require breaking the code, because we couldn't withhold the potential benefits we observed from the scientific community or patients in general." As to whether some groups of outliers could skew the results, Dr. Barry says he and his colleagues considered that. "As part of the statistical validation, we took out every center that had recruited more than 850 patients and ran the whole analysis again to see if there was a bias in favor of a particular institution or clinical center," he says. "And the results came out exactly the same."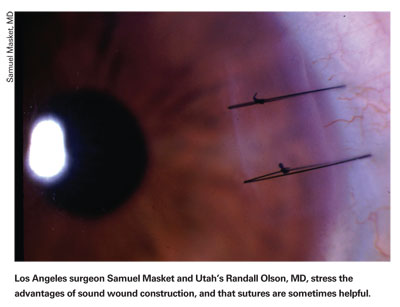
Dr. Olson says the ESCRS study proved definitely that intracameral cefuroxime is superior to no antibiotic in the first 24 hours after surgery, and for this he commends them. But he wonders why they didn't compare injection to the frequent topical use of fourth-generation fluoroquinolones on the day of surgery, since that's a widely used practice.
"That's a very good question," says Dr. Barry. "But, it can only be answered by another trial. Such a trial would be, perhaps, impossible, because I think it would be very difficult for any serious group investigating post-cataract surgery endophthalmitis to randomly allocate patients to any treatment that didn't involve intracameral antibiotics, given the results of our study. Also, if you're comparing intracameral antibiotic A, say Vigamox, vs. B, Cefuroxime, you'd need so many patients to show a meaningful difference that the study would be very expensive to conduct, and, perhaps, futile."
The study has also been criticized for an unusually high rate of endophthalmitis in the minimal treatment group (0.3 percent) compared to that found in previous studies. This group received only povidone iodine, intensive topical placebo drops and routine levofloxacin t.i.d. and q.i.d. for a week. Dr. Barry says several things may be at work here.
"First, most other endophthalmitis studies are retrospective, and all retrospective studies are prone to under-reporting," he says. "Two, people do not report and/or publish bad results, so I'd expect there are poor series of endophthalmitis out there that never got published; and three, I suspect that perhaps povidone iodine isn't as good as we all thought it might be. On its own, it led to an endophthalmitis rate of 0.3 percent."
The results of the study have some ophthalmologists pushing for safe, unit-doses of injectable antibiotic they could use with their surgeries, rather than having to try to mix them themselves and risk dilution errors. However, possibly because of the criticisms of the study, or perhaps because of how much a randomized study of unit-dose agents would cost, no company has stepped up to begin a large-scale trial of antibiotics packaged in unit-doses for injection.
Though no company has applied to the FDA for unit-dose injectable vials yet, the agency's Dr. Chambers still is interested to hear the ESCRS's final results.
"It's an interesting study," he says. "Why was it stopped early? Was the result just random chance or is it a real finding? I would hope whatever final results they eventually publish explain why."
The ESCRS's Dr. Barry promises that their final paper will reveal all. It's due out, he hopes, in March or April. "I think when Paper 3 [the final results] is published in the Journal of Cataract and Refractive Surgery," he says, "it will put all the critics soundly to bed."
1. Apt L, Isenberg SJ, Yoshimori R, Spierer A. Outpatient topical use of povidone-iodine in preparing the eye for surgery. Ophthalmology 1989;96:3:289-92.
2. Ta CN, Egbert PR, Singh K, Shriver EM, Blumenkranz MS, Mino De Kaspar H. Prospective randomized comparison of 3-day versus 1-hour preoperative ofloxacin prophylaxis for cataract surgery. Ophthalmology 2002;109:11:2036-40; discussion 2040-1.
3. McDonnell PJ, Taban M, Sarayba M, et al. Dynamic morphology of clear corneal cataract incisions. Ophthalmology 2003;110:12:2342-8.
4. Wallin T, Parker J, Jin Y, et al. Cohort study of 27 cases of endophthalmitis at a single institution. J Cataract Refract Surg 2005;31:4:735-41.
5. Barry P, Seal DV, Gettinby G, et al. ESCRS study of prophylaxis of postoperative endophthalmitis after cataract surgery: Preliminary report of principal results from a European multicenter study. J Cataract Refract Surg 2006;32:3:407-10.



