Glaucoma surgery is generally undertaken when medical therapy and appropriate laser treatment fail to produce adequate intraocular pressure reduction. In recent years, the options for surgically managing glaucoma in that situation have greatly expanded with the introduction of the new “minimally invasive glaucoma surgeries,” or MIGS. But despite be-ing known for their excellent safety profile, MIGS haven’t replaced the traditional glaucoma surgeries—trabeculectomy and tube shunt implantation. That’s true because tube shunts and trabeculectomies are still the most effective ways to achieve the very low IOP that’s required for some patients.
However, having two surgical options that are capable of producing very low pressures has caused some surgeons to wonder whether one might be superior to the other in different surgical situations. Here, I’d like to share some of what the Primary Tube vs. Trabeculectomy Study is revealing about performing these procedures on eyes that have not undergone previous incisional ocular surgery.
Comparing Tubes and Trabs
Glaucoma specialists continue to have different opinions regarding the best surgical approach for patients with medically uncontrolled glaucoma. Medicare claims data indicate that tube shunts are increasingly being used as an alternative to trabeculectomy. Surveys of the American Glaucoma Society membership have also demonstrated a growing preference for tube shunts over trabeculectomy in many clinical scenarios.
One clinical situation that’s part of this debate is whether a tube shunt or trabeculectomy is preferable when operating on a “virgin” eye that hasn’t undergone previous incisional surgery. In the most recent AGS survey, trabeculectomy has remained the most popular option as an initial procedure for eyes with POAG, but a growing number of glaucoma surgeons prefer the use of tube shunts as an initial glaucoma procedure. Until now, however, this surgical choice was being made with limited data.
The Primary Tube Versus Trabeculectomy (PTVT) Study was undertaken to provide that data. It’s an investigator-initiated trial designed to compare the safety and efficacy of trabeculectomy with mitomycin-C to tube shunt surgery, when performed as an initial procedure in eyes that haven’t had previous incisional ocular surgery.
Study Design
The design of the PTVT Study is similar to that of the Tube Versus Trabeculectomy (TVT) Study, an earlier trial that enrolled patients with prior cataract extraction and/or failed filtering surgery. In contrast, the PTVT Study recruited patients without prior incisional ocular surgery. Participants in the PTVT Study were 18 to 85 years old, with IOP
≥18 mmHg and ≤40 mmHg on maximum tolerated medical therapy. The vast majority of the subjects had primary open-angle glaucoma, and there were no significant differences between baseline demographic and ocular characteristics of the groups.
 |
Reasons for exclusion included a narrow anterior chamber angle; secondary glaucomas such as neovascular, uveitic, iridocorneal endothelial syndrome, epithelial downgrowth and steroid-induced glaucoma; severe posterior blepharitis; conjunctival scarring; a functionally significant cataract; and any anticipated need for additional ocular surgery.
As in the TVT Study, participants in the PTVT Study were randomized to a 350-mm2 Baerveldt glaucoma implant or a trabeculectomy with MM-C, although the dosage of MM-C was lower than in the TVT Study. During the design phase of the study, we considered letting each glaucoma surgeon implant whichever tube shunt was preferred. However, the biostatisticians tasked with analyzing our data advised us to standardize the surgeries as much as possible.
It was a consensus opinion among the investigators in our study that the 350-mm2 Baerveldt implant offered the highest efficacy among available implants due to its large surface area, and this has been supported by two landmark clinical trials—the Ahmed Baerveldt Comparison (ABC) Study and the Ahmed Versus Baerveldt (AVB) Study. So, a 350-mm2 Baerveldt glaucoma implant was used in all patients randomized to the tube group. We also standardized other parts of the procedure, such as having the implant placed in the superotemporal quadrant with the tube in the anterior chamber, and a complete restriction of flow at the time of surgical implantation. We standardized aspects of the trabeculectomies as well. An MM-C dosage of 0.4 mg/ml for two minutes was used in all patients, and the surgery was performed superiorly.
Despite standardization of many aspects of each procedure, surgeons were allowed sufficient flexibility to perform both operations in a manner in which they were comfortable and proficient. For example, surgeons were allowed to decide whether a limbus- or fornix-based conjunctival flap was used. The method of tube ligation, the size of the trabeculectomy flap and the number of flap sutures were also left to the surgeon’s discretion.
Sixteen clinical centers were involved in the study. Patients were randomly assigned to one of the two groups, with follow-up visits scheduled for one day, one week, one month, three months, one year, 18 months and two, three, four and five years postoperatively. The primary outcome measure was the rate of surgical failure, using criteria consistent with recommendations from the World Glaucoma Association.
The Data (So Far)
Results from the PTVT Study at year three were presented at the 2019 Annual AGS Meeting. Highlights included:
• IOP reduction. The pressure reduction was greater in the trabeculectomy group at all time points, and that difference was statistically significant—with the exception of the 18-month and two-year time points. Furthermore, the greater degree of pressure reduction in the trabeculectomy group was achieved with fewer postoperative glaucoma medications. This clearly suggests that trabeculectomy with MM-C was more effective at reducing IOP than tube shunt implantation in this population.
• Rate of surgical failure. The primary outcome measure for the study was the rate of surgical failure, using Kaplan-Meier survival analysis. The rate of surgical failure increased over time, which came as no surprise; even glaucoma surgeries that are initially successful can eventually fail. At one year, the tube shunt group had a significantly higher failure rate than the trabeculectomy group. The cumulative probability of failure in the trabeculectomy group continued to trend somewhat lower than the tube group over time. However, the difference in failure rate between the
groups was no longer statistically significant at three years.
• Reasons for failure. We found some differences in the reasons for surgical failure between the two study groups. The most common reason for failure in both groups was insufficient pressure reduction; either the pressure was above 21 mmHg at two consecutive visits, or it wasn’t reduced 20 percent below baseline at two consecutive visits. Other reasons for failure included needing a reoperation for glaucoma or having hypotony. Interestingly, all of the hypotony failures occurred in the trabeculectomy group, while more reoperations were seen in the tube group.
• Need for adjunctive medical therapy. Patients who didn’t fail were subdivided into complete or qualified successes. Complete successes didn’t need postop adjunctive medical therapy; qualified successes were using glaucoma medications. The rate of complete success was significantly higher in the trabeculectomy group than in the tube shunt group. (That’s an important consideration when man-aging a patient who can’t tolerate medications, or a patient who is poorly compliant.)
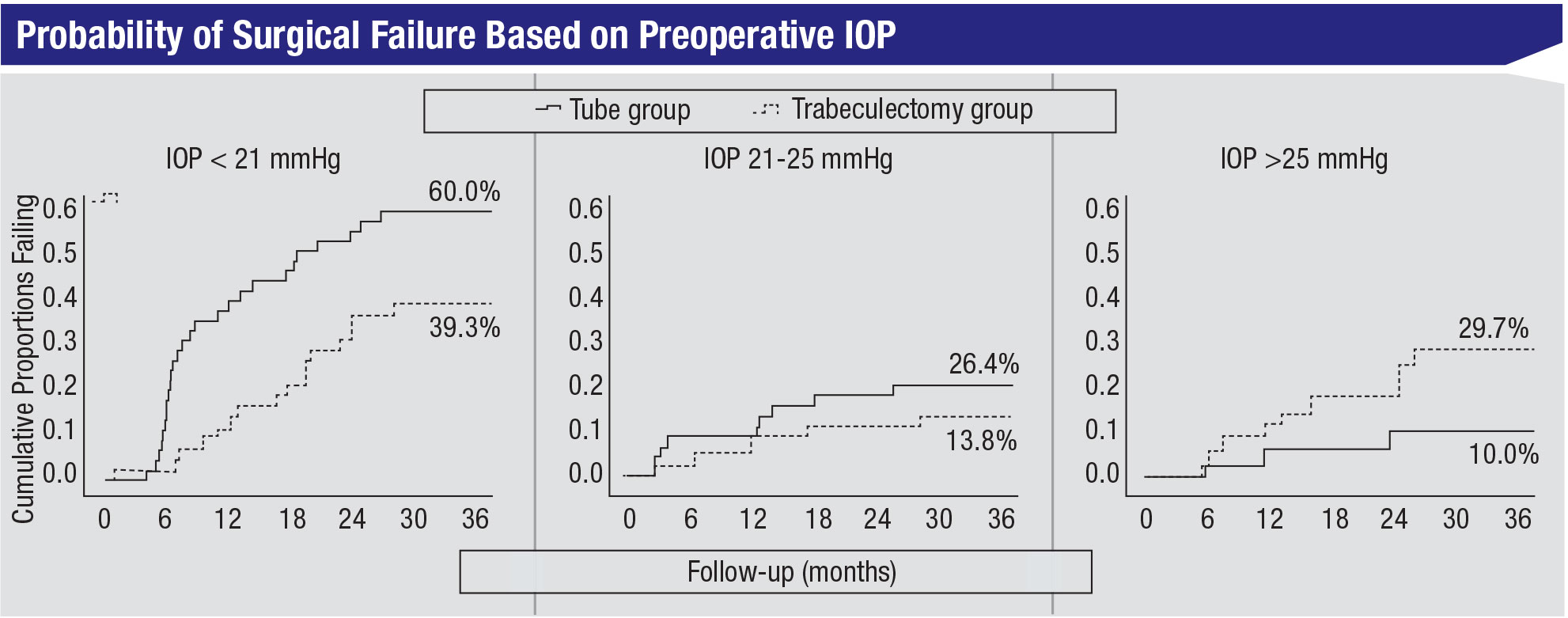 |
• Baseline factors associated with surgical failure. We performed a risk factor analysis to identify baseline factors that were associated with failure in the study. The only significant predictor of failure was preoperative IOP. The patients with lower preoperative pressures had a higher risk of failure, and this was especially true in the tube group. (See figure, above.) In my opinion, this is one of the study’s most interesting and important findings.
• Complications. In terms of complications, the PTVT Study’s findings have been similar to what’s been observed in other prospective glaucoma surgical trials, including the TVT, ABC and AVB studies. Postoperative complications are very common after traditional glaucoma surgery, whether it be a tube shunt or a trabeculectomy. Fortunately, most of these complications are self-limited and resolve without any specific intervention.
We did, however, find some differences between the types of com-plications occurring in the two treatment groups:
— Early postoperative complications, defined as those occurring in the first month after surgery, were more common in the trabeculectomy group than in the tube group.
— The rate of later postoperative complications (occurring after a month) wasn’t significantly different between the two groups.
— The rate of serious complications, defined as those requiring a reoperation to manage the com-plication, and/or resulting in a vision loss of two or more lines of Snellen visual acuity, wasn’t significantly different between the treatment groups.
— Overall, the rate of reoperation for complications trended higher in the trabeculectomy group compared to the tube group.
The complications data suggests that tube shunt surgery may have a slightly better safety profile than trabeculectomy with MM-C, at least during the first three years postop. If you combine all complications together, they were significantly more common in the trabeculectomy group than the tube group. However, the PTVT trial will continue out to five years, and the long-term data will be important for evaluating these two surgical procedures, in terms of both safety and efficacy.
• Number of postoperative interventions. In my experience, postoperative management following tube shunt surgery tends to be less involved than postoperative management following trabeculectomy. However, in this study the number of postop interventions performed in the clinic, such as laser suture lysis or pulling a rip-cord suture, wasn’t significantly different whether the patient received a tube or a trabeculectomy.
• Visual acuity. We didn’t find any significant difference between the treatment groups with regard to visual acuity outcomes.
• Cataract formation. All of the patients in our study were phakic. After three years of follow-up, about a third of the patients in the study had undergone cataract extraction.
There’s ample evidence in the literature to suggest that glaucoma surgery, whether it be trabeculectomy or tube shunt surgery, accelerates the development of cataract. This data suggests that cataract progression occurs at a similar rate among patients undergoing trabeculectomy or tube shunt surgery.
• Patient comfort. One of the complications we specifically asked patients about was dysesthesia, or patient discomfort. We found no significant difference in patient-re-ported dysesthesia between the two procedures.
Take-home Highlights (So Far)
What useful information can we take away from this study at the three-year follow-up mark?
• Preoperative IOP impacts the likelihood of success. When patients have lower levels of preoperative IOP—in our post hoc analysis the cutoff was less than 21 mmHg—trabeculectomy appears to be a more successful operation than tube shunt surgery. I suspect this is related to the titratability of trabeculectomy; it allows us to do laser suture lysis or use releasable sutures to titrate the pressure to lower levels in the early postoperative period. I think that’s particularly valuable in patients who have initial low pressure.
• Both tube shunt surgery and trabeculectomy with MM-C are very effective at lowering IOP. Mean postoperative pressures were in the low teens in both groups throughout three years of follow-up; most patients ended up with pressures of 14 mmHg or less. MIGS procedures seldom achieve this degree of IOP reduction. As previously noted, traditional glaucoma surgeries are still an important part of our armamentarium.
• Your choice of tube or trabeculectomy may ultimately come down to safety vs. efficacy. There seems to be an inevitable tradeoff between safety and efficacy in glaucoma surgery. In our study com-paring tube shunt implantation and trabeculectomy, the data suggests that trabeculectomy is more effective, but more likely to be associated with complications. A similar dichotomy was seen in the ABC and AVB studies; the Baerveldt implant was more effective, while the Ahmed valve was safer. Likewise, when com-
paring traditional glaucoma surgeries to MIGS, we find tube shunts and trabeculectomy are more efficacious, but MIGS are safer. So added efficacy seems to be achieved at the expense of reduced safety—at least with the existing glaucoma surgical options.
This shouldn’t be surprising. Many of the complications that we contend with in glaucoma surgery are related to hypotony, such as anterior chamber shallowing and choroidal effusions, and sometimes more serious complications, including suprachoroidal hemorrhage and hypotony maculopathy. (Of course, it’s possible that some future glaucoma surgery will be both highly effective and low-risk.)
A Few Final Thoughts
The purpose of a trial like the PTVT Study is to provide useful information that will help surgeons select the best glaucoma procedure for an individual patient. Data produced by the trial thus far hasn’t demonstrated clear superiority of one of these procedures over the other.
However, there’s another important consideration when choosing a glaucoma surgical procedure, one that was not addressed in the PTVT Study: the surgeon’s experience and comfort with each procedure. All of the surgeons in the PTVT Study were proficient and experienced with both of the procedures being studied, but this may not be the case with all surgeons. A surgeon’s familiarity with each operation is an important factor when deciding which surgery to perform.
One interesting question that our study may eventually help answer is how tube shunt and trabeculectomy function is impacted by subsequent cataract surgery. All eyes enrolled in our study were phakic, but a third of the patients had undergone cataract surgery by the three-year time point. Multiple studies have demonstrated that when performing cataract surgery in eyes with preexisting trabeculectomy filtering blebs, there’s some risk of bleb failure. However, there’s less information in the medical literature about how tube shunts fare with subsequent cataract removal. I believe the data coming from a prospective, randomized clinical trial will add significant new information to help answer this question. However, we’re waiting for the five-year data to do a full statistical analysis relating to this question.
As noted earlier, glaucoma surgical practice patterns appear to be shifting away from trabeculectomy and toward implanting tube shunts. The TVT Study provided data to support this trend, as tube shunt surgery was found to be more successful, with a lower rate of early postop complications relative to trabeculectomy with MM-C among patients with prior cataract and/or failed glaucoma surgery. Notably, this efficacy result wasn’t seen in the PTVT Study. However, it’s important to realize that different patient populations were studied in the TVT and PTVT Studies.
Randomized clinical trials offer the highest level of evidence-based medicine, but caution should be used in extrapolating study results to dissimilar patients. We now have two trials that compared tube shunts and trabeculectomy with MM-C in different populations, and we’re seeing somewhat different results. So remember to apply the lessons learned only to the appropriate patient population. REVIEW
Dr. Gedde is a professor of ophthalmology and vice chair of education at Bascom Palmer Eye Institute. He is a study chairman for the PTVT Study. He has no personal financial ties to any product mentioned, but notes that the PTVT Study was funded by grants from Johnson & Johnson Vision, the National Eye Institute and Research to Prevent Blindness.



