Descemet’s stripping automated endothelial keratoplasty and Descemet’s membrane endothelial keratoplasty have advantages over full-thickness corneal transplants, namely a decreased risk of tissue rejection, faster visual recovery and the potential for better vision.
According to Winston Chamberlain, MD, PhD, who is in practice in Portland, Oregon, the primary indication for corneal transplant surgery in the Western world is Fuchs’ dystrophy. “Historically, we have thought of it as being a Caucasian disease, but it’s actually probably present in many different populations,” he says. “The genetic variants that drive it may be different, and it probably accounts for about 60 percent of all corneal transplants now in the United States. Most physicians in the U.S. would choose to perform DMEK on a patient with Fuchs’ dystrophy.”
Here, experts review the latest approaches to these transplantation procedures.
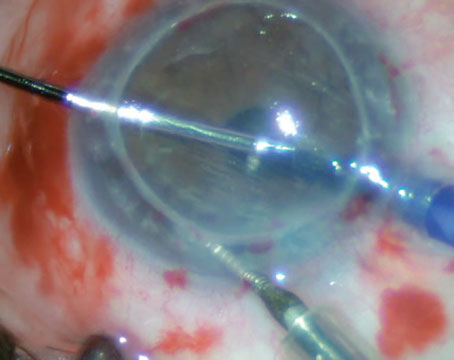
 |
| Figure 1. Insertion of DMEK graft with a Geuder cannula. (Nandini Venkateswaran, MD) |
Candidates for DMEK/DSAEK
Nandini Venkateswaran, MD, who is in practice in Boston, says DMEK is her go-to procedure for patients with Fuchs’ endothelial dystrophy or pseudophakic bullous keratopathy. These are often straightforward cases. “I tend to reserve DSAEK for more complex cases, such as patients who’ve had prior retinal detachment surgery or prior glaucoma surgery, who may be aphakic or have an anterior chamber intraocular lens, or who may have a more complex anatomic structure, such as iris defects and atypical anterior chamber depth, that may hinder a DMEK graft from successfully attaching in the eye,” she says. “As you incorporate both of these techniques in practice, a lot of your DMEK cases tend to go faster than your DSAEK cases because you self-select the tougher cases for DSAEK. For patients with an existing but edematous penetrating keratoplasty, both DSAEK and DMEK can work, but I tend to do more DSAEK in those scenarios.”
Clara Chan, MD, who is in practice in Toronto, agrees. “DMEK is the more anatomically correct way to replace the stripped diseased endothelium and Descemet’s membrane,” she says. “Tissue handling can be more challenging with DMEK, and being able to visualize inside the anterior chamber is crucial because the donor tissue is inserted as a scroll, which requires a variety of tapping maneuvers in a shallowed anterior chamber to open up and position properly. DSAEK more commonly has a role in complex eyes, such as those with prior vitrectomy, an anterior chamber intraocular lens, multiple iris defects, aniridia, aphakia, or advanced corneal edema such that the view into the anterior chamber is very poor.”
Tips and Techniques for DMEK
According to Dr. Chamberlain, the most common surgical method in the United States for DMEK is to use pre-loaded tissue, which has made the surgery accessible to more corneal surgeons. “This basically means that the eye bank does a lot of the work for the surgeon,” he says. “They cut the graft, peel it off, and put it in an injector/shooter. When the surgeon receives it, the injector/shooter is bathing in a media that supports the corneal cells. And then the surgeon just has to inject or pull it into the eye. There are a few injectors out there, and they are all about equivalent in terms of how much they damage the cells. The most popular ones are made of glass, and there is a bit of a trend toward using narrower openings.
“A Jones Tube-based injector was originally modified by Mike Straiko, MD, and more recently remodified by Gunther Weiss, to be narrower and go through a 2.2-mm wound,” Dr. Chamberlain continues. “The advantage is that you have smaller incisions with a more stable anterior chamber when injecting and manipulating the graft in the eye. One of the dangers of having an unstable chamber is that the graft can shoot back out through the wound, and that can damage the endothelial cells.”
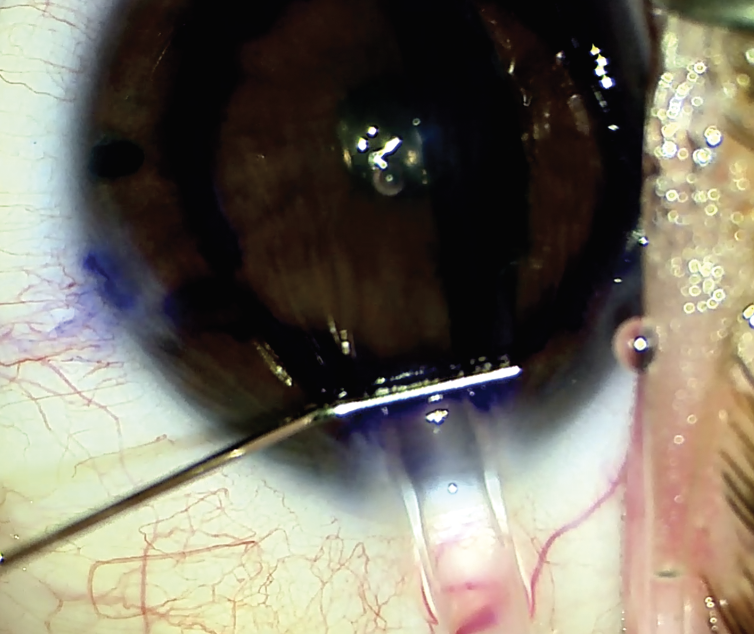
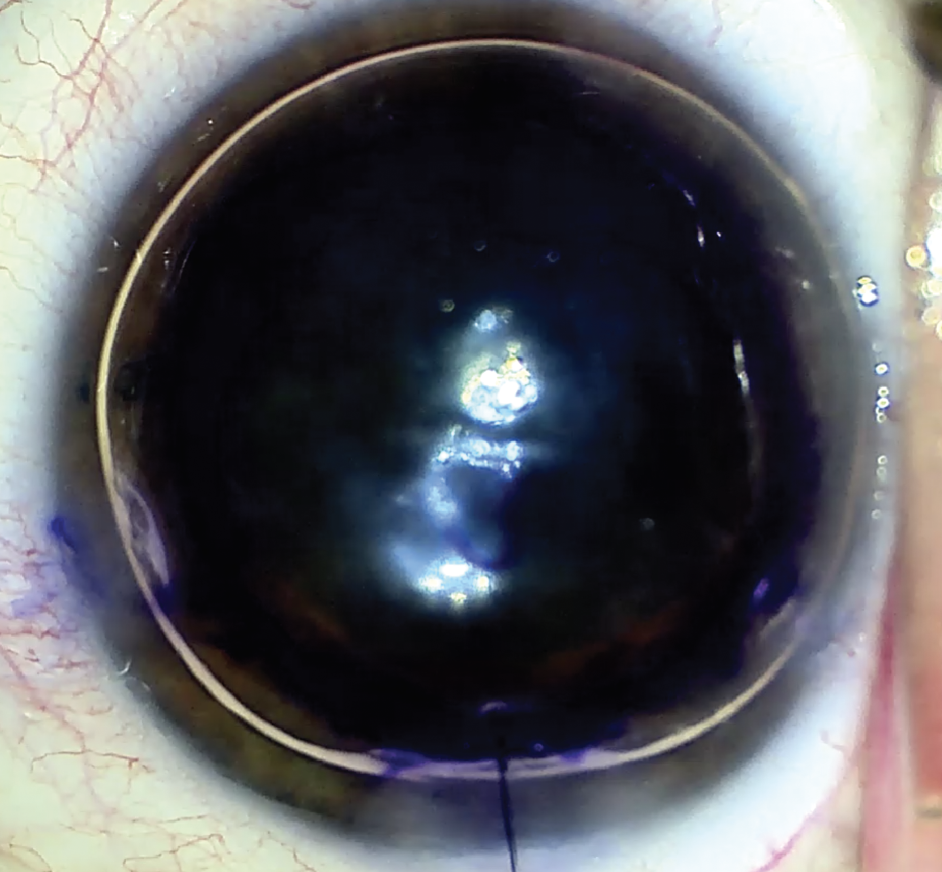 |
| Figure 2. “S” stamp with SF6 gas in DMEK. (Courtesy Nandini Venkateswaran, MD) |
Typically, the endothelium of the graft is rolled out, which is the natural roll. There’s a new trend of forcing the endothelium to face inward by locking the scroll in an inverted orientation inside a narrow injector tube. “We call it endo-in, and the advantage is that you can actually pull it into the eye with an instrument and the graft will be in the right orientation in the anterior chamber,” Dr. Chamberlain says. “Experienced DMEK surgeons are probably not as concerned about this orientation issue on routine DMEK surgeries, but there are some theoretical advantages for more complicated eyes, such as those with a damaged iris, aphakia or post-vitrectomy. Because this method puts the graft in the proper orientation, it can be positioned against the posterior cornea while being held in place by the second instrument to keep the surgeon from losing the graft onto the retina in aphakic eyes or pressing it against an intraocular lens in eyes with large or damaged pupils, or where the chamber can’t be shallowed. Several eye banks are starting to provide surgeons with an option to get tissue prepared in that orientation.”
Dr. Chan says that she marks the tissue to ensure the correct orientation, and she pre-places a 10-0 nylon interrupted suture in the main incision. “I try to get the graft into a double-scroll configuration prior to injecting into the eye because then it is much easier to unfold and to orient the endothelium side down,” she says. “The anterior chamber must be shallowed and have a low pressure during the injection or the DMEK tissue risks being ejected out of the wound. Once the DMEK graft is in the anterior chamber, it has to remain shallow to pin the graft in place. I tie the suture using a slip knot, then I use a couple 27-ga cannulas attached to 3-cc syringes to gently tap open the graft and massage it into position. I do a 10-minute pressurized full fill using approximately 14 percent SF6 gas (0.7 cc SF6 in a 5-cc syringe) and then lock the slip knot while the anterior chamber is in a physiologic shape. Lastly, I reduce the bubble size to about 60 percent, making sure that it covers the diameter of the DMEK graft, by injecting a phenylephrine cocktail similar to that used in floppy iris cataract cases to ensure that the pupil dilates as well. In eyes with tubes or trabs, I will leave in a full gas fill with the IOP around 25. Topical cyclogel is also used followed by a subconjunctival injection of ancef and dexamethasone.”
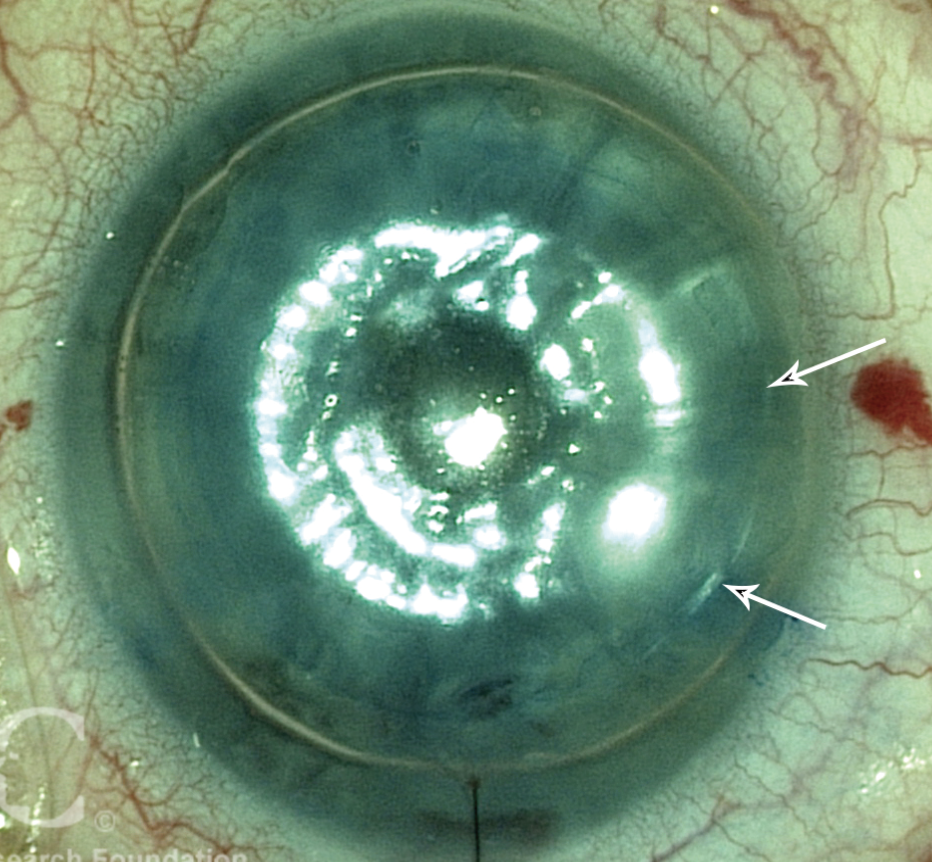 |
| Figure 3. The graft is unfolded and in place; air placed in the anterior chamber helps to hold it there. (Arrows mark the edge of the graft.) (Courtesy Francis W. Price Jr., MD) |
Tips and Techniques for DSAEK
According to Dr. Chamberlain, DSAEK accounts for approximately one-third of the transplants being performed in the United States. “It is still largely performed by surgeons using a pre-cut graft,” he says. “It’s cut at the eye bank and comes intact on a corneoscleral rim. The surgeons trephinate the tissue to the desired size and put it into the eye. There are various ways to do that. Some surgeons slide it into the eye in the correct orientation on a Sheets glide. Many surgeons use an injector or pull-through method, and there are several available. Some eye banks are now taking these injectors and pre-loading them with DSAEK grafts. The surgeon receives the graft, and he or she just has to pull it in the eye with a microforceps or push it in with fluid. That saves the surgeon some steps in the OR and cuts down on OR time. I personally have been a little concerned about storing DSAEK grafts and those injectors because they’re much thicker than DMEK grafts, and there may be some crowding, with a reduction in access and diffusion of storage media over the storage time, but several eye banks have validated this storage technique and vouch for its safety.”
Dr. Chan adds that there are many instruments and inserter systems available for both DMEK and DSAEK, and she recommends practicing using these instruments in a wet lab prior to performing live surgery. “Some cornea surgeons prefer a pull-through technique for DSAEK, while others prefer an injector or push-in technique,” she says.
Dr. Venkateswaran primarily uses the EndoSerter from CorneaGen. “The EndoSerter gets attached to the irrigation/aspiration unit on your phaco machine, and it’s nice because it has ongoing irrigation as you’re inserting the graft into the anterior chamber,” she says. “This allows you to maintain the anterior chamber depth and have a smooth and atraumatic insertion of the graft as compared with other techniques like folding forceps or direct pushing of the graft into the eye on a Sheets glide with a small needle. Some ophthalmologists use the Tan EndoGlide from Innovia Medical, which is also a great tool. I like the fact that the EndoSerter allows for an ongoing infusion with it, so I don’t need to place in an anterior chamber maintainer when I do these cases.”
Dr. Chamberlain adds that some surgeons are using support mechanisms, such as an anchor stitch through the graft, for complicated DSAEKs. “Surgeons can run a stitch through the graft to pull it into the eye and/or anchor it to the back of the cornea,” he explains. “The most common method is probably a single suture superiorly or nasally. It may damage the endothelium at one discrete point, but if it’s done carefully, it’s very effective at reducing postoperative manipulation in the event of a detachment. It doesn’t keep the graft from detaching, but it keeps it from dislocating. This is a useful technique for complicated eyes where the bubble might escape to the back of the eye or go up into a glaucoma shunt or trabeculectomy bleb the night after surgery. It can be easily rebubbled at the slit lamp the next day or at the 1-week visit in clinic if the graft has partially detached from the posterior cornea, similar to a DMEK rebubbling step.”
Dr. Chan says that, for DSAEK, she uses an anterior chamber maintainer hooked up to the irrigation on a phaco machine with IOP set to 40 mmHg with a pull-through technique using the Tan curved DSAEK forceps and a reusable inserter device, such as the Macaluso or Busin glide. For DMEK, she uses the Geuder glass cannula attached to a 3-cc syringe filled with balanced salt solution to inject the tissue. “DSAEK tissue, no matter how thin, will typically open up in the correct orientation,” she says. “I like to pre-place a 10-0 nylon suture in the main incision. Before the forceps lets go of the tissue, I inject a small air bubble underneath the graft to keep it floating against the cornea. Then, I tie the slip knot and bump the graft into position using the 19-gauge cannula, which is usually attached to a large BSS bottle, then pressurize the eye to an IOP of about 40 with a full air fill in the anterior chamber for 10 minutes. I finish tying off the suture at the main incision now that the anterior chamber is physiologic in shape before reducing the air bubble to about 60 percent, ensuring that it covers the diameter of the graft. In complex eyes at no risk for pupil block, I will release some air to bring the IOP down to about 25 mmHg, but will keep the anterior chamber completely filled with air.”
She adds that patients are kept supine in the recovery area for one hour after DSAEK and for two hours after DMEK. Instructions are given to maintain supine positioning as much as possible or on the opposite side to the surgical eye so that the bubble covers the temporal incision (a frequent location for graft detachments especially in DMEK if the graft overlaps the irregular internal main wound). “Patients’ vision and IOP are then checked in the clinic and a drop of cyclogel and antibiotics is instilled, before being repatched and shielded and checked again the next morning,” she says. “Having a consistent technique and being detail-oriented with each surgical step helps to ensure consistent successful results with a low rebubble rate.”
The Future
Dr. Venkateswaran says that advances to these corneal procedures will continue. “DSAEK tissue didn’t come pre-loaded until recently. Now, we’re seeing multiple eye banks and insertion devices having pre-loaded DSAEK tissue, just like we have pre-loaded DMEK tissue. This simplifies and shortens your surgical and procedural time and increases efficiency, and I think many surgeons are transitioning over to these techniques. I do think that corneal surgery will continue to play a role, but we’re seeing the advent of injectable endothelial cell therapy, which may supplant surgical options in the future,” she says.
Dr. Venkateswaran has a financial interest in CorneaGen. Drs. Chamberlain and Chan have no financial interest in any of the products or companies mentioned in this article.