All current commercial OCT systems (and some research systems) use what is called "time-domain" technology. Although this technology has certainly proven to be clinically valuable, it has limitations—most notably limited resolution and slow acquisition time.
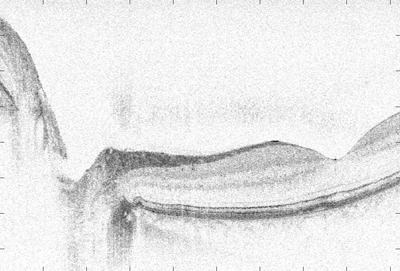 |
| A spectral-domain OCT image of an optic nerve and fovea. Because of its speed, SDOCT can be used for mapping optic nerve head topography, 3-D imaging and in vivo video imaging. Johannes F. de Boer, PhD |
Recently, by changing two key components of the older technology, researchers at the Wellman Center for Photomedicine of the Massachusetts General Hospital in Boston and Harvard Medical School have created a new variation on OCT that has significantly improved both the speed of data acquisition and the resolution of the results. This new version of the technology, referred to as spectral-domain OCT, or SDOCT, promises to make OCT scanning a more powerful tool than ever. (Another name for this technology, Fourier domain OCT, refers to its use of the Fourier transformation to convert spectrum data, provided by the spectrometer, into depth data.)
Eliminating Obstacles
Teresa C. Chen, MD, glaucoma specialist at the Massachusetts Eye and Ear Infirmary and assistant professor of ophthalmology at Harvard Medical School, is participating in the testing of the new version of OCT, which was developed by Johannes F. de Boer, PhD, associate professor at Harvard Medical School.
"Time-domain OCT is fundamentally limited by slow acquisition speed and low resolution," explains Dr. Chen. "In time-domain OCT technology, a mirror has to be moved back and forth during scans and a photodetector is used to analyze the data by measuring different depth positions serially. In contrast, the SDOCT instrument has no need for a moving mirror, and it analyzes the data using a spectrometer that can measure different depth positions simultaneously. These two changes account for the dramatic increase in acquisition speed, as well as the improved resolution."
Notable among previous attempts to improve OCT technology is "ultra-high-resolution OCT," which uses a titanium-sapphire laser as a light source. "This technology improves the resolution, but is still limited by slow acquisition speed," notes Dr. Chen, "and its titanium sapphire laser source is much more expensive and more difficult to maintain. SDOCT can use the same light source used in commercially available clinical OCT instruments."
Multiple Advantages
This new approach to OCT offers several clear benefits:
• Increased data acquisition speed and resolution. Single images ("B-scans") consisting of 1,000 A-scans can be acquired in 34.1 ms, allowing for an axial resolution of 6 µm. Using a different light source, single images consisting of 500 A-scans can be acquired in 34 ms, allowing for an axial resolution of 3.5 µm.
This technology produces data several orders of magnitude more sensitive than that produced by time-domain OCT; one study found that its sensitivity was 150 times greater. This level of resolution may allow clinicians to detect minute changes in the nerve fiber layer, possibly leading to earlier detection of glaucomatous changes.
• Mapping of true optic nerve head topography. "The old OCT technology doesn't create a true map of optic nerve topography, because the data acquisition is so slow," explains Dr. Chen. "With the current Stratus OCT, it can take 1.28 seconds to acquire a 2-D image. During that time, the eye makes a lot of imperceptible movements, even with a very cooperative patient. As a result, you have to re-align the A-lines, similar to A-scans, in order to create an image.
"SDOCT can acquire the same data in milliseconds," she notes. "Therefore, no re-aligning of the A-lines is needed, and we can create a true image of the optic nerve head."
• 3-D imaging. Rapid data acquisition makes it possible to produce 3-D images of ocular anatomy with less interference from patient movement. A 6.2 x 6.2 mm area can be scanned with 62 parallel images 100 µm apart in 2.1 seconds. This could be used to produce a 3-D map of the entire posterior pole for detection of glaucoma, detect the exact location of diabetic macular edema, determine the size and depth of choroidal neovascularization, or measure the size of choroidal lesions. Because of these capabilities, as well as the technology of spectral domain optical Doppler tomography, spectral domain OCT could decrease the need for fluorescein angiography.
• In vivo video imaging. High-speed data acquisition makes it possible to do in vivo video imaging at 29 frames per second (1,000 A-lines per frame) with an axial resolution of 6 µm. (A different light source allows a resolution of 3.5 µm.) Among other things, this can be used to measure retinal blood flow, including real-time imaging of blood vessels as small as 10 µm and as deep as the choroid. (Creating videos or movies of 3-D anatomy is currently not possible with the Stratus OCT.)
Asked whether SDOCT has any limitations, Dr. Chen says that it may still be possible to achieve better resolution or higher speed. "But there's been no trade-off for the improved speed and resolution spectral domain OCT has already achieved," she says. "Dr. de Boer hasn't made the technology more unsafe—just the opposite. Also, we can image larger areas of the eye more quickly. So it improves what we're able to do in almost every respect."
Benefits in Practice
Dr. Chen notes that the increased capabilities of this technology could offer significant benefits for clinical practices. "Having OCT technology with better resolution and higher speed should improve our ability to diagnose and monitor a host of problems," she says. "Being able to more accurately scan the optic nerve and nerve fiber layer may allow us to detect glaucoma earlier and monitor patients more effectively. Being able to image retinal structures affected by macular degeneration more precisely may help with diagnosis and decrease the need for more invasive testing, such as fluorescein angiograms. Because this technology can image larger areas, we should be able to monitor posterior segment tumors better. OCT technology can be used for many other retinal diseases as well."
Dr. Chen says the main focus right now is making SDOCT as practical as possible for clinical use. "For several years Dr. de Boer has been improving the SDOCT instrument that he built here at the Massachusetts General Hospital, making it more patient- and doctor-friendly," she says. "For example, we're finding ways to improve image acquisition despite patient eye movement or attention loss."



