 |
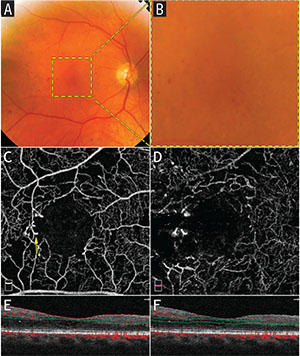
| Figure 1. Multimodal imaging of a 65-year-old patient with moderate non-proliferative diabetic retinopathy. (A) Color photograph shows intraretinal blot-dot hemorrhages and microaneurysms. (B) Yellow-dashed frame identifies the corresponding 3x3-mm area imaged on OCTA. (C) OCTA segmented at the superficial retinal plexus shows an enlarged foveal avascular zone (FAZ). Microaneurysms can also be easily identified (yellow arrow). (D) OCTA segmented at the deep retinal plexus shows further enlargement of the FAZ. (E) and (F) represent corresponding OCT B-scansegmentation of the superficial and deep plexuses, respectively, with decorrelation signal overlay. |
OCTA Explained
OCTA is a noninvasive imaging modality that allows for detection of blood flow and three-dimensional reconstruction of blood vessels using signal decorrelation between consecutive transverse cross-sectional OCT scans. An OCTA image is computed by comparing, on a pixel-by-pixel basis, repeated B-scans acquired at the same retinal location in rapid succession. The rationale behind OCTA imaging is that in non-mobile tissue the reflected signal will be stationary, and thus the repeated B-scans will be identical. Inside vasculature, however, moving erythrocytes cause a time-dependent backscattering of the OCT signal, which manifests as differences among the repeated B-scans.4-6
Basically, OCT angiograms of the posterior pole can be obtained by using one or a combination of two methodologies: amplitude decorrelation and phase-variance. Amplitude decorrelation analyzes amplitude changes in the OCT signal. Split-spectrum amplitude decorrelation partitions the spectrum into smaller spectra and performs the repeated B-scan decorrelation separately for each sub-spectrum, which improves the signal-to-noise ratio.7 Doppler OCT is a phase-based technology from which OCTA has its origins.8,9 It can quantify axial blood flow that’s parallel to the direction of the imaging acquisition device.7,10,11
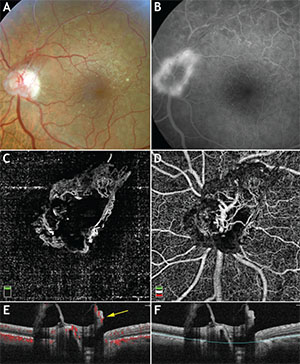 |
| Figure 2. Multimodal imaging of a 60-year-old patient with superotemporal branch retinal vein occlusion. (A) Color photograph shows neovascularization of the disc. (B) Late-phase FA shows hyperfluorescence of the optic nerve due to neovascularization leakage. Optical coherence tomography angiography of the vitreous (C) and optic nerve head (D) demonstrates some well-delineated, fine, abnormal vessels. (E) and (F) represent corresponding OCT B-scan segmentation of the optic nerve head and vitreous. The yellow arrow points to the presence of the decorrelation signal on the vitreous interface, indicative of neovascularization flow. |
OCTA for Vascular Disease
The retinal capillary network is arranged into morphologically distinct layers. The superficial retinal capillary plexus is located predominantly within the ganglion cell layer; and the deep retinal capillary plexus is located at the outer boundary of the inner nuclear layer, with a smaller intermediate retinal capillary plexus at the inner margin of the inner nuclear layer. The vascular layers of the retina are connected by perpendicularly positioned vessels.12 With the ability to analyze each vascular plexus separately, OCTA has become an important tool for studying retinal vascular diseases such as MacTel, RVO, DR and others. For example, OCTA was used to demonstrate that the superficial and deep retinal capillary plexuses may be affected differently by retinal vascular diseases such as retinal artery and vein occlusion.13-15
Another advantage of OCTA is its superior ability to precisely delineate the vessels surrounding the foveal avascular zone (See Figure 1). In contrast, perifoveal leakage of fluorescein dye may blur the FAZ margins, and FA is limited to primarily delineating the superficial vascular plexus. Increased FAZ size has been correlated to reduced visual acuity prognosis in eyes with retinal vascular disease.16 A recent study demonstrated that the perifoveal intercapillary area on OCTA appears to increase in size as the level of diabetic retinopathy progresses.17 Also, neovascularization of the optic nerve, which can occur in proliferative DR and ischemic retinopathy, can be easily detected using OCTA by viewing the inner retina/optic nerve surface at the most superficial level (See Figure 2).18
It’s been hypothesized that OCTA may show retinal microcirculation impairment in the macula prior to the development of retinopathy.19 One study demonstrated that diabetic individuals without clinical evidence of diabetic retinopathy have larger FAZ sizes as well as FAZ remodeling and subtle capillary non-perfusion compared to normal, non-diabetic control eyes.20 Microaneurysms imaged with FA may not always be apparent on OCTA. This may result from stagnant erythrocytes blocking flow, or the flow in the microaneurysms may be below the threshold of detection of OCTA. Microaneurysms on FA may also correspond to capillary loops or preretinal neovascular tufts on OCTA.21
OCTA for Chorioretinal Disease
Evaluation of CNV using OCTA is one of the most important applications of this modality.4,22,23 OCTA can be used to determine the location of the CNV, its morphology and its response to anti-VEGF therapy. As a baseline, in a normal eye the “outer retina” OCTA scan segmenting between the outer plexiform layer and Bruch’s membrane will be devoid of signal, as there’s no vascular flow in this layer. However, this may not be the case in eyes with CNV.
There are different CNV morphologies on OCTA, but the clinical relevance of this is yet to be determined. CNV can be identified as several small filamentous vessels that form anastomoses (known as “seafan” or “lacy-wheel”), or as vessels associated with a central trunk of vessels (“Medusa”) (Figure 3). Recently, researchers analyzed OCTA patterns of CNV and their potential correspondence to quiescent and progressive CNV characteristics seen on multimodal imaging (FA, IGCA and spectral-domain OCT). In this study, the investigators imaged CNV using OCTA and graded it according to the presence or absence of the following features: well-defined CNV; presence of tiny capillaries; presence of anastomoses and loops; morphology of the vessels termini as opposed to a “dead tree” aspect; and presence of a hypo-intense halo surrounding the CNV. They found a 95-percent agreement between OCTA and the traditional multimodal imaging protocol for a “treatment required” decision in eyes with at least three out of the five features. There was a 91-percent agreement for “treatment not required” when patients presented with fewer than three features.24
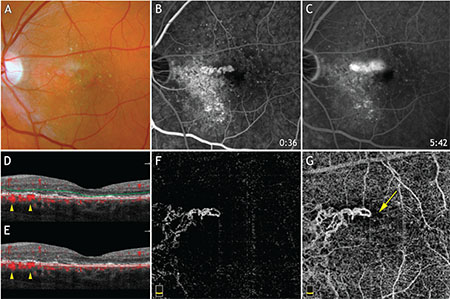 |
| Figure 3. Multimodal imaging of a 63-year-old patient with choroidal neovascularization secondary to central serous chorioretinopathy. (A) Color photograph shows a subretinal hemorrhage at the center of the macula surrounded by retinal pigment epithelium clumps. Early (B) and late-phase (C) fluorescein angiography show leakage from CNV. (D) and (E) represent corresponding OCT B-scan segmentation of the outer retinal and choriocapillaris, respectively. Yellow arrowheads point to the decorrelation signal below the RPE detachment suggestive of CNV. (F) OCT angiogram segmented at the level of the outer retina reveals CNV. (G) OCT angiogram segmented at the level of the choriocapillaris. The yellow arrow highlights the hypo-intense halo surrounding the CNV. |
Depending on the clinical presentation, OCTA’s sensitivity to visualize abnormal vascular networks may vary. When massive hemorrhage, exudate or fibrotic tissue is present, OCTA signals can be blocked, limiting visualization of CNV. Because CNV secondary to chronic CSCR and myopic CNV are rarely associated with large subretinal hemorrhages that limit penetration of the OCT signal, the abnormal vascular network may be identified with a screening OCTA in such cases.
Choroidal neovascularization can be classified according to the abnormal vascular plexus location in various retinal and choroidal layers: below the retinal pigment epithelium (type 1 CNV); above the RPE (type 2 CNV); and intraretinal (type 3 CNV). On OCTA, CNV classification depends on where the evidence of a vascular decorrelation signal is located. It can be located immediately above the RPE (type 2 CNV), and between Bruch’s membrane and the RPE (type 1 CNV).24 Since there can be OCT signal attenuation from the RPE-Bruch’s membrane complex,25,26 it’s possible that visualization beneath the RPE may influence the type 1 CNV identification. Thus, differences in signal penetration may play an important role in this imaging modality.
SD vs. SS OCTA for CNV
There are currently three FDA-approved OCTA systems. Two of them use a spectral-domain platform operating at ~840-nm wavelength. The third uses swept-source OCTA technology with a longer ~1050-nm wavelength and is in limited commercial release in the United States. It’s possible that SS-OCTA may be less affected by ocular opacity and allow a deeper penetration into the choroid.25-28
SS-OCTA also has less variation in sensitivity with depth (sensitivity roll-off) compared to SD-OCTA. In SD-OCTA, the imaging system is most sensitive to signals coming from reflectors close to what is known as the zero-delay line; as a reflector is moved away from the zero-delay, the system becomes less sensitive to the back-reflected signals.29 This sensitivity roll-off is due to the limited spectral resolution of the spectrometers that are used in SD-OCTA systems. In contrast, SS-OCTA systems don’t use spectrometer-based detection. Instead, in SS systems, it’s the instantaneous linewidth of the swept light source, along with the analog-to-digital acquisition rate, that determine the sensitivity roll-off of the system. This difference enables SS-OCTA systems to have improved sensitivity roll-off compared to SD-OCTA systems,30 which improves visualization of the choroid both on cross-sectional as well as en face OCTA imaging, and may also improve visualization of CNV, especially the sub-RPE component of the membrane.31
Limitations of OCTA
There are some limitations to OCTA imaging in its current configuration. One drawback is its restricted field of view (See Figure 4). The automated scan protocols that are currently available are 2x2 mm, 3x3 mm, 6x6 mm and 8x8 mm. In order to generate an OCT angiogram, OCT scans need to be repeated at least twice at each position in a volumetric raster scan. However, since the number of cross-sectional OCT scans is limited by the scanning speed of the instrument, a larger field of view will have reduced density and resolution.
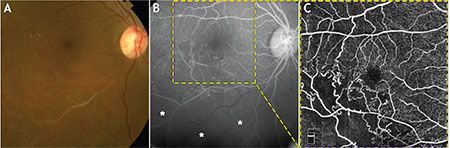 |
| Figure 4. Multimodal imaging of a 54-year-old patient with branch retinal vein occlusion of the inferior temporal arcade. (A) Color fundus photography shows non-perfused vessels and hyperpigmented laser scars on the inferior temporal retina. (B) Fluorescein angiography. Yellow-dashed frame identifies the corresponding 6x6-mm area imaged on optical coherence tomography angiography. White asterisks denote an ischemic retina not captured due to the small-field coverage. (C) En face OCTA segmented at the superficial retinal plexus shows a non-perfused area of the retina temporal-inferior to the fovea. |
Additionally, superficial vessels may erroneously appear in segmented views of deeper layers, such as the outer retina and choriocapillaris.32 These are termed “projection artifacts,” and may lead to incorrect diagnosis if not promptly identified (i.e., the retinal vessels’ projection may be misinterpreted as CNV).
Another issue is that commercially available OCTA devices are currently expensive, although this may change as the technology becomes more common. Also, there is currently no code beyond the standard OCT imaging code with regards to reimbursement. Finally, in terms of utility, while OCTA provides additional clinical information for the practitioner, such as noninvasive identification of CNV and retinal vascular abnormalities, its usefulness with regards to therapeutic monitoring is unclear.
FA and ICGA demonstrate the dynamic properties of dye within vascular networks, such as leakage in disorders that produce vascular incompetence and exudation, and dye-based angiography is still an important tool for diagnosis and management of chorioretinal diseases, especially when assessing the periphery of the fundus. OCTA is a promising technology, however, as it allows simultaneous assessment of both structural and vascular flow. OCTA is also safer, faster, more easily repeated and more comfortable for the patient than dye-based angiography, and it may provide more detailed information about blood flow in retinal and choroidal vasculature. The ability to rapidly obtain images of vascular plexuses and assess the integrity of retinal and choroidal perfusion should prove invaluable as a screening and diagnostic strategy for chorioretinal disorders.
Dr. Novais is a retinal specialist in the department of ophthalmology at the Federal University of São Paulo in Brazil and was, until recently, a postdoctoral research fellow at the New England Eye Center at Tufts Medical Center. Dr. Baumal is an associate professor of ophthalmology at Tufts University School of Medicine in Boston. She may be reached at the New England Eye Center at Tufts Medical Center, 800 Washington St, Box 450, Boston, MA 02116. Email: cbaumal@tuftsmedicalcenter.org, phone: 617-636-7950 or fax: 617-636-4866.
The authors have no financial interest in any of the products mentioned.
1. Stanga PE, Lim JI and Hamilton P. Indocyanine green angiography in chorioretinal diseases: Indications and interpretation: An evidence-based update. Ophthalmology 2003; 110:15-21;quiz 22-13.
2. Spaide RF, Klancnik JM, Cooney MJ. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA ophthalmology 2015;133:45-50.
3. Ha SO, Kim DY, Sohn CH, Lim KS. Anaphylaxis caused by intravenous fluorescein: Clinical characteristics and review of literature. Intern Emerg Med 2014;9:325-330.
4. de Carlo TE, de Carlo TE, Bonini Filho MA, Chin AT, et al. Spectral-domain optical coherence tomography angiography of choroidal neovascularization. Ophthalmology 2015;122:1228.
5. Jonathan E, Enfield J, Leahy MJ. Correlation mapping method for generating microcirculation morphology from optical coherence tomography (OCT) intensity images. J Biophotonics 2011;4:583-587.
6. An L, Wang RK. In vivo volumetric imaging of vascular perfusion within human retina and choroids with optical micro-angiography. Optics express 2008;16:11438-11452.
7. Jia Y, O Tan, J Tokayer, et al. Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Optics Express 2012;20:4710-4725.
8. Fingler J, D Schwartz, C Yang, SE Fraser. Mobility and transverse flow visualization using phase variance contrast with spectral domain optical coherence tomography. Optics express 2007; 15:12636-12653.
9. Kim DY, J Fingler, JS Werner. In vivo volumetric imaging of human retinal circulation with phase-variance optical coherence tomography. Biomedical Optics Express 2011;2:1504-1513.
10. White B, Pierce MC, Nassif N, et al. In vivo dynamic human retinal blood flow imaging using ultra-high-speed SD-OCT. Optics Express 2003;11:3490-3497.
11. Leitgeb RA, Werkmeister RM, Blatter C, et al. Doppler optical coherence tomography. Prog Retin Eye Res 2014;41:26-43.
12. Snodderly DM, Weinhaus RS, Choi JC. Neural-vascular relationships in central retina of macaque monkeys (Macaca fascicularis). J Neurosci 1992;12:1169-1193.
13. Sarraf D, Rahimy E, Fawzi AA, et al. Paracentral acute middle maculopathy: a new variant of acute macular neuroretinopathy associated with retinal capillary ischemia. JAMA Ophthalmol 2013;131:1275-1287.
14. Freiberg FJ, Pfau M, Wons J, et al. Optical coherence tomography angiography of the foveal avascular zone in diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 2016;254:6:1051.
15. Bonini Filho MA, Adhi M, de Carlo TE, et al. Optical coherence tomography angiography in retinal artery occlusion. Retina 2015;35:2339.
16. Parodi MB, Visintin F, Della Rupe P, et al. Foveal avascular zone in macular branch retinal vein occlusion. Int Ophthalmol 1995;19:25-28.
17. Salz DA, de Carlo TE, Adhi M, et al. Select features of diabetic retinopathy on swept-source optical coherence tomographic angiography compared with fluorescein angiography and normal eyes. JAMA Ophthalmol 2016;134:644-650.
18. de Carlo TE, Bonini Filho MA, Baumal CR, et al. Evaluation of preretinal neovascularization in proliferative diabetic retinopathy using optical coherence tomography angiography. Ophthalmic Surg Lasers Imaging Retina 2016;47:115-119.
19. Takase N, Nozaki M, Kato A, et al. Enlargement of foveal avascular zone in diabetic eyes evaluated by en face optical coherence tomography angiography. Retina 2015;35:2377.
20. de Carlo TE, Chin AT, Bonini Filho MA, et al. Detection of microvascular changes in eyes of patients with diabetes but not clinical diabetic retinopathy using optical coherence tomography angiography. Retina 2015;35:2364-2370.
21. Hwang TS, Jia Y, Gao SS, et al. Optical coherence tomography angiography features of diabetic retinopathy. Retina 2015;35:11:2371-6.
22. Kuehlewein L, Bansal M, Lenis TL et al. Optical coherence tomography angiography of type 1 neovascularization in age-related macular degeneration. Am J Ophthalmol 2015;160:4:739.
23. Baumal CR, de Carlo TE, Waheed NK, et al. Sequential optical coherence tomographic angiography for diagnosis and treatment of choroidal neovascularization in multifocal choroiditis. JAMA Ophthalmol 2015;133:1087-1090.
24. Coscas GJ, Lupidi M, Coscas F, et al. Optical coherence tomography angiography versus traditional multimodal imaging in assessing the activity of exudative age-related macular degeneration: A new diagnostic challenge. Retina 2015;35:2219.
25. Saito M, Iida T, Nagayama D. Cross-sectional and en face optical coherence tomographic features of polypoidal choroidal vasculopathy. Retina 2008;28:459-464.
26. Ueno C, Gomi F, Sawa M, et al. Correlation of indocyanine green angiography and optical coherence tomography findings after intravitreal ranibizumab for polypoidal choroidal vasculopathy. Retina 2012;32:2006-2013.
27. Povazay B, Hermann B, Unterhuber A, et al. Three-dimensional optical coherence tomography at 1050 nm versus 800 nm in retinal pathologies: Enhanced performance and choroidal penetration in cataract patients. J Biomed Opt 2007;12:041211.
28. Unterhuber, Povazay B, Hermann B A, et al. In vivo retinal optical coherence tomography at 1040 nm—enhanced penetration into the choroid. Optics Express 2005;13:3252.
29. Imamura Y, Fujiwara T, Margolis R, Spaide RF, et al. Enhanced depth imaging optical coherence tomography of the choroid in central serous chorioretinopathy. Retina 2009;29:1469-1473.
30. Grulkowski I, Liu JJ, Potsaid B, et al. Retinal, anterior segment and full eye imaging using ultrahigh speed swept-source OCT with vertical-cavity surface emitting lasers. Biomedical Optics express 2012;3:2733-2751.
31. Novais EA, Adhi M, Moult EM, et al. Choroidal neovascularization analyzed on ultra high-speed swept-source optical coherence tomography angiography compared to spectral-domain optical coherence tomography angiography. Am J Ophthalmol 2016;164:80-88.
32. de Carlo TE, Romano A, Waheed NK, Duker JS, et al. A review of optical coherence tomography angiography (OCTA). International Journal of Retina and Vitreous 2015;1:1:1-15.



