Open globe injuries are a significant cause of vision loss worldwide, and the wide variety of presentations and severities can make them a challenge to manage. These injuries may be categorized as rupture, penetrating, perforating or an intraocular foreign body injury, and can lead to complications such as retinal detachment and traumatic endophthalmitis.1 While posterior segment open globe injuries have been associated with poorer outcomes, with appropriate management at each stage of repair it’s possible to improve vision for many patients with these injuries.2 In this article, we’ll delve into the steps for evaluating and treating posterior segment trauma.
Initial Evaluation
When a patient presents with an open globe injury, a detailed history may offer important clues about the nature and extent of the ocular damage. The mechanism of injury, including whether a blunt or sharp object caused the trauma, will help determine whether the injury resulted in a rupture or laceration. It’s essential to assess for the possibility of an intraocular foreign body; injuries from fireworks, explosives, nail guns, hammering metal and firearms are associated with an increased risk of IOFB injury. The velocity and size of the object and the material of the IOFB may help with preoperative planning. If the patient needed to pull the object out of the eye, there’s an increased risk of a posterior strike site and posterior segment trauma, even if the entry wound is only present anteriorly.
Once you’ve obtained a detailed history, conduct a thorough examination. Check visual acuity carefully, and note the presence or absence of an afferent pupillary defect. VA has been found to be the most important determinant of final visual outcome in penetrating eye injuries.3 Checking intraocular pressure is usually avoided to prevent expulsion of intraocular contents. On exam, 360-degree bullous subconjunctival hemorrhage and no posterior view warrants exploration, due to concern for an occult rupture.
Anterior segment findings of potential posterior segment IOFB include focal cataract or iris transillumination defect associated with a full-thickness corneal wound. Attempt to perform a dilated fundus exam, without scleral depression, to assess for posterior pathology and IOFB. Avoid putting pressure on the globe, since this could lead to further prolapse of intraocular contents. Assessment for endophthalmitis is also essential for preoperative planning; significant anterior chamber and/or vitreous inflammation is concerning and warrants intravitreal injection of antibiotics.
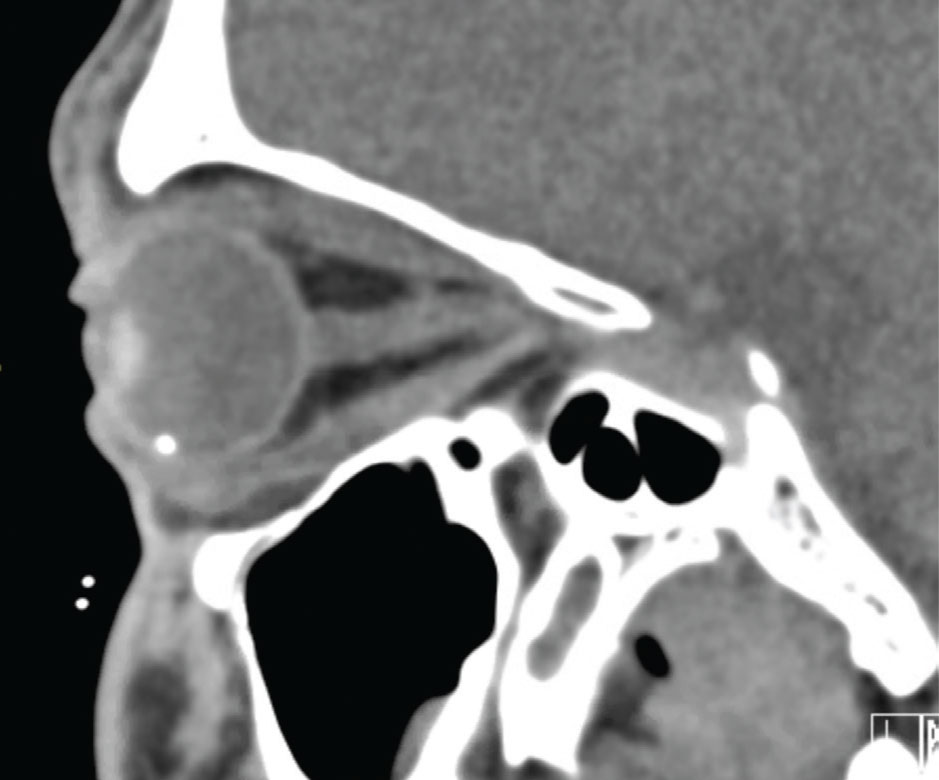 |
| Figure 1. CT scan of orbit with an intraocular foreign body. |
A computed tomography scan of the orbits with thin cuts is the imaging modality of choice to assess for an IOFB (Figure 1) and can sometimes support the diagnosis of an open globe injury, although a normal CT does not rule out the possibility of a full-thickness wound. An occult rupture on CT often appears as flattening of the posterior scleral contour.4 Other signs of open globe on CT include intraocular air or frank globe deformity. Magnetic resonance imaging may be more sensitive in detecting wood or plastic IOFB, but is generally avoided due to the risk of metallic IOFB causing further damage.5 It’s also preferable to avoid B-scan ultrasound, to minimize pressure on the globe. The clinician should then place a fox shield over the eye, start broad-spectrum systemic antibiotics, determine tetanus vaccination status and place the patient on bedrest to minimize pressure on the globe.
Primary Closure of an Open Globe
An open globe should be repaired within 24 hours of the injury to decrease the risk of traumatic endophthalmitis. Ideally, if there’s an IOFB you should remove it at the time of the primary surgery. The goal of the initial surgery is to have watertight closure of all wounds in a manner that will allow you to complete a secondary vitrectomy, if necessary. It’s preferable to use general anesthesia rather than periocular anesthesia, to minimize pressure on the globe. During induction, succinylcholine can cause contraction of extraocular muscles, and therefore should be avoided.
When repairing a corneal laceration, close it with sutures that are at least 90 percent deep in order to minimize posterior wound gape and postoperative corneal edema. Corneal glue may not provide strong enough closure and may hinder your view of the posterior segment. Perform a conjunctival peritomy to identify all scleral wounds. Unless prolapsed tissue is obviously contaminated or necrotic, reposition it with an instrument, such as an iris or cyclodialysis spatula, rather than excising it. Cut prolapsed vitreous flush against the globe to limit vitreoretinal traction and prevent incarceration. Closure of wounds from anterior to posterior will minimize the prolapse of intraocular contents. Don’t engage the underlying retinal or uveal tissue with scleral passes, in order to limit incarceration. If you can’t visualize the posterior-most edge of a zone-3 wound, you can leave it to close by secondary intention rather than disinserting extraocular muscles, as it can be difficult to isolate muscles for scleral buckle placement during the secondary surgery after they’re reinserted. The operative note must be detailed, including the location and length of the wound and whether a complete closure was achieved, so the secondary vitrectomy can be planned appropriately.
Management of an Intraocular Foreign Body
Ideally, you should remove IOFBs from the eye within 24 hours of the injury, at the time of primary closure.
First, close the entry wound to ensure that the eye will maintain adequate intraocular pressure during the IOFB removal. Then, perform a 23- or 25-gauge vitrectomy with elevation of the posterior hyaloid in order to decrease traction on the retina during IOFB removal. Prior to the object’s removal, you can use endolaser to surround the IOFB, since the visualization necessary to perform the laser might be limited by any intraocular hemorrhage that occurs because of manipulation of the IOFB.
For a magnetic IOFB extraction, you can use external magnets, intraocular forceps, soft-tip cannulas, the vitreous cutter and intraocular magnets.6 When there’s good visualization of the retina and the intravitreal foreign body, an external magnet held over a pars plana sclerotomy may be effective. However, the results are often best when the IOFB is removed with vitrectomy and an intraocular magnet,7 especially when visualization of the retina and IOFB is poor. It may be helpful to bring the IOFB anteriorly with an intraocular magnet and then transfer to a foreign body forcep that you can pull through a limbal or sclerotomy wound.8 If the IOFB has a lower specific gravity than perfluorocarbon liquid, PFO can also be used to lift the IOFB off of the retina for easier atraumatic grasping and may also protect the macula in case the IOFB is dropped.9 Make sure that the wound through which the IOFB will be removed is large enough to allow its passage. You can also inject intravitreal antibiotics—usually vancomycin and ceftazidime—at the time of IOFB removal for endophthalmitis prophylaxis.10
Postoperative Care After Primary Closure
If the patient’s vision is no light perception, the clinician should recheck it carefully with the brightest setting on the indirect ophthalmoscope to confirm this finding, as these eyes generally are poor candidates for subsequent intervention. Initiate a cycloplegic agent and frequent topical steroids to control postoperative inflammation. At each postop visit, especially when there is a limited view, perform a B-scan ultrasound to assess for vitreous or retinal traction, retinal detachment and/or hemorrhagic choroidal detachments. When there’s significant vitreous hemorrhage, proliferative vitreoretinopathy and tractional retinal detachments commonly develop later in the course of recovery, so you should pursue prophylactic vitrectomy in these cases even in the absence of retinal detachment on B-scan. Refer these patients to a retina specialist within the first week so that they can be evaluated in a timely fashion.
Secondary Vitrectomy: Indications and Timing
Many open globe injury eyes require secondary vitrectomy due to posterior segment complications. Indications for a secondary vitrectomy include persistent media opacity, progressive vitreoretinal traction, retinal detachment and endophthalmitis. Visual acuity usually needs to be light perception or better to warrant additional surgery. The only exception is if the eye has massive hemorrhagic choroidal detachments, since this is the one situation where the vision can actually improve from NLP.11
Favorable outcomes have been associated with recovery of light perception after initial closure and secondary vitrectomy in one to two weeks.12 Surgery within seven to 14 days allows adequate time for healing of the primary wounds, and also decreases the chance of encountering significant membrane proliferation during the secondary surgery. If there are hemorrhagic choroidal detachments, it’s best to wait until they’re liquified on B-scan before proceeding with secondary surgery, so that they can be thoroughly drained (Figures 2a and 2b).
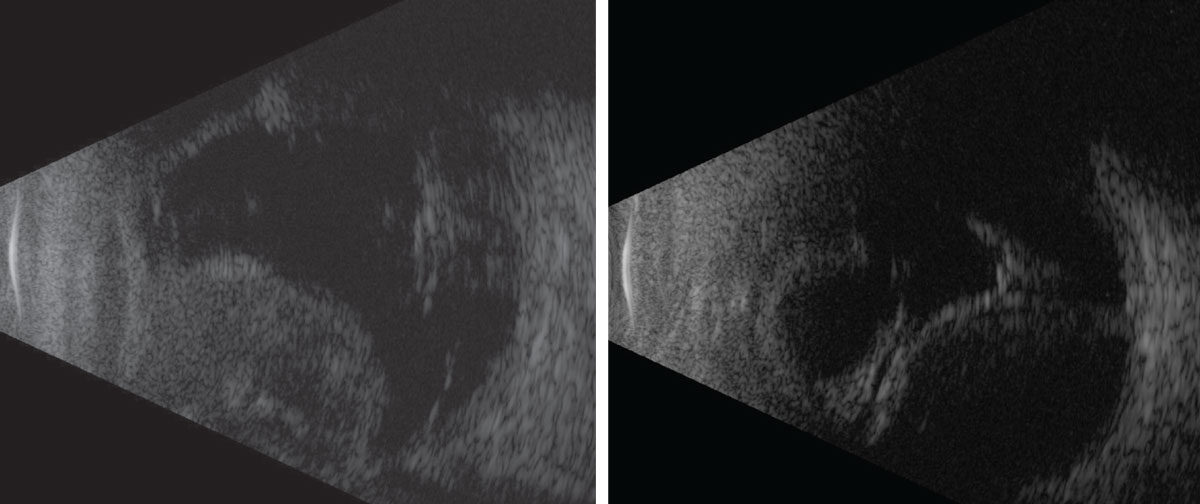 |
|
Figure 2a (left): Solid hemorrhagic choroidal detachments one day after open globe injury. Figure 2b (right): Liquified hemorrhage choroidal detachments 10 days after open globe injury. |
Secondary Vitrectomy Procedure
The main goals of the secondary vitrectomy are to clear the ocular media by either removing vitreous hemorrhage and/or cataract; to remove the scaffold from the scleral laceration site; to remove the posterior hyaloid; and to identify and treat retinal tears or detachment.6
The first step involves placing the infusion cannula. If there’s vitreous hemorrhage that prevents a view of the pars plana cannulas, initiate the infusion with an anterior chamber maintainer. To avoid subretinal or suprachoroidal placement, consider using a 6-mm long infusion cannula at the pars plana, rather than the traditional 4-mm cannula, particularly if there’s a known history of choroidal hemorrhage. In the presence of choroidal hemorrhages, a preoperative B-scan ultrasound should be used to assess for liquification of the choroidal hemorrhages and the location of the preferential drainage site, which should be the quadrant where the choroidal hemorrhage is the highest. You can drain the blood through a scleral incision while concurrently infusing balanced salt solution or air. A long radial sclerotomy is the preferred method, as it allows for drainage of residual clots as well.
Unless there are large choroidal hemorrhages, adjuvant placement of a scleral buckle is usually indicated. Traumatic retinal detachment is often associated with multiple retinal breaks and has a higher risk for PVR. An encircling band will support the vitreous base and can decrease the risk of future retinal detachment. In the case of a known preoperative total funnel retinal detachment where there’s a higher likelihood of needing a 360-degree retinectomy, the surgeon can hook and isolate the extraocular muscles and then perform the vitrectomy. If a 360-degree retinectomy is ultimately needed, then the buckle can be deferred. However, if there’s peripheral retina that will benefit from support, then the buckle can be placed easily, since the conjunctival peritomy and muscle isolation have already been completed.
In addition, if the lens is opacified or has been dislocated, it’ll need to be removed for adequate posterior segment visualization. To decrease the likelihood of anterior loop PVR, perform a lensectomy with total removal of the capsule. You can remove the lens with the vitrectomy probe or a fragmentation device through the pars plana.6
Once you’ve cleared the view to the posterior segment, you can then initiate the core vitrectomy, starting anteriorly and moving posteriorly. The main initial goal of the core vitrectomy is to find a plane between the retina and the hyaloid that can then be extended. A preoperative B-scan ultrasound may be helpful in identifying an area in the periphery where the retina appears attached and the hyaloid appears elevated.
If a natural space between retina and hyaloid isn’t apparent on B-scan, consider gradually using the cutter to remove more superficial vitreous hemorrhage, then burrowing down layer by layer towards the retina in a limited area where, if an inadvertent retinal break were created, it would be supported by the buckle and/or tamponade agent. Assess the peripheral retina for any retinal tears or detachments and treat with endophotocoagulation. It’s important to remove all preretinal membranes and assess for areas of retinal incarceration, the location of which can be predicted based on the laceration site noted in the primary repair. Once you’ve identified any retinal incarceration, perform a retinectomy to release it.
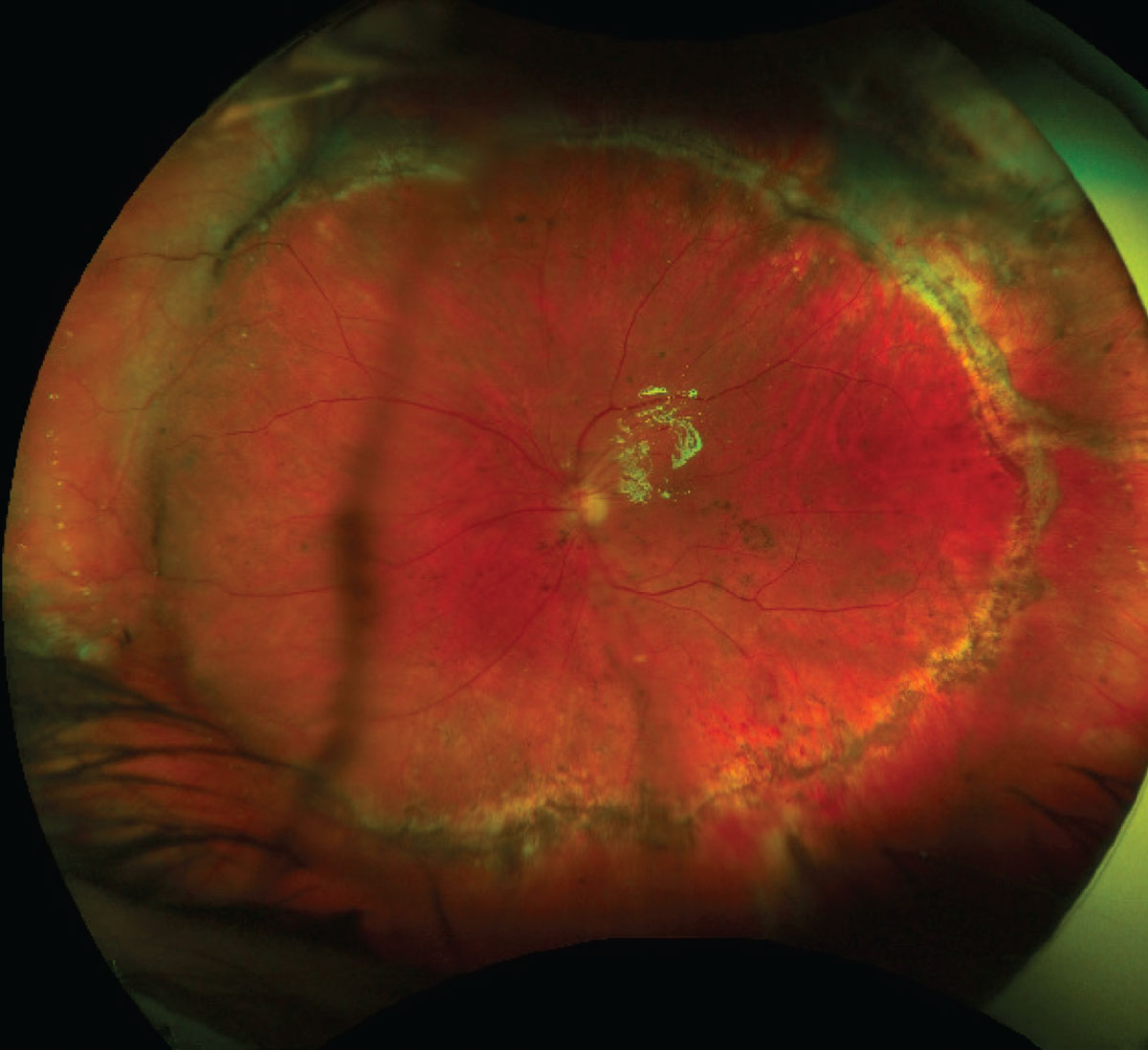 |
| Figure 3. Widefield fundus photo after an open globe injury and secondary vitrectomy to repair traumatic retinal detachment. The scleral buckle contour can be seen supporting the nasal retina, and the 270-degree retinectomy edge can be seen superiorly, temporally and inferiorly. |
When proliferative vitreoretinopathy causes retinal foreshortening, a large retinectomy may be required to reattach the retina (Figure 3). The retina can be flattened with perfluorocarbon liquid or fluid-air exchange with subsequent laser to the retinectomy edge.13
When deciding between gas versus silicone oil tamponade, consider using silicone oil, particularly with larger retinectomies and in eyes with anterior membrane proliferation. If the patient is aphakic with oil fill, you must perform an inferior peripheral iridotomy.
If the eye is aphakic and aniridic with oil fill, place silicone oil retention sutures across the plane of the iris to keep oil away from the cornea. Lastly, instruct the patient to position face down postoperatively.
Patient Counseling
Open globe injuries are a traumatic experience for everyone involved—patients, caretakers and surgeons. Patient counseling is a crucial component of the management of eye trauma and is essential in preparing the patient for the long journey to recovery and rehabilitation.
Patients naturally wish to know what their vision will be like after surgery, but it’s difficult to predict this with any certainty. When counseling patients, then, it’s important to strike a balance: Avoid being overly pessimistic, since some eyes can regain meaningful vision, but set realistic expectations. Most eyes with poor vision prior to secondary vitrectomy have limited vision postoperatively as well. However, there is a chance for improvement, and the best results will only be achieved if you enter each case mentally prepared to repair the eye to the best of your ability and to be meticulous about the repair at each step of the process.
Dr. Chee is an assistant professor of ophthalmology at the University of Washington in Seattle.
Dr. Tom is a vitreoretinal surgery fellow at the Retina Institute in St. Louis.
1. Kuhn F, et al. The Birmingham Eye Trauma Terminology System (BETT). Journal Français d’Ophtalmologie 2004;27:2:206–10.
2. Rahman I, et al. Open Globe Injuries: Factors predictive of poor outcome. Eye 2006;20:12:1336–41.
3. Sternberg P Jr, et al. Multivariate analysis of prognostic factors in penetrating ocular injuries. American Journal of Ophthalmology 1984;98:4. 467–72.
4. Sevel D, et al. Value of computed tomography for the diagnosis of a ruptured eye. Journal of Computer Assisted Tomography 1983;7:5:870–75.
5. Modjtahedi B, et al. Imaging characteristics of intraocular foreign bodies. Retina 2015;35:1:95–104.
6. Schachat AP, Recchia FM, Sternberg P. Chapter 114: Surgery for Ocular Trauma Principles and Techniques of Treatment. In: Ryan’s Retina E-Book. Elsevier Health Sciences, 2018. 2081-2106.
7. Mester V, Kuhn F. Ferrous intraocular foreign bodies retained in the posterior segment: Management options and results. International Ophthalmology 1998;22:6:355–62.
8. Sborgia G, et al. 25-gauge vitrectomy in open eye injury with retained foreign body. Journal of Ophthalmology Jan. 2017;3161680.
9. Guevara-Villarreal DA, Rodríguez-Valdés PJ. Posterior segment intraocular foreign body: Extraction surgical techniques, timing, and indications for vitrectomy. Journal of Ophthalmology;Nov 2016.Article ID: 2034509.
10. Ahmed Y, et al. Endophthalmitis following open-globe injuries. Eye 2012;26:2:212–17.
11. Chee YE, Kanoff JM, Eliott D. Remarkable visual recovery after severe open globe injury. AJO Case Reports 2016;3:34.
12. Feng K, et al. Case–control study of risk factors for no light perception after open-globe injury. Retina 2011;31:10:1988–96.
13. Shalaby, Khaled Abdel-Galil. Relaxing retinotomies in the management of retinal detachment with severe proliferative vitreoretinopathy (PVR). Clinical Ophthalmology 2010 Oct 5;4:1107-14.



