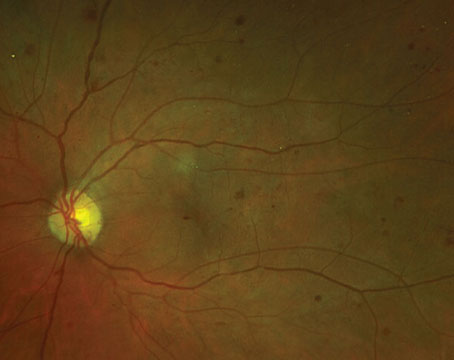
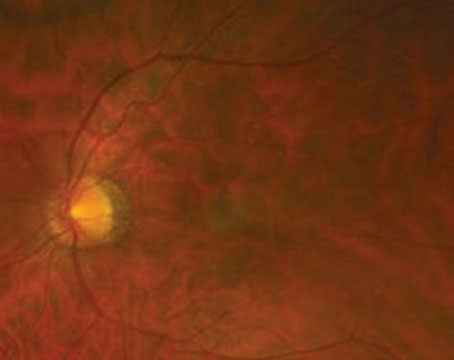
Age-related macular degeneration is a complex, multifactorial disease characterized by the degeneration of photoreceptors and retinal pigment epithelial cells with or without choroidal neovascularization. While significant progress has been made in the treatment of the exudative or wet form of AMD characterized by CNV, no therapy has been approved for the treatment of the nonexudative or dry form of AMD, which accounts for 85 percent of all AMD cases.1
While the exact causes of AMD remain unclear, factors such as aging, oxidative stress, light damage and genes are all known to play a significant role. Clinical manifestations of early AMD include the appearance of extracellular deposits of drusen between Bruch’s membrane and the RPE, visible clumps of pigment in the macula, and subretinal deposits of oxidized proteins and lipids (lipofuscin) within the RPE. For nonexudative AMD patients, RPE dysfunction progresses to RPE cell damage and death, potentially leading to regions of geographic atrophy (See Figure 1), scotopic sensitivity and photoreceptor cell death.2,3 Geographic atrophy and the accompanying retinal changes of dry AMD account for approximately 20 to 25 percent of severe visual loss and for a much larger portion of moderate visual loss in AMD patients.4,5
Recent biological and histological findings have supported an association between rod photoreceptor cells and nonexudative AMD. Symptoms of nonexudative AMD such as parafoveal scotomas related to GA and scotopic sensitivity can be rationally linked to rod photoreceptor cells because of the higher density of rod cells parafoveally (See Figure 2) and the primary role of rod photoreceptor cells in low-light vision.6 The disruption of rod photoreceptor cells is also thought to be associated with RPE cell damage due to the role of RPE cells in the regeneration of visual pigments for the vitamin A visual cycle in rods.6 Many novel treatments currently in preclinical development or in early-stage clinical trials target pathological schemes of rod photoreceptor cell metabolism in order to slow the progression of dry AMD and protect the RPE (See Table 1). The disease processes of the RPE are currently of particular interest because the Food and Drug Administration has recently begun approving treatments based on the reduction or maintenance of GA lesion size in the RPE, rather than basing drug approval on visual acuity improvements alone. Research has therefore become more focused on the development of GA rather than other pathological changes such as drusen accumulation and pigment clumping that are more difficult to measure and compare.
The Visual Cycle and AMD
Rod photoreceptor cells convert light energy into electrical signals through the visual cycle. In order to carry out this process, the photoreceptors of the retina are dependent on the metabolic support of the RPE for the regeneration of visual pigment and breakdown of photoreceptor byproducts.7,8 In rod photoreceptor cells of vertebrates, 11-cis-retinal generated in the RPE is isomerized by light to all-trans-retinal, which is then reduced to all-trans-retinol (See Figure 3). In order for the visual cycle to continue, the 11-cis-retinal must be regenerated in the RPE from all-trans-retinol with the help of the isomerase RPE65.3 Throughout this regeneration process, the toxic metabolite retinylidene-N-retinylethanolamine (A2E) is created as a byproduct within the outer segment disks of the rod photoreceptor cells. In patients suffering from AMD, A2E and other biretinoid compounds are not properly digested by the RPE and disrupt RPE cell function, thus causing RPE cell death and increasing the metabolic workload of adjacent RPE cells.9-11 This disease progression is discernible on fundus autofluorescence photography due to the distinct autofluorescent emission of A2E and related cytotoxic fluorophores (See Figure 4). Areas of increased FAF as associated with A2E accumulation have been observed both at the boundaries of GA lesions and in areas of future atrophic development, indicating that areas of increased FAF precede both development and enlargement of GA lesions.12-18
Pathway-Based Therapies
Developing therapies for dry AMD aim to slow A2E accumulation by targeting various steps within the visual cycle in order to slow the progression of GA and protect the photoreceptors. Two oral medications are currently in clinical testing in order to determine their safety and efficacy in the treatment of GA.
The first medication, the oral agent emixustat (ACU-4429; Acucela), is a small non-retinoid molecule that functions as a modulator of isomerase RPE65, which is required for the conversion of all-trans-retinol to 11-cis-retinal in the RPE. By inhibiting RPE65, emixustat reduces the levels of 11-cis-retinal and suppresses rod photoreceptor function and the visual cycle, thus theoretically reducing A2E accumulation and subsequent GA development and enlargement. Emixustat is specific to the visual cycle in rod photoreceptor cells and therefore should not cause the same vitamin-A deficiency systemic effects or ophthalmic side effects such as nyctalopia as do visual cycle modulators that act systemically.19
The Phase Ia study for emixustat has been completed (
Clinicaltrials.gov identifier: NCT00942240). Forty-six healthy male and female subjects were randomized to either a placebo group or one of five doses ranging from 2 mg up to 75 mg per day. The drug was well-tolerated and exhibited a dose-dependent modulation of rod ERG signals. A Phase IIb/3 clinical trial is currently under way to determine the efficacy of emixustat in patients with GA.
The second drug therapy involving visual cycle modulation is fenretinide (RT-101, ReVision Therapeutics). Fenretinide is an oral synthetic retinol derivative originally developed to treat diseases including rheumatoid arthritis, psoriasis and various cancers. These past studies have found limited efficacy but have demonstrated long-term safety and tolerability of fenretidine.20-25
In the normal physiological process, the retinol necessary for the regeneration of 11-cis-retinal is delivered to the RPE in a complex formed by RBP, retinol and transthyretin (TTR). In circulation, fenretinide competes with retinol for RBP binding and the subsequently formed RBP-fenretinide complex is removed by the kidneys through excreted urine, thus reducing the circulating quantity of RBP.26,27 The systemic inhibition of retinol by fenretinide is dose dependent and reversible upon drug cessation.20-23 Because the delivery of retinol to the RPE is uniquely dependent on the binding of retinol and RBP, fenretinide treatment preferentially reduces retinol concentrations in the retina rather than other areas of the body, although visual disturbances such as delayed dark adaption have commonly been reported as a side effect of fenretinide.20,22,23,27
Reducing the delivery of RBP-retinol to the RPE is theorized to reduce the accumulation of A2E and related compounds and thus slow GA lesion growth. A two-year Phase II proof-of-concept trial (Identifier: NCT00429936) was designed to determine whether fenretinide-mediated reductions of RBP would lead to a reduction of lesion growth in subjects with GA secondary to dry AMD. The study utilized two doses (100 mg and 300 mg) and a placebo group. For the placebo and 100-mg groups, no correlation was observed between reductions in lesion growth and reduction in RBP levels. However, for the 300-mg group there was a clear trend for reduced lesion growth rates in patients who achieved serum RBP levels ≤2 mg/dL. Fifty-one percent of 300 mg patients who completed the two-year study achieved this level of RBP reduction. Variabilities in the production of fenretinide are hypothesized to be partially responsible for inconsistent bioavailability of administered fenretinide and fluctuations in systemic results in the 300-mg dosage group. Additionally, fenretinide treatment at both the 100-mg and 300-mg doses was associated with a 45-percent reduction of CNV incidence relative to placebo.27 Therefore, fenretinide was found to have both dose-dependent properties for the reduction of GA lesion growth and dose-independent anti-angiogenic properties for the prevention of CNV if adequate drug bioavailability was achieved. Recently, Acucela acquired fenretinide and the combined efforts will ideally produce significant advances in visual cycle modulation and excess A2E reduction.
The theoretical mechanisms of visual cycle modulators such as fenretinide and emixustat is analogous to that of beta-adrenergic blocking agents (also known as beta-blockers) in congestive heart failure. Beta-blockers work by lowering blood pressure, thus reducing the heart rate of the patient and the force of each heartbeat. By reducing the workload of the heart, beta-blockers slow the progression of heart failure and reduce patient mortality.28 In a similar fashion, visual cycle modulators aim to reduce the workload of the RPE by decreasing rod photoreceptor metabolism and A2E accumulation, thus potentially slowing the progression of AMD and improving patient outcomes.
Byproduct Accumulation
The chronic inflammation associated with AMD is additionally associated with a second byproduct other than A2E—amyloid beta.29 Amyloid beta, a waste product of the central nervous system associated with diseases such as atherosclerosis and Alzheimer’s disease, has been found as a differentiating compound between dry AMD eyes and control age-matched donor eyes.30-32 Amyloid beta, which has been identified as a component of drusen, is a known activator of the complement system and may lead to damage in RPE cells and play a role in inflammation that contributes to AMD progression.32,33 The presence of proinflammatory molecules of the complement system within drusen may stimulate chronic inflammation in the RPE-Bruch’s membrane-choriocapillaris complex and induce vascular endothelial growth factor expression, thus explaining why soft drusen and theoretically amyloid beta accumulation are risk factors for CNV in AMD eyes.32,33
Reducing Accumulation
Amyloid diseases and drusen in AMD eyes are associated with abundant amyloid beta fibrils of various lengths. Two humanized anti-amyloid beta antibodies, RN6G (PF-4382923; Pfizer) and GSK933776 (GlaxoSmithKline), bind to amyloid beta 40/42 and amyloid beta 40 respectively in peripheral circulation in order to decrease its accumulation within the retina by sequestering the byproduct in the periphery. Animal mouse models have shown the efficacy of anti-amyloid beta antibodies in preserving photoreceptors and reducing retinal atrophy without disrupting normal photoreceptor function.34 Both drugs have successfully completed Phase I clinical trials (Identifiers: NCT00877032 and NCT01424436). GSK93376 is currently being tested for potential efficacy in a Phase II clinical trial (Identifier: NCT01342926) and the Phase II clinical trial for RN6G has recently resumed patient recruitment (Identifier: NCT01577381).
While the treatment of exudative AMD has transformed with anti-angiogenic therapies, therapeutic agents that prevent and delay the progression of dry AMD remain elusive. While current anti-angiogenic therapies address CNV, treatment for the underlying RPE changes and retinal atrophy is still under investigation. Ongoing clinical trials may shed light on new therapies basesd on the reduction of byproduct accumulation and the modulation of rod photoreceptor cell activity.
REVIEW
Ms. Maldonado is a clinical research technician at Retina Consultants of Houston and plans on attending Baylor College of Medicine. Dr. Brown is the director of the Greater Houston Retina Research Center and practices at Retina Consultants of Houston at the Methodist Hospital in Houston. Dr. Wykoff also practices at Retina Consultants of Houston at the Methodist Hospital after serving as chief resident and co-director of Ocular Trauma at Bascom Palmer Eye Institute. Contact Dr. Brown at Retina Consultants of Houston, 6560 Fannin, Ste. 750, Houston, Texas 77030; phone: (713) 524-3434; e-mail:
dmbmd@houstonretina.com.
Figure 2. Modified from Osterberg, G. Topography of the layer of rods and cones in the human retina. Acta Ophthalmol 1935:Suppl. 13:6, 1-102.
Figure 3. Modified from
http://lpi.oregonstate.edu/infocenter/vitamins/vitaminA/visualcycle.html Copyright 2010.
1. Seddon JM, Chen CA. Epidemiology of age-related macular degeneration. In: Ryan SJ, Hinton DR, Schachat AP, Wilkinson CP, eds. Retina. Medical Retina. Vol. 2. 4th ed. St. Louis: Mosby, 2005:1017-28.
2. Curcio CA. Photoreceptor topography in ageing and age-related maculopathy. Eye 2001;15:376-383.
3. Lamb TD, Pugh EN Jr. Dark adaptation and the retinoid cycle of vision. Prog Retin Eye Res 2004;23:307-380.
4. Klein R, Klein BE, Jensen SC, Meuer SM. The five-year incidence and progression of age-related maculopathy: The Beaver Dam Eye Study. Ophthalmology 1997;104:7-21.
5. Sunness JS. Geographic atrophy in Age Related Macular Degeneration (Berger JW, Fine SL, Maguire MG, eds.). St. Louis: Mosby, 1999;155.
6. Snodderly DM, Sandstrom MM, Leung I, Zucker C, Neuringer M. Retinal pigment epithelial cell distribution in central retina of rhesus monkeys. Invest Ophthalmol Vis Sci 2002;43:2815-2818.
7. Bok D. Retinal photoreceptor-pigment epithelium interactions: Friedenwald lecture. Invest Ophthalmol Vis Sci 1985;26:1659-1694.
8. Besharse JC, Defoe DM. Role of the retinal pigment epithelium in photoreceptor membrane turnover. In: Marmor MF, Wolfensberger TJ, eds. The Retinal Pigment Epithelium. New York: Oxford University Press, 1998:chap. 8.
9. Dorey CK, Wu G, Ebenstein D, et al. Cell loss in the aging retina. Relationship to lipofuscin accumulation and macular degeneration. Invest Ophthalmol Vis Sci 1989;30:1691-1699.
10. Mata NL, Weng J, Travis GH. Biosynthesis of a major lipofuscin fluorophore in mice and humans with ABCR- mediated retinal and macular degeneration. Proc Natl Acad Sci USA 2000;97:7154-7159.
11. Karan G, Lillo C, Yang Z, et al. Lipofuscin accumulation, abnormal electrophysiology, and photoreceptor degeneration in mutant ELOVL4 transgenic mice: A model for macular degeneration. Proc Natl Acad Sci USA 2005;102:4164-4169.
12. Holz FG, Bellman C, Staudt S, et al. Fundus autofluorescence and development of geographic atrophy in age-related macular degeneration. Invest Ophthalmol Vis Sci 2001;42:1051-1056.
13. Schmitz-Valckenberg S, Bültmann S, Dreyhaupt J, et al. Fundus autofluorescence and fundus perimetry in the junctional zone of geographic atrophy in patients with age-related macular degeneration. Invest Ophthalmol Vis Sci 2004;45:4470-4476.
14. Holz FG, Bindewald-Wittich A, Fleckenstein M, et al; FAM-Study Group. Progression of geographic atrophy and impact of fundus autofluorescence patterns in age-related macular degeneration. Am J Ophthalmol 2007;143:463-472.
15. Schmitz-Valckenberg S, Bindewald-Wittich A, Dolar-Szczasny J, et al. Fundus Autofluorescence in Age-Related Macular Degeneration Study Group. Correlation between the area of increased autofluorescence surrounding geographic atrophy and disease progression in patients with AMD. Invest Ophthalmol Vis Sci 2006;47:2648-2654.
16. Bindewald A, Schmitz-Valckenberg S, Jorzik JJ, et al. Classification of abnormal fundus autofluorescence patterns in the junctional zone of geographic atrophy in patients with age related macular degeneration. Br J Ophthalmol 2005;89:874-878.
17. Scholl HP, Bellmann C, Dandekar SS, et al. Photopic and scotopic fine matrix mapping of retinal areas of increased fundus autofluorescence in patients with age-related maculopathy. Invest Ophthalmol Vis Sci 2004;45:574-583.
18. Owsley C, Jackson GR, White M, et al. Delays in rod-mediated dark adaptation in early age-related maculopathy. Ophthalmology 2001;108:1196-1202.
19. Malpeli G, Folli C, Berni R. Retinoid binding to retinol-binding protein and the interference with the interaction with transthyretin. Biochim Biophys Acta 1996;1294:48-54.
20. Formelli F, Clerici M, Campa T, et al. Five-year administration of fenretinide: Pharmacokinetics and effects on plasma retinol concentrations. J Clin Oncol 1993;11:2036-2042.
21. Conley B, O’Shaughnessy J, Prindiville S, et al. Pilot trial of the safety, tolerability, and retinoid levels of N-(4-hydroxyphenyl) retinamide in combination with tamoxifen in patients at high risk for developing invasive breast cancer. J Clin Oncol 2000; 18:275-283.
22. Camerini T, Mariani L, De Palo G, et al. Safety of the synthetic retinoid fenretinide: Long-term results from a controlled clinical trial for the prevention of contralateral breast cancer. J Clin Oncol 2001;19:1664-1670.
23. Mariani L, Formelli F, De Palo G, et al. Chemoprevention of breast cancer with fenretinide (4-HPR): Study of long-term visual and ophthalmologic tolerability. Tumori 1996;82:444-449.
24. Baglietto L, Torrisi R, Arena G, et al. Ocular effects of fenretinide, a vitamin A analog, in a chemoprevention trial of bladder cancer. Cancer Detect Prev 2000;24:369-375.
25. Veronesi U, Mariani L, Decensi A, et al. Fifteen-year results of a randomized phase III trial of fenretinide to prevent second breast cancer. Ann Oncol 2006;17:1065-1071.
26. Berni R, Formelli F. In vitro interaction of fenretinide with plasma retinol-binding protein and its functional consequences. FEBS Lett 1992;308:43-45.
27. Mata N, Lichter J, Vogel R, Han Y, Bui T, Singerman L. Investigation of oral fenretinide for treatment of geographic atrophy in age-related macular degeneration. Retina 2013;33: 498-507.
28. Meghani SH, Becker D. Beta-blockers: a new therapy in congestive heart failure. Am J Crit Care 2001 Nov;10(6):417-27.
29. Johnson LV, Leitner WP, Rivest AJ, Staples MK, Radeke MJ, Anderson DH. The Alzheimer’s A beta-peptide is deposited at sites of complement activation in pathologic deposits associated with aging and age-related macular degeneration. Proc Natl Acad Sci USA 2002;99:11830-11835.
30. Anderson DH, Mullins RF, Hageman GS, Johnson LV. A role for local inflammation in the formation of drusen in the aging eye. Am J Ophthalmol 2002;134:411-431.
31. Isas JM, Luibl V, Johnson LV, et al. Soluble and mature amyloid fibrils in drusen deposits. Invest Ophthalmol Vis Sci 2010;51:1304-1310.
32. Dentchev T, Milam AH, Lee VM, Trojanowski JQ, Dunaief JL. Amyloid-beta is found in drusen from some age-related macular degeneration retinas, but not in drusen from normal retinas. Mol Vis 2003:9;184-190.
33. Hoh Kam J, Lenassi E, Jeffery G. Viewing ageing eyes: Diverse sites of amyloid beta accumulation in the ageing mouse retina and the up-regulation of macrophages. PLoS One 2010;5:e13127.
34. Ding JD, Johnson LV, Herrmann R, Farsiu S, Smith SG, Groelle M, et al. Anti-amyloid therapy protects against retinal pigmented epithelium damage and vision loss in a model of age-related macular degeneration. Proc Natl Acad Sci USA 2011;108(28):E279-87.