Posterior Vitreous Detachment
Posterior vitreous detachment is the critical event leading to the development of retinal tears and retinal detachment. PVD occurs when the posterior vitreous separates from the retina and collapses anteriorly toward the vitreous base. This event is the result of vitreous syneresis (liquefaction). PVD usually follows a benign course. However, when the separating vitreous remains firmly adherent to an area of the retina, localized vitreoretinal traction results. The PVD may produce retinal breaks in areas of firm vitreoretinal attachments and on narrow posterior extensions of the vitreous base. Mild traction causes tenting of the retina. With additional traction, a horseshoe tear (flap tear) develops. With still further traction, the flap may separate from the surrounding retina, leaving a round or oval hole with an overlying operculum.
Typical symptoms of PVD are "flashes and floaters." Photopsia, the subjective impression of flashing lights, is caused by vitreous traction on the peripheral retina. Floaters are described as "cobwebs," "spots," or "hair" in the field of vision. They are caused by vitreous opacities, such as epipapillary glial tissue torn from the optic disc, condensations of vitreous collagen, and/or blood. There are no symptoms that can distinguish a PVD alone from a PVD with an associated retinal break. If a RD is also present, patients may complain of a "curtain," "shade," or "shadow" obscuring their peripheral vision.
Patients with acute symptomatic PVD without peripheral retinal breaks require no immediate treatment but may be examined in one to two weeks, since some retinal breaks appear to develop days to weeks after the onset of symptoms. The development of RD is unlikely if no retinal tears are present at four to six weeks follow-up.2 All patients are counseled and instructed to return immediately if they have a significant change in their symptoms, such as an increase in photopsias and/or floaters, which might indicate progression of PVD with a retinal tear, or a "shade" over their vision, which may indicate RD.
Symptomatic Retinal Breaks
A symptomatic retinal break is defined as a break in the presence of new or increased flashes and/or floaters. Ten to 15 percent of patients with acute symptomatic PVD are found to have a retinal tear.3,4 Vitreous hemorrhage is an important sign. When present, there is a 70-percent incidence of retinal tears, compared with a 2 to 4 percent incidence without hemorrhage.5 Hemorrhage results from the avulsion of superficial retinal or prepapillary vessels or from torn retinal vessels that cross retinal tears. In some cases, an intact vessel may cross over a retinal tear, and continued vitreoretinal traction on the bridging vessel may cause vitreous hemorrhage to occur later.
|
|
|
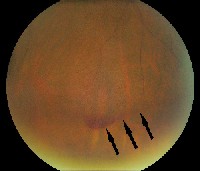
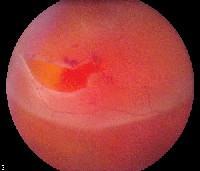
| A small, layered preretinal hemorrhage at inferior vitreous base in a patient with floaters and an acute PVD. |
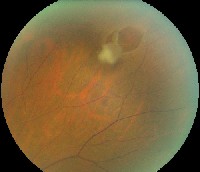
The same patient with a superior peripheral retinal tear. |
Acute symptomatic retinal breaks resulting from a PVD may result in RD. Acute symptomatic horseshoe tears require prompt treatment in an attempt to prevent RD.2,5 Thirty to 50 percent of acute symptomatic horseshoe tears have been found to result in RD, and prophylactic treatment reduces the frequency of RD to 5 percent.2,6 Symptomatic operculated breaks typically do not progress to RD unless residual vitreous remains adherent to retinal vessels adjacent to the break;2 however, most retinal specialists treat these lesions regardless of the status of the vitreous.
The goal of treatment is to create a chorioretinal adhesion surrounding a break so that no communication exists between the vitreous cavity and the subretinal space. While either cryopexy or laser can be used, laser is generally the preferred method. Cryotherapy is used in cases in which media opacity precludes transmission of laser energy. Barrier laser is delivered at the slit lamp using a contact lens or with the laser indirect ophthalmoscope using scleral depression. Breaks and adjacent subretinal fluid should be completely surrounded with three to four rows of almost confluent laser spots, 500 µm in size. In eyes with peripheral horseshoe tears, treatment must extend anteriorly to the ora serrata since persistent vitreoretinal traction can pull the flap through a cryopexy or laser scar and cause an RD.7 Patients should be followed closely after treatment since retinal breaks in new locations develop in 8 percent and vitreous hemorrhage occurs in 4 percent (due to an avulsed vessel).7
|
|
|
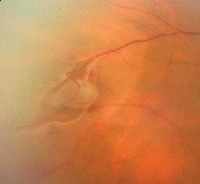
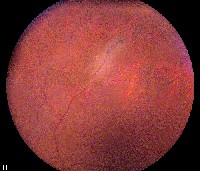
| A bridging vessel over a retinal tear with subretinal fluid. | Laser demarcation surrounding an area of subretinal fluid. |
Asymptomatic operculated holes and atrophic round holes very rarely lead to detachment. Eyes with signs and symptoms of acute PVD may have atrophic retinal breaks unrelated to vitreoretinal traction, and these breaks are considered to be pre-existing and not symptomatic. Asymptomatic operculated holes and atrophic round holes are generally not prophylactically treated; however, if cataract surgery is planned, treatment may be considered.2
Horseshoe tears and operculated holes may lead to a subclinical retinal detachment (SCRD), which is defined as a retinal break with surrounding subretinal fluid extending at least 1 disc diameter (DD) away from the break, but no more than 2 DD posterior to the equator. If there is less than 1 DD of fluid, then it is a break without detachment.2
There is some data to suggest that eyes with subclinical detachment are more likely to progress to clinical RD than eyes with breaks without detachment.2 In one study, untreated asymptomatic SCRD were compared with untreated asymptomatic retinal breaks. In phakic eyes, 31 percent of the SCRD progressed to clinical RD, whereas only 5 percent of the retinal breaks progressed to a clinical RD. The author recommended consideration of prophylactic treatment for SCRD.2 A more recent study, however, found progression to clinical RD in only 11 percent of phakic patients with asymptomatic SCRD, and an equal number of patients with SCRD (11 percent) had spontaneous regression and disappearance. The author recommended careful follow-up and monitoring of the SCRD, rather than prophylactic treatment.8
Lattice Degeneration
Lattice degeneration is one of the most important vitreoretinal abnormalities known to predispose a patient to RD. The condition is bilateral in 45 percent of cases,9 and patients with lattice more frequently have myopia than the general population. Approximately 30 percent of patients with retinal detachment also have lattice degeneration. Because the prevalence of lattice is high in the general population (7 to 8 percent in clinical series, 11 to 16 percent in autopsy series),9 the lifetime risk of a RD in a patient with lattice is less than 1 percent.10
Clinically, lattice lesions appear as sharply defined, round or oval areas of retinal thinning or irregularity. Variable features of lattice lesions include abnormal pigmentation (present in 82 percent), yellow-white surface flecks (present in 80 percent) and branching white lines, which represent occluded or sheathed vessels (present in 17 percent). Atrophic retinal holes may occur within lattice (present in 25 to 35 percent) and result from progressive retinal thinning,10 and tractional retinal tears (present in 1 to 2 percent) may occur at the lateral edges or the posterior margin of the lattice lesion. Lattice lesions usually are circumferentially oriented and located between the 11 to 1 o'clock and 5 to 7 o'clock meridians, anterior to the equator. They are best seen using binocular indirect ophthalmoscopy with scleral depression. In certain systemic conditions (Stickler's syndrome), lattice may be perivascular and radially oriented.
|
|
|
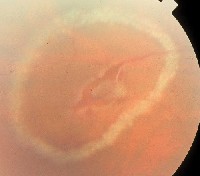
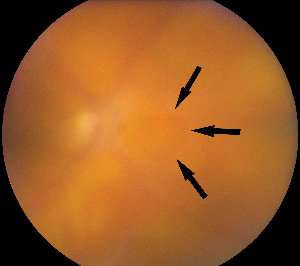
| A large, tractional retinal tear at the edge of lattice with associated retinal detachment. | A radial, perivascular lattice in a patient with Stickler's syndrome. |
The management of lattice degeneration is based on an increased understanding of the natural history of this condition. For lattice degeneration without retinal breaks, the risk of RD is very low (less than 1 percent), and prophylactic therapy is not indicated. For lattice degeneration with atrophic round holes, the incidence of clinical or progressive subclinical detachment is approximately 2 percent.10 Prophylactic therapy for atrophic holes in lattice is not indicated; if subclinical detachment is present, treatment is indicated only if the patient cannot be followed at regular intervals on a long-term basis.10 When retinal detachment is caused by round atrophic holes, it is unrelated to posterior vitreous detachment and usually occurs in patients younger than 30 years of age.
For lattice degeneration with tractional tears, treatment is dependent on a careful history. If asymptomatic (patient denies flashes and floaters), treatment is indicated in aphakic eyes, fellow eyes of patients with RD, and patients with a family history of RD. If symptomatic, prophylactic treatment is also indicated since these eyes have a relatively high risk of progression to RD. Tractional tears occur at the time of posterior vitreous separation, generally in patients older than 40 years of age.
For asymptomatic fellow eyes with lattice degeneration in patients with lattice-associated RD in the first eye, the benefit of prophylactic therapy is controversial. The rate of RD in untreated fellow eyes is approximately 5 percent11 (in patients with myopia and lattice, it may be has high as 25 percent). In one study, prophylactic treatment reduced the risk of RD to 1.8 percent over a seven-year period; there was, however, no additional benefit in the subgroups with extensive lattice (greater than 6 clock hours) or high myopia (6 D or more).11
Additional Conditions
Myopia is an independent risk factor for RD, since more than 50 percent of non-traumatic RD's occur in myopic eyes. If the level of myopia is from 1 to 3 D, there is a fourfold increase in risk over the general population, and if myopia is greater than 3 D, there is a tenfold risk.12 There is no proven treatment to prevent RD in this patient population.
A cystic retinal tuft is an elevated, round or oval peripheral lesion composed primarily of glial tissue. The lesion appears sharply circumscribed and chalky white (unlike the translucency of a retinal flap). The tuft contains crypts of vitreous and has traction at its apex from condensed vitreous. These lesions occur in 5 percent of autopsy cases and are bilateral in 20 percent. They are associated with 6.5 to 10 percent of non-traumatic retinal detachments, but the risk of RD in a patient with a cystic retinal tuft is only 0.28 percent.13 Since the risk of RD is very low, prophylactic treatment is not recommended; however, careful monitoring of patients with cystic retinal tufts is required.
A zonular traction tuft is a thickened zonule that is displaced posteriorly and attached to the anterior retina. This results in an abnormal tuft of tissue drawn from the surrounding retinal surface toward the ciliary body. This lesion is usually found in the nasal quadrants and occurs in 15 percent of autopsy cases and is bilateral in 15 percent. With continual traction from the zonules on the tuft, thickening and degeneration occurs, and there is a 2.2 percent incidence of tears at the base of these lesions. These tufts do not require prophylactic treatment, but do require careful monitoring.14
Acquired (senile, degenerative) retinoschisis occurs when the cysts of typical peripheral cystoid degeneration or reticular peripheral cystoid degeneration coalesce, with resultant lamellar splitting of the retina. It is characterized by a smooth, dome-shaped elevation of the inner layer of the schisis cavity in the peripheral fundus and may be confused with a RD. These lesions occur in about 1 percent of the population and usually are bilateral in the inferotemporal quadrant. Outer layer holes occur in approximately 17 percent and inner layer holes occur in less than 4 percent.15 A progressive retinal detachment requires breaks in both the inner and outer retinal layers; this occurs very rarely.15 Less than 2.5 percent of RD are associated with retinoschisis16 and prophylactic therapy (barrier retinopexy) may not be beneficial, since the schisis may progress through the treated area or may develop in new areas.15 Treatment is indicated in retinoschisis when patients have symptomatic, progressive RD.
Another condition leading to retinal detachment is a giant retinal tear, defined as a retinal tear that extends 3 clock hours (90 degrees) or more around the circumference of the globe. A giant retinal tear typically develops slightly posterior and parallel to the ora serrata, with the vitreous base attached to the anterior margin of the tear. The vitreous is usually not attached to the posterior margin of the tear, allowing it to be relatively mobile; in some cases this results in an inverted retinal flap. In addition there may be a posterior radial extension at one or both ends of the giant tear. Giant retinal tears are particularly difficult to manage, and they are associated with a high rate of proliferative vitreoretinopathy. Non-traumatic giant retinal tears are also associated with a high rate of vitreoretinal pathology in the fellow eye. In natural history studies of fellow eyes, the incidence of retinal tears is 59 percent, (17 percent with giant retinal tears), and the incidence of RD is 18 percent.17 In one series of fellow eyes prophylactically treated with 360 degree cryotherapy, the incidence of retinal tears was reduced to 8 percent (2 percent with giant retinal tears), and the incidence of RD was 6 percent over a seven-year period.18 Some authors recommend a prophylactic scleral buckle in fellow eyes of patients with a particularly high risk of bilateral giant retinal tears (high myopes, Stickler's syndrome).17 Since giant retinal tears are associated with significant visual morbidity, consideration may be given to prophylactic therapy in the fellow eye.
We are indebted to the work of the many investigators who have meticulously studied the risk factors and precursors of RD. Although retinal surgeons differ in their approach to prophylactic treatment of vitreoretinal abnormalities, we can offer evidence-based advice to our patients at risk for RD by understanding the natural history of these conditions and the results of prophylactic treatment. Additional studies will shed some light on these issues, but the debate over prophylaxis will likely continue for some time.
Dr. Adrean is a vitreoretinal Fellow at Kresge Eye Institute, Wayne State University School of Medicine. Dr. Eliott is associate professor of ophthalmology and director of the Vitreoretinal Service at Kresge. Contact Dr. Eliott at Kresge Eye Institute, 4717 St. Antoine, Detroit, Mich. 48201. Phone: (313)993-0871, fax: (313)577-2905, E-mail: deliott@med.wayne.edu.
1. Wilkes SR, Beard CM, Kurland LT, Robertson DM, O"Fallon WM. The incidence of retinal detachment in Rochester, Minnesota, 1970-1978. Am J Ophthalmol 1982;94:670-3.
2. Davis MD. Natural history of retinal breaks without detachment. Arch Ophthalmol 1974;92:183-94.
3. Linder B. Acute posterior vitreous detachment and its retinal complications. Acta Ophthalmol 1966 Suppl 87. 1-108.
4. Tasman WS. Posterior vitreous detachment and peripheral retinal breaks. Trans Am Acad Ophthalmol Otolaryngol 1968;72:217-24.
5. Benson WE. Retinal detachment: Diagnosis and Management, Ed 2. Philadelphia: Harper & Row Publishers Inc, 1988:4.
6. Colyear BH Jr, Pischel DK. Clinical tears in the retina without detachment. Am J Ophthalmol 1956;41:773-92.
7. Robertson DM, Norton EW. Long-term follow-up of treated retinal breaks. Am J Ophthalmol 1973;75:395-404.
8. Byer NE. Subclinical retinal detachment resulting from asymptomatic retinal breaks: Prognosis for progression and regression. Ophthalmology 2001;108:1499-503.
9. Foos RY, Simons KB. Vitreous in lattice degeneration of retina. Ophthalmology 1984;91:452-7.
10. Byer NE. Long-term natural history of lattice degeneration of the retina. Ophthalmology 1989;96:1396-401.
11. Folk JC, Arrindell EL, Klugman MR. The fellow eye of patients with phakic lattice retinal detachment. Ophthalmology 1989;96:72-9.
12. The Eye Disease Case-Control Study Group. Risk factors for idiopathic rhegmatogenous retinal detachment. Am J Epidemiol 1993;137:749-57.
13. Byer NE. Cystic retinal tufts and their relationship to retinal detachment. Arch Ophthalmol 1981;99:1788-90.
14. Foos R. Zonular traction tufts of the peripheral retina in cadaver eyes. Arch Ophthalmol 1969;82:620-32.
15. Byer NE. Long-term natural history study of senile retinoschisis with implications for management. Ophthalmology 1986;93:1127-37.
16. Hagler WS, Woldoff HS. Retinal detachment in relation to senile retinoschisis. Trans Am Acad Ophthalmol Otolaryngol 1973;77:OP99-113.
17. Freeman HM. Fellow eyes of non-traumatic giant retinal breaks. In: Ryan SJ, ed. Retina Vol 3: St. Louis: Mosby, 2001:2366-2370
18. Wolfensberger TJ, Aylward GW, Leaver PK. Prophylactic 360 degrees cryotherapy in fellow eyes of patients with spontaneous giant retinal tears. Ophthalmology 2003;110:1175-7.