Corneal refractive correction continues to be extremely popular, despite pandemics and constantly expanding alternatives for addressing refractive error. But as with any popular procedure, the more individuals come into your office for treatment, the more likely you are to encounter cases in which you have to think twice about whether or not to proceed with the surgery.
Here, surgeons discuss how they make these decisions in four challenging situations: when the patient has herpes simplex; when the cornea is borderline thin; when the patient has rheumatoid arthritis; and when the patient has previously undergone radial keratotomy.
1: Patients with Herpes Simplex
“The typical scenario [in our office] is that the patient presents with a distant history of herpes simplex,” says Gregory D. Parkhurst, MD, Physician CEO of Parkhurst NuVision in San Antonio, Texas (https://sanantonio-lasik.com). “The question is, is the patient eligible for refractive surgery if the disease is quiet or dormant? Generally speaking, in this situation we have a conversation with the patient around informed consent. We talk about the potential risks, benefits and alternatives—both surgical and nonsurgical. That conversation usually includes a discussion of other criteria, including the patient’s age, health, refractive error and goals.
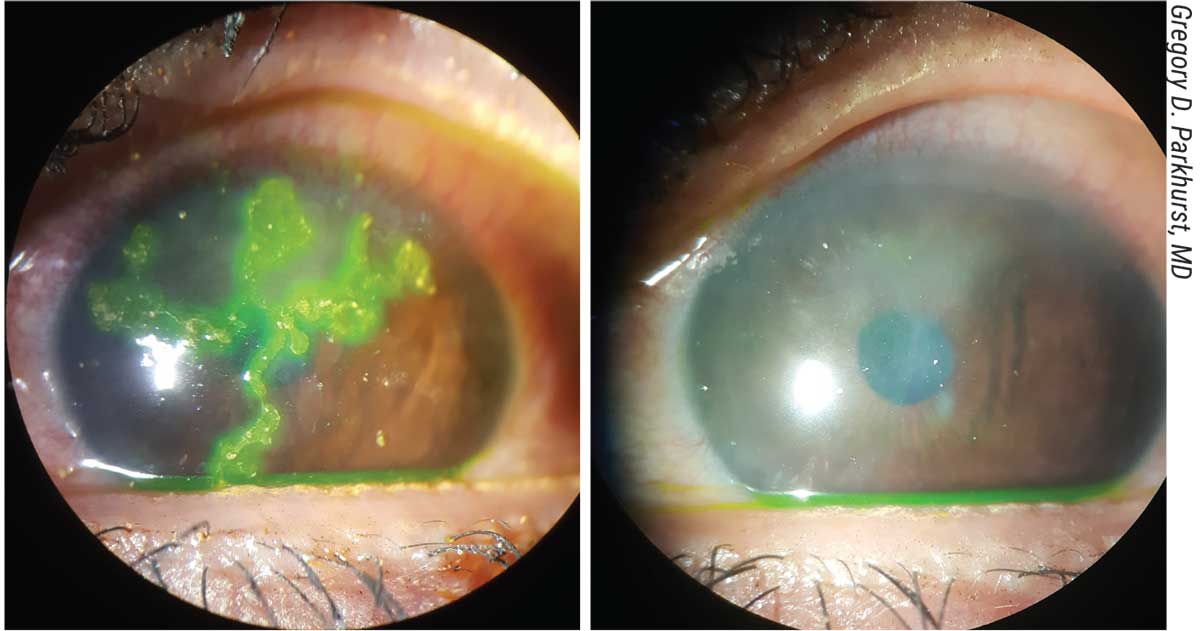 |
| An acute herpes simplex infection, illuminated with fluorescein (left), followed by late stromal scarring many months later. This patient is not a good candidate for LASIK. |
“In the case of corneal herpes, the disease is usually limited to the corneal stroma and/or epithelium,” he continues. “For that reason we often consider refractive procedures that would bypass the cornea. I’m not saying we’d rule out laser vision correction in every case; there might be some cases, depending on the severity of the disease and other factors, in which we’d consider performing a laser procedure on the cornea in a well-informed patient. But generally, our preference is to bypass the cornea and use another approach.
“One of the approaches we often consider with a younger myope is a phakic IOL,” he notes. “If we were going to do a phakic IOL in this patient, we’d likely put the patient on oral antivirals for a couple of weeks around the time of surgery, in the event that the surgical intervention incites some recurrence of the active condition—even if it’s an intraocular surgery like a phakic IOL.
“Of course, if a patient has an active infectious process—their eye is red and inflamed—they’d need to have that addressed first with appropriate therapy such as anti-viral drops, and sometimes antiviral oral medication,” Dr. Parkhurst notes. “We need to get past the acute phase before we think about any surgical intervention.
“In terms of timing, we don’t have any hard-and-fast rule about how long the eye has to be quiet before proceeding, but I’d want to see at least many months of a quiet eye, and I’d feel most comfortable waiting a year,” he notes. “This might be an option if the patient was limited to a single episode of herpes simplex keratitis. On the other hand, if the patient has a history of stromal recurrences, we’d want them to have long-term antiviral suppressive therapy. However, that would also give us a reason to avoid doing surgery on the cornea at all.
“In addition, the severity of the herpetic infection and its location are also considerations,” he notes. “Was it limited to the epithelium? Was there stromal involvement? If it was stromal, was it recurrent? The more the disease approached the severe side of that spectrum, the less likely I’d be to offer laser vision correction.”
Karl Stonecipher, MD, medical director of TLC Laser Eye Centers in Greensboro, North Carolina, and a clinical associate professor of ophthalmology at the University of North Carolina, says he’s never done LASIK on a patient who had active or sequestered herpes. “I also wouldn’t do PRK on a patient in that situation,” he says. “I don’t want to test those waters.
“I know certain people will try to ablate herpes scars with the patient on 500 mg or 1 gram of Valtrex twice a day,” he says. “If I were to do that, I’d definitely do it under systemic antiviral prophylaxis. However, that’s a therapeutic treatment, not a refractive treatment.
“If the patient has stromal herpetic keratitis but the cornea is clear, and I’m choosing between putting in an ICL or doing a laser vision correction with a UV light that might activate the virus, I’d be more likely to do the intraocular procedure,” he continues. “I think it would be a better option because you’re not lasering the cornea. The contradictions for an ICL include being pregnant, breastfeeding, younger than 21, older than 45, having a chronic disease that causes hormone fluctuations, using medications that cause vision changes, having wound-healing issues or having a poor endothelial cell count, but I don’t see herpes on that list.”
Dr. Stonecipher notes that he has no problem performing cataract surgery on a patient who’s had herpes. “We do a lot of intraocular lenses in those patients,” he says. “They have no other option because they have a cataract. We quiet their herpetic challenges with antivirals, and then treat them and keep them on antivirals for six months to a year postop. That’s one reason I’d feel more comfortable doing an ICL in a patient with herpetic eye disease. I’ve sent patients who needed a very low correction outside the country, to the Dominican Republic for example, where they have access to very low-powered ICLs.”
Dr. Stonecipher points out that performing LASIK on a patient with herpes is questionable from a medico-legal standpoint, because most of the laser systems have warning labels that say it’s an issue. “This might be the case because it’s possible that UV light can activate the virus,” he explains. “Various stresses have been suggested as virus triggers, including UV light. I don’t believe it’s been statistically proven that UV light triggers herpes; some studies have failed to find a connection, such as the Herpetic Eye Disease Study II, in which UV light exposure, contact lens wear and systemic infection weren’t found to be associated with recurrence of ocular herpes.1 On the other hand, many people have anecdotally noticed a connection in the clinic. So if a connection does exist, it’s not clear.”
Dr. Stonecipher adds that he’s seen patients develop herpes after LASIK. “They did well with antivirals,” he notes. “But the only time I’ll do laser correction in any type of patient with pre-existing herpes is as a therapeutic treatment when there are no other options available for that person—even if they’ve been quiet for a while.”
2: A Borderline Thin Cornea
“This is a common scenario,” says Dr. Parkhurst. “In these thin-cornea patients we have to decide whether the cornea is thick enough that we can perform safe and effective laser vision correction that’s going to work both in the short and long term, and yet won’t put the patient’s eye at future risk for rare processes like ectasia.
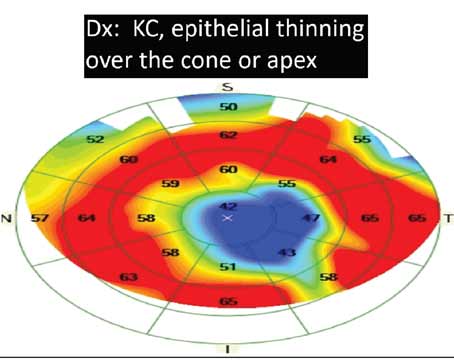
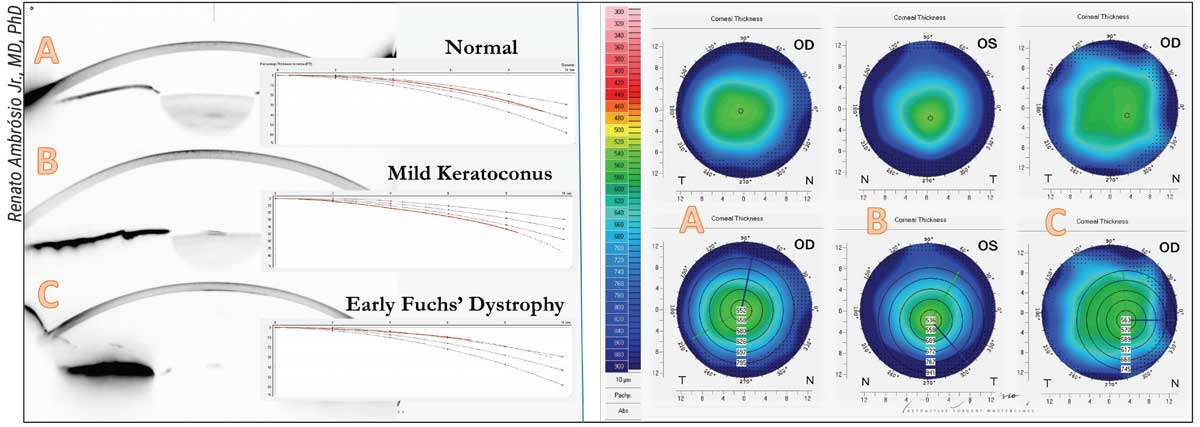 |
| Whether laser vision correction is appropriate for a given patient shouldn’t be decided solely on the basis of central corneal thickness, surgeons say. Above: Three cases with similar central thickness but different prognoses for LVC. A) A cornea presenting with a normal percentage of increased thickness moving outward from the thinnest point. B) A mild case of keratoconus—i.e., a thinned cornea. C) A case with moderate guttata considered to be subclinical edema and mild Fuchs’ Dystrophy. |
“I generally prefer to leave 300 µm in the stromal bed if I’m going to do either LASIK or SMILE,” he continues. “Again, it depends on the magnitude of the refractive error and the corneal thickness. For example, if I had a patient with a very thin cornea that was completely normal, with a very low level of myopia—let’s say -1 D—that patient may very well be eligible for laser vision correction. But if there’s not sufficient corneal thickness relative to the magnitude of myopia needing correction, we’d lean toward a phakic IOL. A phakic IOL will let us correct even extreme amounts of myopia without removing any tissue at all.
“When talking about this with patients,” he adds, “I’ll often explain that the ideal scenario is to start with thin glasses and a thick cornea, because then there’s plenty of tissue for making the correction. Patients who have the opposite—thick glasses and thin corneas—aren’t ideal candidates for correcting the entire refractive error with laser vision correction. Of course, that’s true whether the option is LASIK, PRK or SMILE.”
“We consider a cornea to be thin if the mean thickness is two standard deviations below the norm,” explains Renato Ambrósio Jr., MD, PhD, president of the International Society of Refractive Surgery and director of refractive surgery at the Instituto De Olhos Renato Ambrósio/Visarerio Refracta Personal Laser in Rio De Janeiro, “Based on a study published in the Journal of Cataract and Refractive Surgery in 2003, the CCT is about 555 µm, ±35 µm.2 When you go to the thinnest point, it’s around 540 ±32 µm, which can vary based on different populations.”
If you’ve decided to proceed with LASIK, does the tool you use to make the flap matter? “We pretty much always use a femtosecond laser if we’re going to be performing stromal surgery, whether we’re making a LASIK flap or performing SMILE,” notes Dr. Parkhurst. “One of the reasons is that a femtosecond laser gives us precision in our flap thickness to the level of a few microns. With a mechanical microkeratome, there’s a little more deviation in the planned vs. the resulting flap thickness. I believe the femtosecond laser adds a layer of precision and safety to our corneal laser vision correction.”
Dr. Stonecipher says he prefers using a femtosecond laser to create the flap whether the cornea is thin or not. “The femtosecond laser is much more accurate in terms of standard deviation,” he notes. “I haven’t used a microkeratome in quite some time.”
Beyond Corneal Thickness
Dr. Ambrósio notes that the issue isn’t just whether a cornea is thick or thin, but whether it’s stable or unstable. “When I encounter a thin cornea, I ask if this is a stable thin cornea or a thinned, unstable cornea,” he says. “Thin is different from thinned—and the same concept applies to a thick vs. a thickened cornea. Based on single-point thickness, even a relatively thick cornea can be unstable. That’s why refractive surgeons need to understand the importance of performing advanced complementary exams when evaluating a patient for laser vision correction.
“The idea is to go ‘beyond but not over’ front-surface topography,” he continues. “Data from tomography and biomechanics can help determine whether the patient is suitable for a laser vision correction procedure and help us customize our planning to increase safety and efficiency. Furthermore, it’s important to detect mild keratoconus before proceeding with any refractive surgery, but we should again go ‘beyond but not over’ this towards characterizing the intrinsic biomechanical susceptibility of the cornea to undergoing ectatic progression. Considering such a concept, we can estimate how much impact the cornea may have from the laser vision correction procedure and still be stable over time.
“Twenty years ago when I did my fellowships, ultrasound-measured central corneal thickness and Placido-disk-based topography were the mandatory exams,” he recalls. “At that time, I participated in a retrospective study that found that 0.9 percent of patients who came in as candidates for laser vision correction had abnormalities detected by axial topography curvature maps, while more than half of such cases had 20/20 or better distance corrected vision and normal slit-lamp biomicroscopy. This study demonstrated the fundamental need for corneal topography and CCT; nevertheless, we have to go beyond such evaluation in order to enhance both safety and efficiency.
“Front-surface corneal topography is still a mandatory exam because it provides data related to the ocular surface and the stability of the tear film,” he says. “However, some corneas with high susceptibility to ectasia won’t yet show changes on the front surface. This is why a 3-D representation of the cornea using tomography is crucial. From the 3-D evaluation of the cornea, we can derive front surface elevation, back surface elevation and the thickness profile.
“This data is summarized in the Pentacam Belin/Ambrósio Enhanced Ectasia display (BAD),” he points out. “The BAD was designed to facilitate clinical interpretation using the enhanced elevation concept, originated by Michael Belin, MD, and the relational corneal thickness spatial profile that I developed. Because of the plethora of data, machine-derived metrics such as ‘deviation from normality’ on the BAD have gained momentum.
“Besides performing tomography with Scheimpflug technology,” he adds, “OCT can provide layered or segmental evaluation for epithelial thickness mapping, an idea that was originally pioneered by Dan Reinstein, MD, using high-frequency ultrasound.”
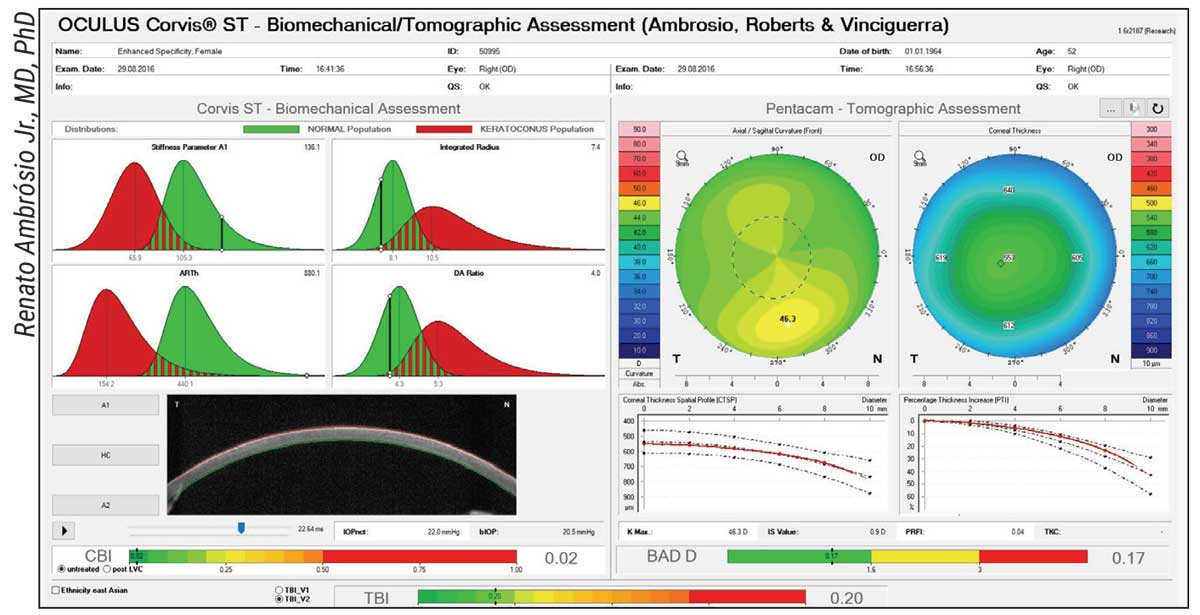 |
| A patient in which the enhanced tomographic and biomechanical assessment enhanced specificity, considering the mild inferior steepening. This patient had successful LASIK in both eyes in 2016 with documented stability in November 2021. |
Corneal Biomechanics
Prof. Ambrósio also considers it essential to investigate the biomechanical status of the patient's cornea. “Biomechanics can tell us a lot about the possible future state of the cornea,” he says. “I believe any cornea can progress to ectasia, which is a biomechanical failure of the corneal stroma to resist the stresses caused by IOP, extraocular muscles and eyelids. That’s why I think the issue isn’t just detecting existing mild keratoconus, but characterizing the individual patient’s risk for ectasia progression.
“At first, we evaluated the biomechanical status of the cornea with the ORA from Reichert; now we can also use the Corvis ST from Oculus,” he explains. “To the best of my knowledge, the Corvis ST is approved by the FDA only for non-contact tonometry and pachymetry, while the software capable of analyzing corneal biomechanical properties using the deformation corneal response is available internationally. This approach includes the Corvis corneal biomechanical Index, or CBI,3 and also integrates with the Pentacam to calculate the Tomographic Biomechanical Index, or TBI,4 which is available in the Ambrósio-Roberts & Vinciguerra Display, and uses artificial intelligence to improve the detection of patients who are at higher risk for postop ectasia. I’m very grateful and honored to be among the ones who have developed and refined the TBI.”
Prof. Ambrósio notes that the ultimate goal of the preoperative evaluation of candidates for refractive surgery is to understand the inherent properties of the patient’s cornea and refractive error, so he can determine how to modify them safely and efficiently. “This doesn’t just mean biomechanical properties,” he notes. “It also means the shape of the cornea and eventually its optical properties as well,” he says. “This will help us evaluate whether the patient is a good candidate for surface ablation, SMILE or LASIK, and it will help us customize the cap or flap geometry. It will also help us determine if the patient is a better candidate for phakic IOL implantation or refractive cataract surgery, to improve safety and also the refractive outcome.”
Dr. Ambrósio admits that evaluating the cornea meticulously may not eliminate risks, but can minimize them. “For example, the risk of ectasia is significantly reduced, although we still have cases with unexplained causes,” he says. “Meanwhile, when we enhance our ability to evaluate patients, we’re not only increasing sensitivity for detecting patients at risk, we’re also enhancing specificity. That means we’ll find some borderline candidates that may be safely operated on, as long as we do proper measurements to determine whether the corneal procedure could weaken the patient’s cornea.”
What about genetic testing? “Many doctors are doing genetic testing of patients with borderline thin corneas,” Dr. Stonecipher points out. “Those tests are now available through Avellino. It’s a good precaution to check their keratoconic status with these genetic tests. That data can give you some statistical elements to discuss with the patient.”
“We know there are tests that can screen for genes associated with a risk for keratoconus or ectasia, but I don’t think those tests are beneficial if a cornea is just thin,” says Dr. Parkhurst. “Thin but normally shaped corneas haven’t been shown to have a high risk of going on to develop keratoconus. It’s an abnormal shape that’s the bigger concern. If a thick cornea has an abnormal shape, that’s more likely to progress to keratoconus than a thin cornea with a normal shape.”
LASIK with Cross-linking?
“Today, many of my colleagues feel more comfortable doing laser vision correction on relatively thin corneas because they do it in combination with cross-linking,” Dr. Stonecipher notes. “Many surgeons outside the United States will cross-link a patient and then feel totally comfortable doing laser vision correction. John Kanellopoulos, MD, and Rohit Shetty, MD, have published multiple papers showing that when dealing with irregular topography, like that seen in a keratoconus patient, trying to improve the patient’s vision by cross-linking with laser vision correction is an option.”5,6
“Cross-linking is a great procedure,” says Prof. Ambrósio. “I believe Prof. Seiler and his group deserve enormous credit for developing it. However, the idea of using it as a ‘green card’ for elective refractive surgery in borderline corneas doesn’t resonate with me. Cross-linking has opened a new horizon for the treatment of corneal ectatic diseases, but using it to prevent possible future ectasia isn’t a good idea. If a patient can undergo LVC, we don’t need to do cross-linking.
“A therapeutic procedure designed to rehabilitate vision shouldn’t be confused with an elective procedure,” he points out. “Cross-linking with a topo-guided surface ablation to help a patient with moderate to advanced keratoconus as an alternative for keratoplasty makes sense. However, the improvement that results from such a treatment, which could be a great result for the keratoconus patient, would be a nightmare outcome for an elective laser vision correction patient. The point is that elective refractive surgery aims for excellent uncorrected vision, while therapeutic procedures for vision rehabilitation aim for distance-corrected vision.”
Dr. Parkhurst says that if a patient has mild keratoconus, he might perform corneal cross-linking to stabilize the condition. “Then we’ll do a topo-guided PRK to correct the refractive error and normalize the shape of the cornea,” he explains. “Generally speaking, we’re not advocates for doing cross-linking prophylactically in a cornea undergoing laser vision correction based on corneal thickness alone. If a cornea is borderline for LASIK, we’ll generally opt for a phakic IOL.”
Dr. Parkhurst explains that it’s not a question of risk. “I don’t think cross-linking is risky for the patient,” he says. “I just don’t think it’s been shown to have benefit when used prophylactically in a normal cornea. If you only operate on normal corneas and don’t do extreme refractive error treatment, it’s not necessary to cross-link. We reserve cross-linking for abnormally shaped corneas.”
“We must remember that cross-linking may alter the predictability of future disease,” Prof. Ambrósio adds. “In fact, there have been reports of ectasia after LASIK combined with prophylactic cross-linking.”7
3. Rheumatoid Arthritis Patients
“We’ve treated patients with systemic immune deficiencies, systemic autoimmune diseases such as HIV, and other riskier categories such as juvenile rheumatoid arthritis,” notes Dr. Stonecipher. “With all the immunosuppressants available today we can collaborate with the patient’s rheumatologist. If the rheumatologist feels that the patient’s condition is controlled, whether it’s HIV or one of the autoimmune diseases, we’ll consider that patient to be eligible.
“Of course, we consider all of the other relevant parameters as well,” he adds. “We make sure the patient has a stable refractive error and a typical, normal cornea. The biggest issue is dry eyes, because many of these conditions are associated with a dry-eye problem. We want to make sure their dry eyes are in check, and that they have a good tear film.”
Dr. Parkhurst says he would potentially do LASIK on a patient with rheumatoid arthritis, with good informed consent. “Some peer-reviewed publications have suggested that laser vision correction can be done safely in the setting of autoimmune disease,” he points out. “But of course, this is off-label. None of the FDA-approved excimer lasers have an indication for use in the setting of autoimmune disease.”
Dr. Parkhurst notes that the healing response in these patients can be less predictable. “When performing LASIK or PRK, you’re anticipating that the patient’s cornea will have a predictable healing response to a set dose of excimer laser,” he says. “A patient with a known autoimmune disorder may under-respond or over-respond to the dose of laser you use. For that reason, as in the other situations, we’d consider bypassing the cornea to correct the refractive error with a phakic IOL or possibly with a refractive lens exchange procedure. That way we don’t have to depend on the corneal healing response to get the therapeutic effect we’re looking for.
“The final decision about whether to proceed comes down to many factors,” he says. “What’s the patient’s age? How well-controlled is the autoimmune disease? Is the rheumatologist involved? Is it a small or large refractive error? What are the patient’s alternatives? Is this related to addressing the patient’s condition? For example, is the patient having chronic infections from contact lens abuse, and this is seen as a way to reduce the infection risk? The bottom line is that I wouldn’t rule out an autoimmune disease patient in every single case, but it’s definitely an important consideration in the decision process.”
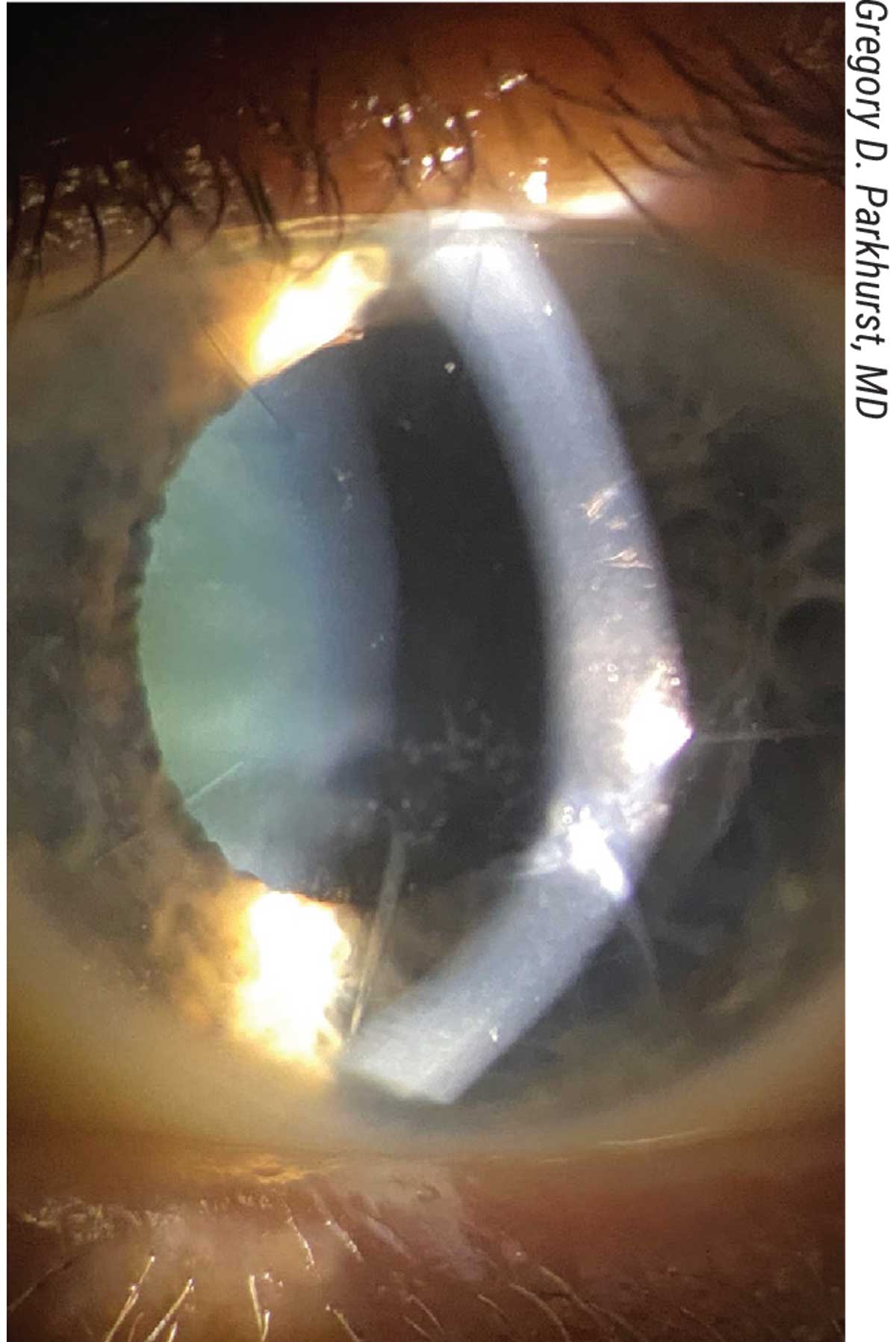 |
| A fairly typical presentation of an eight-cut radial keratotomy with early cataract. This would be best addressed by cataract surgery, rather than by treating hyperopia on the cornea with laser vision correction. |
4. Prior Radial Keratotomy
“Seeing patients who’ve had previous radial keratotomy is pretty common,” notes Dr. Parkhurst. “We see RK patients monthly, sometimes weekly. They don’t often present asking for LASIK; more often they present with vision distortion and blurring, wondering if there’s anything we can do to help them see better.
“By definition these patients are an older demographic,” he continues. “RK hasn’t been done much for 20 to 25 years, so these patients are usually in their 60s or older. Often, they were myopic before the RK but now they have hyperopia; depending on how much RK was done, they can have pretty high hyperopia, sometimes greater than +5 D. Of course, that’s not a refractive error or a cornea that would inspire us to suggest laser vision correction.
“In this scenario we almost always recommend a lenticular refractive surgery approach,” he says. “However, we don’t usually recommend a phakic IOL; usually we recommend a refractive lens exchange, or even cataract surgery. Many of these patients present with cataract, and we can cure both the cataract and the hyperopia by removing the natural lens and replacing it with an IOL.
“That’s not to say that corneal surgery is off the table,” he continues. “In certain circumstances I’d consider a laser vision correction approach, but it’s not routinely my first recommendation. Let’s say we have an atypical RK patient who isn’t
a +5 D hyperope. Maybe they’re
-1 D, and they don’t have a cataract. In that scenario I might consider doing a PRK to correct the -1 D. However, that’s not a typical RK patient. So it’s not that I’m ruling out laser vision correction because of their RK; it’s that when this factor is present, there are usually other factors that lead us toward treating the refractive error in a different way.”
Dr. Stonecipher says he stopped making flaps in post-RK patients in early 2002. “We’ve just done correction on the surface for the past 20 years,” he says. “Recently, when the T-CAT procedure was approved, we started doing topographic treatments on those patients with myopia and astigmatism (hyperopia with astigmatism isn’t approved). Most of these patients have higher-order aberrations related to their topography, so a topography-guided treatment is therapeutic as well as vision-correcting. Working on the corneal surface is a much better option.
“In the old days we did do LASIK in some of those post-RK patients,” he recalls. “We did it under a flap made with a microkeratome. The challenges we had were epithelial ingrowth and poor wound healing, and sometimes the incisions would split when making the flap. When using the femtosecond laser, the bubbles would often track the RK incisions and cause other issues.
“Typically, when we do enhancements on those patients, I look at the root-mean-square of their higher-order aberrations, or RMSH, and try to determine it that’s 0.45 or less,” explains. “In many cases, if they have a lower RMSH, I’ll do a trans-epithelial PRK where I laser through the epithelium and treat the refractive error. But if the RMSH is higher than 0.45, I’m more inclined to do laser vision correction using topography-guided profiles. That way, not only am I enlarging the older optical zone, which is typically small in RK patients, I’m making the patient see qualitatively and quantitatively better. I’m not only getting rid of their refractive error, I’m also getting rid of some of their halos, glare and starbursts related to the RK treatment.”
Dr. Parkhurst says that if he were to proceed with laser vision correction in a young RK patient with a clear lens and low-level myopia, he’d usually stick to PRK and avoid LASIK and SMILE because of the vertical incisions in the cornea. “The incisions would bisect the plane of the lenticule in the case of SMILE, and the flap in LASIK,” he points out. “Yes, it’s possible to do LASIK, despite this; but a PRK to treat the -1 D would be very effective, and you wouldn’t have to worry about possibly putting the cornea back together.”
Dr. Parkhurst notes that although an eye that’s had RK can be challenging in terms of calculating IOL lens power, today’s IOL-power formulas are actually quite good at predicting the power that will correct most of the refractive error. “At the least, this type of procedure will dramatically reduce the patient’s hyperopia,” he points out. “We’ve also found that using a wavefront aberrometry device like the ORA during the surgery improves our accuracy, even beyond using the latest formulae. And more recently, we’ve had a lot of luck using the light-adjustable lens. So in some cases we remove the cataract and replace it with a light-adjustable lens. Then we have the ability to fine-tune the refractive result in the office.”
Dr. Parkhurst adds that RK corneas often exhibit an extended range of focus. “These corneas are effectively multifocal,” he says. “Because of that, it’s often surprising how much visual range a patient with prior RK gets, even with a monofocal IOL. These patients often have a really great result in terms of addressing their hyperopia, astigmatism and even their presbyopia.”
A Few General Considerations
These pearls may apply to any situation in which a patient asking about LASIK may not be an ideal candidate:
• Always consider an intraocular alternative. “You have to look at intraocular procedures as an option,” notes Dr. Stonecipher. “Today, the designs of our ICLs and IOLs have gotten better, and some of these patients are better served by an ICL or a refractive lensectomy, depending on their age, occupation and expected outcome. In the early days, talking about refractive lensectomy was considered unacceptable. Now it’s mainstream.”
• Consider the patient’s occupation. Dr. Stonecipher says that when he’s considering the risk of a procedure, he takes into account what the patient does for a living. “In many states you must have a certain level of uncorrected vision to become a police officer or firefighter,” he points out. “That makes sense—you don’t want to go into a burning building and lose your glasses or have them fog up. So that’s part of the informed consent. If a patient says, ‘I’m thinking of becoming a police officer,’ and he or she is educated and understands the risks related to their condition—whether it’s a thin cornea, or something else—we definitely talk to the patient about it.”
• Consider what outcome parameters the patient is willing to accept. “When you’re managing a patient with a potentially problematic condition, you have to look at what the patient’s expectations are and what the patient is willing to give up,” Dr. Stonecipher notes. “For example, a patient might need a large refractive correction but have a thinner cornea. A patient like that might be happy with just being less nearsighted and still wearing glasses when we’re done. Would we operate on a patient like that? Absolutely, if they understand and are well-informed. Of course, if the cornea is thin and we’re concerned about ectasia, we must discuss the possibility of additional interventions such as cross-linking.
“I operated on a patient last week who was outside the normal parameters,” he continues. “This patient was a -12 D case. I said, ‘Look, this is what we can expect. I can’t do your full correction. I’ll do the best I can; we’ll do a tissue-sparing LASIK, which means we do basic wavefront-optimized LASIK.’ He said, ‘If you just make my glasses thinner, I’ll be happy. I’ve looked at ICLs, and I don’t want one of those in my eye. I’ve looked at the other options, too. I know I may still have to wear glasses, but I want to be able to get out of my house if it’s on fire.’ We had that conversation. He’s now about -0.75 to -0.5 D, and he’s incredibly happy. So, I think if a patient understands the risks and caveats and probable outcome and can verbally relate that to me, it’s OK to proceed.
“These patients take a little more time,” he adds. “They need to understand the risks and what their options are, so you’ll need to have more than one conversation with them. I also have other members of the staff spend time with them to make sure the patient’s expectations are in line.”
• Educating the patient is essential. “Patient education is of utmost importance,” says Dr. Ambrósio. “We, as refractive surgeons, have to promote refractive surgery and practice it with excellence. However, explaining each candidate’s options for elective refractive procedures doesn’t mean convincing the patient to proceed; it just means explaining the limitations and possible risks and highlighting the benefits.
“Refractive surgery procedures are among the ones with the highest levels of patient satisfaction,” adds Dr. Ambrósio. “Nevertheless, the premise of underpromising and overdelivering will always be the strategy for patient care.”
• Don’t confuse refractive surgery with laser vision correction. Prof. Ambrósio points out that it’s important to remember that refractive surgery is much more than corneal laser vision correction. “Laser vision correction is an elective procedure meant to improve the ability of the patient to functionally see without refractive corrections such as glasses or contact lenses,” he says. “Refractive surgery, including phakic IOLs and refractive cataract surgery, goes beyond laser vision correction on the cornea. These should be considered as part of our armamentarium for refractive surgery. Furthermore, refractive surgery technologies may be used for therapeutic procedures, aiming for visual rehabilitation. While every refractive surgeon has to be conscious of his ability to help patients with corneal diseases such as keratoconus, there’s a fundamental need to differentiate the goals from elective and therapeutic refractive procedures.
“Finally,” Dr Ambrósio adds, “the mindset of the refractive surgeon should go towards individualized refractive planning that will meet the needs and characteristics of each patient.”
Dr. Parkhurst is a consultant for Alcon, Johnson and Johnson Vision, Staar and Carl Zeiss. Dr. Stonecipher is a consultant for Alcon, Bausch + Lomb, Johnson and Johnson Vision, Carl Zeiss and Staar. Dr. Ambrósio is a consultant for Oculus, Alcon, Zeiss and Mediphacos.
1. Wilhelmus KR, Dawson CR, Barron BA, et al. Risk factors for herpes simplex virus epithelial keratitis recurring during treatment of stromal keratitis or iridocyclitis. Herpetic Eye Disease Study Group. Br J Ophthalmol 1996;80:11:969-972.
2. Ambrósio Jr., R, Klyce, SD, Wilson, SE. Corneal topographic and pachymetric screening of keratorefractive patients. J Refract Surg 2003;19:1:24–29.
3. Vinciguerra R, Ambrósio Jr R, Eisheikh A, et al. Detection of keratoconus with a new biomechanical index. J Refract Surg 2016;32:12:803-810.
4. Ambrósio Jr R, Lopes BT, Faria-Correia F, et al. Integration of Scheimpflug-based corneal tomography and biomechanical assessments for enhancing ectasia detection. J Refract Surg 2017;33:7:434-443.
5. Shetty R, D'Souza S, Srivastava S, Ashwini R. Topography-guided custom ablation treatment for treatment of keratoconus. Indian J Ophthalmol 2013;61:8:445-450.
6. Kanellopoulos AJ. Initial outcomes with customized myopic LASIK, guided by automated ray tracing optimization: A novel technique. Clin Ophthalmol 2020;14:3955-3963. Published 2020 Nov 17.
7. Taneri S, Kiessler S, Rost A, Dick HB. Corneal ectasia after LASIK combined with prophylactic corneal cross-linking. J Refract Surg 2017;33:1:50-52.