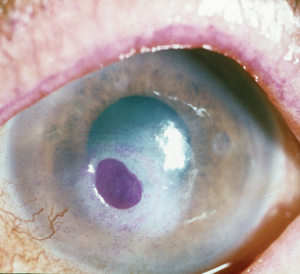
For those of us focused on the anterior segment, we recognize that chronic keratitis comes in many forms, and these present significant therapeutic challenges. From the filamentary keratitis of dry eye and the interstitial keratitis seen in autoimmune diseases to the persistent corneal defects associated with neurotrophic keratitis, there are treatments to alleviate pain and treatments that reduce the risk of disease progression, but there is still much room for improvement in our therapeutic choices.
This month, we’ll briefly consider the healthy cornea, discuss some of the worst-case scenarios for keratitis, and describe how these concepts are driving some of the newest ideas in therapies designed to heal the damaged ocular surface.
The Healthy Cornea
The healthy cornea is a unique tissue designed and maintained to transmit and refract light to the lens and the retina. The layered structure of epithelium, stroma and endothelium, along with intervening Bowman’s and Descemet’s membranes, is key to this function. In addition, there are centripetal aspects to corneal homeostasis. The peripheral cornea interacts with adjacent limbal cells, receives input from surrounding vasculature and is the site of epithelial progenitor cells. The central cornea is an avascular structure with a fully differentiated epithelial layer. Corneal health depends upon the tear film and the aqueous humor for nutritional support and, under normal conditions, exhibits a robust, rapid response to minor surface abrasions and other incidental traumatic stimuli.
While we spend much of our time defining the cause-and-effect relationships underlying corneal pathologies, once in a while it’s a useful exercise to consider all the barriers and redundancies built into the cornea and ask what mechanisms and systems are in place to keep the cornea healthy and functional.2,3 For example, while each of the layers participates as a barrier to environmental and microbial contaminants, they also have specific functions that allow the cornea to conduct the business of light transmission and refraction. The endothelial cells, for example, actively regulate fluid and solute movement into the stromal layer to maintain the deturgescence necessary for corneal transparency.4 The epithelial layer, in contrast, participates in this process indirectly by maintenance of high resistance, tight junctional barriers at the epithelial basement membrane.
|
In cases where tissue damage does not penetrate the epithelial layer (about 60 to 70 µm thick) this process can be completed in as little as 24 hours. More severe traumatic events require epithelial proliferation, migration and reformation of epithelial tight junctions at the basement membrane. With this in mind it’s easy to imagine how modest trauma to the corneal surface, such as that which occurs with chronic allergy or dry eye, can have additive effects that challenge the normal repair mechanisms. And when these pathways are further disrupted by defects in tear formation or sensory nerve function, the cornea’s ability to respond to any traumatic insult can be significantly impaired.
Neurotrophic Keratitis
Neurotrophic keratitis is a rare, degenerative corneal disease that exemplifies the potential for ocular damage when neural feedback loops are lost or impaired.6,7 Any condition that reduces the function of the neural pathways emanating from the trigeminal nerve can lead to NK, but most commonly this occurs subsequent to herpes zoster infection, diabetic neuropathy or surgical trauma to the nerve. The resulting loss of corneal sensitivity (anesthesia) leads to a reduction in lacrimation and a decline in metabolism and mitosis of corneal epithelial cells. NK patients exhibit deficiency in epithelial repair, and can ultimately experience stromal edema, loss of microvilli and abnormal basal lamina homeostasis.
For ordinary subjects, the cornea is constantly responding to environmental insults, but patients with NK are ill-equipped to mount a healthy response to these events. They develop poorly healing, recurrent corneal abrasions or defects that are slow to respond to existing treatments. Most of the current treatments for keratitis simply augment the natural process of corneal remodeling; when this process is compromised, the effectiveness of treatments such as artificial tears, steroids or non-steroidal anti-inflammatories is also compromised.
An important question that remains unanswered is the extent to which neurotrophic action is mediated directly by neural inputs—peptides and other signaling molecules released by epithelial nerve endings—or by their ability to direct the volume of output and constituents of the lacrimal apparatus. Studies dating back a decade or more have suggested that substance P, a peptide secreted by trigeminal nerves in the cornea, exhibited trophic effects and regulated normal epithelial function.8 More recent research has implicated insulin-like growth factor 1—a known, inducible constituent of tears—as a synergistic regulator of corneal epithelium together with substance P.9 In addition, elevated levels of IGF-1 binding protein, a negative regulator of IGF-1, have been reported in tears of diabetic patients.10 Collectively, these data suggest that the combination of neural activity in the corneal epithelial layer and feedback regulation to the lacrimal apparatus work together to modulate corneal epithelial homeostasis.
Treatment Options
Staging for conditions such as NK provides a guideline for both progression of the disease and the types of therapeutic approaches in current use.6 Stage-1 NK features include: corneal anesthesia; epithelial growth dysplasia; punctate keratitis; tear-film anomalies and rose bengal staining of the conjunctiva. As the disease progresses, patients may also show corneal neovascularization or stromal scarring. Patients with a stage-1 defect are typically encouraged to avoid preservatives in ophthalmic medications and also advised to use an artificial tear to augment the lubrication of the ocular surface.11 Topical antibiotics are also commonly used to reduce the risk of corneal infection.
A hallmark of the corneal surface defects seen in NK is that they are irregular in nature and may include one or more raised edges that can exacerbate tissue damage. When defects have rounded edges, a tarsorrhaphy or a therapeutic contact lens is a common treatment strategy. Stage-2 NK may also involve stromal swelling. This can ultimately lead to stromal melting and corneal perforation.
Traditional treatments for stage-2 or stage-3 NK also include penetrating keratoplasty in combination with tarsorrhaphy or a soft bandage contact lens. Another strategy to prevent the need for corneal transplant is the use of amniotic membranes, a tissue graft that provides both epithelial donor cells and a basement membrane matrix to support corneal re-epithelialization. As with other severe forms of keratitis, responses to these treatments vary from patient to patient.
Various preparations of artificial tears containing autologous serum have been used to treat a number of corneal defects, including dry eye, graft vs. host disease (GVHD) and NK. It’s thought that serum can serve to replenish growth factors and other nutrients typically provided by a healthy tear flow. While there is a wealth of literature to support this strategy, most studies are retrospective or single-treatment studies. There are few masked, placebo-controlled trials of serum-supplemented eye drops for any form of keratitis.11 One recent study compared artificial tears to cord blood serum or autologous serum for treatment of chemical injury, and showed that cord blood was superior to the other two treatments; despite this, the artificial tears were superior to autologous serum treatment.12 This result suggests that the method of collection and preparation of serum drops may be critical to their efficacy.
A reasonable alternative to serum treatments would be to identify those serum components responsible for their efficacious effect. Two candidates for this approach are nerve growth factor and thymosin β4. Both of these are polypeptide components of normal serum and tear fluids, and both have significant data to support their use in treatment of corneal defects. NGF is a growth factor that is necessary for survival and differentiation of sympathetic and sensory neurons. It also can enhance the function of injured neurons and has additional effects on both neurons and neuronal target tissues.13 NGF has been used as an experimental therapy for dry eye14 and for vernal keratoconjunctivitis. A clinical trial is currently under way examining use of recombinant human NGF as a treatment for stage-2 and stage-3 NK.15
Like NGF, thymosin β4 is an endogenous constituent of serum, but Tβ4 is normally found in much higher concentrations. Thymosin β4 is a major constituent protein of platelets, macrophages and polymorphonuclear cells where it acts as a G-actin binding molecule and regulator of actin polymerization.16 These cell types function in trauma response, and Tβ4 gene expression is among the earliest to be upregulated during the process of wound repair. In addition to regulating actin polymerization, Tβ4 is released into the extracellular environment of wounds where it promotes tissue remodeling and repair in dermal, ocular, cardiac and central nervous system animal models.
Several studies have established the potential for Tβ4 as a treatment for corneal wounds. It was effective in treatment of corneal defects in patients with GVHD, and was also efficacious in a study of severe dry-eye patients. (Sosne G, IOVS 2013; 54:ARVO E-Abstract 6033) In a 2010 study of patients with stage-2 or -3 NK, Tβ4 treatment resulted in complete healing of persistent corneal defects in six of six patients studied.17 It’s interesting that a protein that functions as a major regulator of the cytoskeletal protein actin also affects tissue remodeling; whether the two functions are linked remains to be established. Another potential treatment on the horizon for epithelial wound healing that has a clear connection to cytoskeletal function is a peptide mimetic of connexin43. This polypeptide, like Tβ4, has shown promise in treatment of both skin and corneal wound healing.18
Perhaps even farther into ophthalmology’s future, we may see stem-cell technologies or other genome-based strategies (such as interfering RNA) applied in treatments for corneal wound healing.19,20 These theoretical treatments are a long way off, but are still in sight, giving patients hope for a lasting solution for their wounds that won’t heal. REVIEW
Dr. Abelson is a clinical professor of ophthalmology at Harvard Medical School. Dr. McLaughlin is a medical writer at Ora Inc.
1. Lutty GA. Effects of diabetes on the eye. Invest Ophthalmol Vis Sci. 2013; 54: ORSF81-ORSF87.
2. Chi C, Trinkaus-Randall V. New insights in wound response and repair of epithelium. J Cell Physiol. 2013; 228: 925-9.
3. Evans DJ, Fleiszig SM. Why does the healthy cornea resist Pseudomonas aeruginosa infection? Am J Ophthalmol 2013; 155: 961-970.
4. Srinivas SP. Dynamic regulation of barrier integrity of the corneal endothelium. Optom Vis Sci 2010;87:239-54.
5. Kowtharapu BS, Stahnke T, Wree A, Guthoff RF, Stachs O. Corneal epithelial and neuronal interactions: Role in wound healing. Exp Eye Res 2014;125:53-61.
6. Sacchetti M, Lambiase A. Diagnosis and management of neurotrophic keratitis. Clin Ophthal 2014;8:571-579.
7. Lambiase A, Sacchetti M, Mastropasqua A, Bonini S. Corneal Changes in Neuro-surgically Induced Neurotrophic Keratitis. JAMA Ophthalmol 2013;131:1547-1553.
8. Nagano T, Nakamura M, Nakata K, et al. Effects of substance P and IGF-1 in corneal epithelial barrier function and wound healing in a rat model of neurotrophic keratopathy. Invest Ophthalmol Vis Sci 2003;44:3810-5.
9. Ko JA, Mizuno Y, Ohki C, Chikama T, Sonoda KH, Kiuchi Y. Neuropeptides released from trigeminal neurons promote the stratification of human corneal epithelial cells. Invest Ophthalmol Vis Sci 2014;55:125-33.
10. Wu YC, Buckner BR, Zhu M, Cavanagh HD, Robertson DM. Elevated IGFBP3 levels in diabetic tears: A negative regulator of IGF-1 signaling in the corneal epithelium. Ocul Surf 2012;10:100-7.
11. Watson SL, Lee MH, Barker NH. Interventions for recurrent corneal erosions. Cochrane Database Syst Rev 2012;9:CD001861.
12. Sharma N, Goel M, Velpandian T, Titiyal JS, Tandon R, Vajpayee RB. Evaluation of umbilical cord serum therapy in acute ocular chemical burns. Invest Ophthalmol Vis Sci 2011;52:1087-92.
13. Kawamoto K, Matsuda H. Nerve growth factor and wound healing. Prog Brain Res 2004;146:369-84.
14. Meerovitch K, Torkildsen G, Lonsdale J, Goldfarb H, Lama T, Cumberlidge G, Ousler GW. Safety and efficacy of MIM-D3 ophthalmic solutions in a randomized, placebo-controlled Phase 2 clinical trial in patients with dry eye. Clin Ophthalmol 2013;7:1275-85.
15. NCT01756456: Evaluation of Safety and Efficacy of rhNGF in Patients with Stage 2 and 3 Neurotrophic Keratitis http://clinicaltrials.gov. Accessed 18 Aug 2014.
16. Goldstein AL, Hannappel E, Kleinman HK. Thymosin β4: Actin-sequestering protein moonlights to repair injured tissues. Trends Mol Med 2005;11:421-429.
17. Dunn SP, Heidemann DG, Chow CYC, Crockford D, Turjman N, Angel J, Allan CB, Sosne G. Treatment of Chronic Nonhealing Neurotrophic Corneal Epithelial defects with Thymosin β4. Ann NY Acad Sci 2010;1194:199-206.
18. Moore K, Ghatnekar G, Gourdie RG, Potts JD. Impact of the controlled release of a connexin 43 peptide on corneal wound closure in an STZ model of type 1 diabetes. PLOS One 2014;9:1:e86570.
19. Hanson C, Hardarson T, Ellerström C, et al. Transplantation of human embryonic stem cells onto a partially wounded human cornea in vitro. Acta Ophthalmol 2013;91:127-130.
20. Mulik S, Bhela S, Rouse BT. Potential function of miRNAs in herpetic stromal keratitis. Invest Ophthalmol Vis Sci 2013;54:563-573.