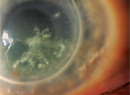
The past two decades have seen a series of microbial “epidemics” due to endemic protozoans in the genus Acanthamoeba. In the eye, these organisms can cause a keratitis which is difficult to diagnose, resistant to treatment and has the potential to be sight-threatening. While the infection caused by Acanthamoeba is often associated with contact lens use,1 particularly in Western countries, a retrospective assessment of outbreaks suggests that there are a number of factors involved in the epidemiology of Acanthamoeba keratitis. In this month’s column, we’ll take a look at recent AK outbreaks, consider the relative importance of various factors implicated in these outbreaks, and discuss the latest approaches to AK diagnosis and treatment.
Acanthamoeba is one of the more abundant protozoa on earth.2 Members of the genus Acanthamoeba have been isolated from soil, treated and untreated tap water, swimming pools, hot tubs and numerous other environments.3 The protozoa’s life cycle consists of an active, feeding trophozoite phase and a dormant, cyst phase. Formation of the cysts is activated by any of a number of unfavorable conditions including extremes of temperatures, pH or low humidity.4 Surveys of water supplies and other environmental sources have identified several dozen species, but among these is one group (sometimes referred to as T4), which includes three or four species particularly associated with keratitis in humans. While it’s not unusual for individuals to exhibit Acanthamoeba-specific antibodies, it’s not clear why the rates of infection are so low, given the prevalence of the organism in the environment. Cases of central nervous system Acanthamoeba-mediated encephalitis, which are often fatal, appear to be the work of opportunistic pathogens as they occur primarily in immune-compromised patients.3 In contrast, keratitis from an Acanthamoeba infection typically occurs in immune-competent individuals, so the precise mechanisms underlying Acanthamoeba virulence are unclear.2-4
Sources and Modes of Infection
AK was first documented in the United States in the early 1970s, and was traced back to ocular exposure to contaminated water. Information regarding the disease remained sparse, but the suggestion that the disease may be associated with contact lens use was established in the 1980s.6 Initial studies blamed increased incidence on the use of “homemade” saline solutions by contact lens wearers; however, infection was later observed among lens wearers using store-bought multipurpose solutions. Most of these solutions are effective in killing Acanthamoeba trophozoites, but don’t effectively kill cysts.5
Several spikes in the incidence of AK in the United States have been reported in the past two decades. The first outbreak was seen between 1985 and 1995 in Iowa towns served by the University of Iowa Hospitals and Clinics.7 This increase was thought to be associated with flooding and subsequent water supply contamination.
A second spike in cases was reported between 2003 and 2006; this included significant increases in multiple geographic areas including Chicago, San Francisco, Boston and Philadelphia.8,9 In Chicago, 63 reported cases in a period from 2004 to 2006 was greater than 10 times the rate previously seen in that area. Epidemiological research found that around half of all AK patients in the Chicago area outbreak had been using the same brand of a “no-rub” contact lens solution (Complete Moisture plus Multi-Purpose; AMO) for cleaning and storing their contacts.9 This finding led to a Food and Drug Administration investigation and a voluntary recall by AMO. Subsequent studies have shown that contact solutions employing the “no-rub” approach to disinfection have minimal efficacy for killing Acanthamoeba, particularly when compared to hydrogen peroxide-based disinfection solutions.10,11 An FDA workshop in 2009 recommended that, despite the low prevalence of AK, lens solution testing should include measurements of efficacy against Acanthamoeba.12
While the annual number of AK cases in the United States is certainly low relative to other types of ocular infections, historical approximations of ~250 cases per year are likely to be underestimates, based upon the difficulty of diagnosis and similarity to other forms of keratitis.9 Acanthamoeba keratitis occurs at higher rates, and is not so closely associated with contact lens use in nations or regions where reliable water supplies are lacking. In these areas, the combination of poor water quality and inadequate medical resources also leads to poorer final visual outcomes of AK infections.13
Beyond Contact Lenses
Following the outbreaks in Chicago, the Centers for Disease Control and Prevention set up an informal network of hospitals, practitioners and public health officials to monitor the prevalence of the disease, as there were some doubts about actual versus reported rates of infection. Despite studies that showed a strong correlation between the AMO solution and the most recent outbreak, rates of reported AK did not decline following voluntary withdrawal of the solution from the market. This point is highlighted in a recent review article, which states that while the “role of contact lens solutions should not be minimized, it has become clear that other, more global risk factors likely underlie the most recent Acanthamoeba keratitis outbreaks.”14
At the same time, a number of studies showed that regardless of sources, Acanthamoeba were common contaminants of home water systems both in the United States and abroad.15-18 In a recently published report, ~50 percent of the homes screened in an Ohio study were positive for Acanthamoeba.16 Prevalence of the organism in community water supplies may also be a factor in the rates of disease: In parts of the United Kingdom where AK incidence is 15 times that of the United States, infrequently flushed-out rooftop tanks serve as holding tanks for public water supplies.18 Most U.S. public water systems use disinfectants such as chlorine that effectively destroy the free trophozoites and inhibit protozoan growth and reproduction.8,9
The CDC formalized its investigation with the formation of the AK investigation team (AKIT) in March 2011.19 This program is designed to follow up with AK patients and eye-care providers using a standardized interview focused on their eye-care practices before and around the time of diagnosis. At the same time, CDC will collect similar data from control patients matched to the AK group for age, geographic area and contact lens use. The emphasis on standardized interviews and careful data collection on controls should ensure that this study will provide an unbiased assessment of health risks associated with AK, as well as valuable information on how to combat future outbreaks. In the CDC report regarding the 2006 outbreak in Chicago, the authors pointed out that unavoidable bias was introduced because of the post-hoc nature of the investigation, and the AKIT study is designed to avoid that pitfall. While the final report is not scheduled for release until later this year, “preliminary analysis identified an association with contact lens use ... but did not reveal the need to recall any particular contact-lens associated product.”19 This underscores the principal message from all recent studies: The best way to avoid AK is for patients to be conscientious in following the hygiene guidelines for their specific lens products.
Diagnosis and Treatment
The first step in what researchers refer to as the “pathogenic cascade”4 associated with AK is the binding of an Acanthamoeba surface protein to mannose-glycoproteins on the ocular epithelial surface. Expression of these glycoproteins is variable, and may explain the disconnect between high levels of Acanthamoeba in the environment and the relatively low rates of infection. Two other factors point to these glycoproteins as players in AK: Their expression is upregulated at sites of corneal trauma and in contact lens wearers.4 Once bound to the ocular surface, the pathogen produces cytotoxic proteases and additional factors which lead to the destruction of corneal tissue.
|
A careful slit-lamp exam can reveal evidence of Acanthamoeba, either directly or by the presence of inflammation. It’s essential that AK be differentiated from the more common viral keratitis. The lack of bulbous dendrites can be diagnostic of AK, as they are typical of herpes infections. Corneal scrapings can be stained for the presence of either trophozoites or cysts, and these samples can also be cultured for diagnostic confirmation of viable Acanthamoeba. However, while they represent the gold standard, protozoan cultures can take days to give results.
Recently, advances in corneal confocal imaging have allowed its use in many practices, where it can be an invaluable tool in timely, accurate identification of Acanthamoeba. In a recent survey of ophthalmologists,21 77 percent reported using both examination and culturing in diagnosis of AK, and 44 percent reported using confocal microscopy. A majority of this last group were in university-based practices, so instrument availability still plays a role. In comparative studies, confocal microscopy showed high sensitivity and specificity as a diagnostic tool.22 Despite this, most practitioners do not have access to these instruments, so traditional methods will likely remain essential for proper AK diagnosis. When these methods fail to produce a diagnosis, corneal biopsy may become necessary.
Therapies haven’t advanced to the extent that diagnostics have, but the goal of prompt, aggressive treatment is well-established. Most practitioners employ a cationic antiseptic such as polyhexamethylene biguanide 0.02% or chlorhexidine 0.02%, in combination with a diamidine such as Brolene.20,23 This combination of agents with complementary mechanisms of action is usually effective, and with time can eradicate both active amoebae and cysts. Use of either agent alone can also be effective, but combination therapy was favored by greater than 90 percent of surveyed ophthalmologists.21 A similar proportion focus on medical therapy before considering surgical options, and most employ steroids after about a month of antimicrobial therapy.
Acanthamoeba infection requires a long duration of therapy. A recent study examining clearance of detectable Acanthamoeba in the course of a long-term combination therapy reported a median clearance time of just over six weeks in the 37 patients surveyed.24 The study also showed that either early use of steroids or stromal involvement was associated with clearance times twice that of the median.
Acanthamoeba keratitis is a rare but serious disease that, if undetected, can cause permanent corneal damage and result in blindness or significant loss in visual acuity. The ephemeral nature of AK outbreaks has reinforced the misconception that the disease is primarily the result of contaminated or otherwise faulty contact lens solutions. The reality is that the organism is common, and contact lens wearers must use good hygiene to minimize their risk. Patients need to understand that showering or swimming without removing their contacts may cost them their eyesight. Also, the upcoming report from the CDC should provide guidance in terms of the role public health officials can play. Ultimately, our response to AK outbreaks should focus on proper contact lens habits, and when needed, prompt diagnosis and treatment.
Dr. Abelson, a clinical professor of ophthalmology at Harvard Medical School and Senior Clinical Scientist at the Schepens Eye Research Institute, consults in ophthalmic pharmaceuticals. Dr. Udell is professor and chairman of the department of ophthalmology, Hofstra-North Shore Long Island Jewish School of Medicine. Dr. McLaughlin is a medical writer at Ora, Inc.
1. Moore MB, McCulley JP, Luckenbach M, et al. Acanthamoeba keratitis associated with soft contact lenses. Am J Ophthalmol. 1985;100:396-403.
2. Schuster Fl, Visvesvara GS. Free-living amoebae as opportunistic and non-opportunistic pathogens of humans and animals. Int J Parasitology 2004;34:1001-27.
3. Marciano-Cabral F, Cabral G. Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev 2003;16:273-307.
4. Clarke DW, Niederkorn JY. The pathophysiology of Acanthamoeba keratitis. Trends in Parasitology 2006;22:175.
5. Hiti K, Walochnik J, Haller-Schober EM, Faschinger C, Aspöck H. Viability of Acanthamoeba after exposure to a multipurpose disinfecting contact lens solution and two hydrogen peroxide systems. Br J Ophthalmol. 2002; 86:144-6.
6. Visvesvara GS, Moura H, Schuster FL. Pathogenic and opportunistic free-living amoebae. FEMS Immunol Med Microbiol. 2007;50:1-26.
7. Meier PA, Mathers WD, Sutphin JE, Folberg R, Hwang T, Wenzel RP. An epidemic of presumed Acanthamoeba keratitis that followed regional flooding. Arch Ophthalmol 1998;116:10904.
8. Joslin CE, Tu EY, McMahon RR, Passaro OJ, Stayner LT, Sugar J. Epidemiological characteristics of a Chicago-area Acanthamoeba keratitis outbreak. Am J Ophthalmol 2006; 142:212-217.
9. Verani JR, Lorick SA, Yoder JS, et al, The Acanthamoeba Keratitis Investigation Team. National outbreak of Acanthamoeba keratitis associated with use of a contact lens solution, United States. Emerg Infect Dis 2009;15:1236-42.
10. Butcko V, McMahon RR, Joslin CE, Jones L. Microbial keratitis and the role of rub and rinsing. Eye Contact Lens 2007;33:421-3.
11. Shoff ME, Joslin CE, Tu EY, Kubatko L, Fuerst PA. Efficacy of contact lens systems against recent clinical and tap water Acanthamoeba isolates. Cornea 2008; 27:713-9.
12. Steinemann TL. Micobiological testing for contact lens care products-public workshop: FDA white oak conference center, Silver Spring, Maryland. Eye Contact Lens. 2009; 35:115-6.
13. Lam OS, Houang E, Fan OS, Lyon D, Seal 0, Wong E; Hong Kong Microbial Keratitis Study Group. Incidence and risk factors for microbial keratitis in Hong Kong: Comparison with Europe and North America. Eye 2002;16:608-18.
14. Tu EY, Joslin CE. Recent outbreaks of atypical contact lens-related keratitis: What have we learned? Am J Ophthalmol. 2010;150:602-608.
15. Shoff ME, Rogerson A, Kessler K, Schatz S, Seal DV. Prevalence of Acanthamoeba and other naked amoebae in south Florida domestic water. J Water Health 2008;6:99-104.
16. Stockman LJ, Wright CJ, Visvesvara GS, Fields BS, Beach MJ. Prevalence of Acanthamoeba spp. and other free-living amoebae in household water, Ohio, USA—1990–1992. Parasitol Res. 2011;108:621–627.
17. Magliano ACM, Maia da Silva F, Teixeira MMG, Alfieri SC. Genotyping, physiological features and proteolytic activities of a potentially pathogenic Acanthamoeba isolated from tap water in Brazil. Experimental Parasitology 2009; 123: 231–235.
18. Kilvington S, Gray T, Dart J, Morlet N, Beeching JR, Frazer DG, et al. Acanthamoeba keratitis: the role of tap water contamination in the United Kingdom. Invest Ophthalmol Vis Sci.2004;45 :165–9.
19.http://www.cdc.gov/parasites/Acanthamoeba/outbreaks/2011/index.html accessed December 11, 2011.
20. Dart JKG, Saw VPJ, Kilvington S. Acanthamoeba Keratitis: Update 2009. Am J Ophthalmol 2009;148:487–499.
21. Oldenburg CE, Acharya NR, Tu EY, et al. Practice Patterns and opinions in the Treatment of Acanthamoeba Keratitis. Cornea 2011;30:1363–1368.
22. Tu EY, Joslin CE, Sugar J, et al. The relative value of confocal microscopy and superficial corneal scrapings in the diagnosis of Acanthamoeba keratitis. Cornea. 2008; 27:764-72.
23. Hammersmith KM. Diagnosis and management of Acanthamoeba keratitis. Curr Opin Ophthalmol 2006;17:327-331.
24. Oldenburg CE, Keenan JD, Cevallos V, et al. Microbiological cure times in Acanthamoeba keratitis. Eye 2011;25:1155–1160.