The idea of testing visual fields outside the office isn’t new. However, novel technology is making it easier to check visual function more reliably at home. Ideally, this would facilitate monitoring of presumed stable or controlled glaucoma patients or suspects, to reduce the overall number of office visits per year. Innovative solutions have shifted from internet and desktop-based testing to more portable and virtual methods that help to reduce fixation losses, improve engagement and possibly help reshape the paradigm of repeatable visual field testing—especially in low-resource settings. Here’s a look at the current state of “at-home” visual field testing.
The Benefits
While visual fields remain the gold standard for monitoring visual function, the perimetry machine is a large, carefully calibrated bowl that’s mostly used in an office setting and is typically administered by a perimetrist to help patients maintain focus. Unfortunately, this gold-standard medical test, which is highly subjective, can’t be repeated as often as physicians would like. There are problems with patient attention, fatigue, comprehension, boredom, etc., and typically, patients only perform one or two fields per year in the office (or even fewer if lost to follow-up). Thus it would be nice to have a way for patients to test themselves at their own convenience to fill in the gaps.
The additional data would be useful in two main situations:
• for reassure that a glaucoma suspect is still normal; and
• to assess whether a glaucoma patient is progressing.
Of course, office examination would likely still be needed for confirmatory testing and treatment recommendations, but a faster, easier self-administered test outside the office could be used as a screening method to help determine when patients need to return to the specialist.
Many glaucoma patients routinely see the doctor on a schedule, every three, six or 12 months, depending on the level of risk or stage of disease. At-home testing affords the possibility of remote monitoring and enabling tele-glaucoma care.
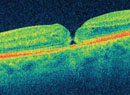
 |
| Melbourne Rapid Fields is an iPad app that uses a moving fixation target to test up to 30 degrees of field. |
A Potential Disadvantage?
Visual fields alone, whether done in-office or at home, normally don’t provide enough clinical data to manage glaucoma patients, despite serving as a clinical end point. Specialists also review glaucoma risk factors, optic nerve appearance, intraocular pressure, structural macular and nerve fiber layer imaging, and a complete eye exam. If portable visual field data were relied upon to make the diagnosis of progression, the diagnosis might come too late, as glaucoma is irreversible and identification of progression prior to field loss is optimal. With that in mind, it’s imperative to continue to examine a glaucoma patient at regularly scheduled office visit intervals. Furthermore, sometimes too much additional data results in noise, creates additional work interpreting test results, and may even be more costly, if there’s overtesting.
Artificial Intelligence
Researchers are exploring how to apply artificial intelligence algorithms to all sorts of ophthalmic test results, with most of the recent excitement centering on deep learning. Multiple publications have analyzed automation of visual field interpretation for the detection of glaucoma.1-4
While physicians currently plot VF glaucoma progression analysis using a visual function index for event-based and trend-based progression analysis relative to a normative database, one can imagine that with more data, the algorithms could optimize comparison of patients with similar severity, or their own baseline, with better predictive analytics. Additionally, when some risk factors change, self-directed, out-of-the-office testing allows for additional, concentrated repeat testing at that time point to make a more informed decision about the timing or risk/benefit ratio of a given therapeutic intervention.
Finally, even if a lower-resolution, simple, portable test isn’t as accurate as a high-resolution, in-office gold standard, there’s still value in collecting a high volume of lower-resolution data. Aggregating more data over a longer period of time may actually be more predictive than the smaller amount of higher-quality data that’s acquired only during annual or semi-annual office visits.
 |
| A limitation of using portable devices to perform VF testing is the inability to control fixation and the distance from the eye to the screen. Virtual reality headsets may solve those issues through adjustable fixation using gaze tracking. |
Portable Tests
Portable visual fields continue to evolve with the advent of tablets, smartphones and virtual reality headsets. Some of the most notable tests, both past and present, include the following:
• Moorfields Motion Displacement Test. One of the early portable visual field tests was the Moorfields Motion Displacement Test. Developed on a laptop PC, the MDT was designed for speed and patient ease of use. In the test, patients saw 32 white lines on a gray background while fixating in the center. When any of the lines moved or “wiggled,” the patient would click a button. The two-minute test could be completed without glasses and even when a cataract was present. While the MDT is no longer available, it represented the early push to move away from an office-based perimeter and onto existing computer screens, a test that could be administered both online and offline. (Other similar computerized perimetric strategies include rarebit perimetry, Peristat and campimetry.)
• Melbourne Rapid Fields Test. Dr. George Kong created Visual Field Easy (https://itunes.apple.com/us/app/visualfields-easy/id495389227), a free iPad app, now known as the Melbourne Rapid Fields test. The MRF takes advantage of a moving fixation target in order to effectively increase the tablet surface area to test up to 30 degrees of field. Several published studies have validated the device as a screening tool similar to a tangent perimeter.5-7
MRF denoted a further shift toward portability of VF tests, thanks to the advent of iPads making software more portable. It’s easy to use, inexpensive and can be used anywhere, which is a big advantage in remote locations. Over time, threshold testing strategy, stimulus size and screen contrast have been standardized across tablets.
A recent study by Australia’s Selwyn Prea, BOptom, Mphil, and colleagues included 60 patients; it was conducted to compare the repeatability of the MRF vs. the Humphrey Field Analyzer SITA-standard and fast programs. The investigators found that MRF had good test-retest repeatability and that it correlated strongly with HFA across four visits over six months.6
In terms of speed, MRF was similar to SITA-fast and was significantly faster than SITA-standard (MRF 4.6 ±0.1 minutes vs. SITA-fast 4.3 ±0.2 minutes vs. SITA-standard 6.2 ±0.1 minutes, p<0.001).6 MRF proved to be very repeatable, with intraclass correlation coefficients (with a value of 1 being a perfect correlation) for baseline and the six-month visit being 0.98, compared to 0.95 and 0.93 for SITA-fast and SITA-standard, respectively.6
• Virtual Reality Peripheral Vision Testing. One of the major limitations of using portable devices to perform VF testing is the inability to control fixation and the distance from the eye to the screen. Virtual reality headsets solve those issues through adjustable fixation using gaze tracking. With VR, no matter where the patient looks, the stimulus can be shown relative to fixation at that moment. Gyroscopes can account for head movement, and the immersive environment can improve user engagement. VR headsets can take advantage of smartphones and easily test individual eyes without one needing to be patched.
There are multiple inexpensive, lightweight, mobile VR applications and software platforms that are either available or in development, such as Vivid Vision (https://www.seevividly.com), BioFormatix’s VirtualEye Perimeter (http://bioformatix.com/perimetry.html), MicroMedical Devices’ PalmScan VF2000 (https://micromedinc.com/our-devices/palmscan-vf2000-visual-field-perimeter/) and Elisar’s eCloud Perimeter (https://www.elisar.com/). These applications can be used with a variety of input “clickers.”
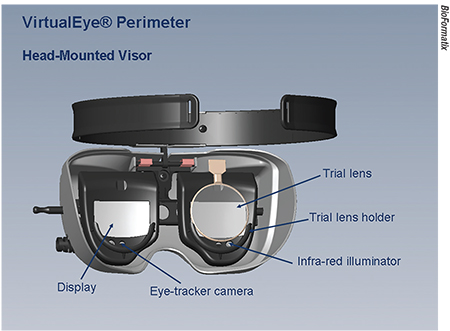 |
| With virtual reality, no matter where the patient looks, the stimulus can be shown relative to fixation at that moment. Gyroscopes can account for head movement, and the immersive environment can improve user engagement. |
One early VR study by Athens’ Stylianos Tsapakis and co-workers found a high correlation between the reliability of VF testing using a VR testing system they developed and the Humphrey test.8 The central 24 degrees of visual field were tested in 20 eyes of 10 patients using VR glasses, a smartphone with a 6-inch display and software that implemented a fast-threshold 3-dB step staircase algorithm. The results were compared to those using the Humphrey perimeter test on the same group of patients, which resulted in a correlation coefficient of r=0.808 (p<0.0001).8
The next level of improvement over virtual reality visual field testing would be to eliminate the subjective aspect of clicking to indicate when a visual stimulus is seen. Could there be a way to detect a patient’s response to a given stimulus via the brain, without any input? This is the goal of nGoggle, a portable brain-computer interface (like an electroencephalograph) that detects visual function through recorded electrical responses directly from the visual cortex.
In a study involving the nGoggle, researchers (some of whom have a financial interest in nGoggle Inc.) sought to objectively assess visual function loss in 62 eyes of 33 glaucoma patients. Each subject underwent VF testing using the nGoggle and a standard automated perimetry test over the course of three months. The nGoggle was able to distinguish between healthy eyes and those with glaucomatous neuropathy.9 Researchers measured values such as receiver operating characteristic curves summarizing diagnostic accuracy, intraclass correlation coefficients and coefficients of variation for assessing repeatability. The ROC curve area for the nGoggle mfSSVEP was 0.924 (95% CI: 0.863-0.964), which was larger than SAP MD (AUC=0.813; 95% CI: 0.716-0.896), SAP MS (AUC=0.797; 95% CI: 0.687-0.880; p=0.030) and SAP PSD (AUC=0.768; 95% CI: 0.657-0.858; p=0.012).9
As the nascent field of portable visual field testing continues to develop, we’re reminded of how much technological advancements play a role in enabling new diagnostics, that don’t need to be restricted to the medical office. These new enhancements may make visual field exams more interesting, faster and more reliable. Moving forward, generating more functional visual data will be useful for quantifying rates of progression more accurately to match with optimally timed interventions. Automated algorithms will continue to improve as we enter the era of big data, driven by Internet-connected portable devices. REVIEW
Robert Chang, MD, is an ophthalmologist at Byers Eye Institute at Stanford Eye Institute.
1. Kucur S, Holló G, Sznitman R. A deep learning approach to automatic detection of early glaucoma from visual fields. PLoS One 2018;13:11:e0206081. doi: 10.1371/journal.pone.0206081. eCollection 2018.
2. Ting DSW, Pasquale LR, Peng L, Campbell JP, Lee AY, Raman R, Tan GSW, Schmetterer L, Keane PA, Wong TY. Artificial intelligence and deep learning in ophthalmology. Br J Ophthalmol 2019;103:167-175.
3. Li F, Wang Z, Qu G, Song D, Yuan Y, Xu Y, Gao K, Luo G, Xiao Z, Lam DSC, Zhong H, Qiao Y, Zhang X. Automatic differentiation of glaucoma visual field from non-glaucoma visual filed using deepconvolutional neural network. BMC Med Imaging 2018;18:1:35.
4. Asaoka R, Murata H, Iwase A, Araie M. Detecting preperimetric glaucoma with standard automated perimetry using a deep learning cassifier. Ophthalmology 2016;123:1974-80.
5. Johnson CA, Thapa S, George Kong YX, Robin AL. Performance of an iPad application to detect moderate and advanced visual field loss in Nepal. Am J Ophthalmol 2017;182:147-154.
6. Prea SM, Kong YXG, Mehta A, He M, Crowston JG, Gupta V, Martin KR, Vingrys AJ.
Six-month longitudinal comparison of a portable tablet perimeter with the Humphrey Field Analyzer. Am J Ophthalmol 2018;190:9-16.
7. Vingrys AJ, Healey JK, Liew S, Saharinen V, Tran M, Wu W, Kong GY. Validation of a tablet as a tangent perimeter. Transl Vis Sci Technol 2016;5:4:3. eCollection 2016 Jul.
8. Tsapakis S, Papaconstantinou D, Diagourtas A, Droutsas K, Andreanos K, Moschos MM, Brouzas D. Visual field examination method using virtual reality glasses compared with the Humphrey perimeter. Clin Ophthalmol 2017;11:1431-1443
9. Nakanishi M, Wang YT, Jung TP, Zao JK, Chien YY, Diniz-Filho A, Daga FB, Lin YP, Wang Y, Medeiros FA. Detecting glaucoma with a portable brain-computer interface for objective assessment of visual function loss. JAMA Ophthalmol 2017;135:6:550-557.