Tonometers
IOP Measurement Comes Home
Icare USA announced FDA 510(k) clearance for its next-generation self-tonometer, the iCare Home2. The company says the tonometer is designed for additional ease of use in measuring patients’ real-world intraocular pressure outside of normal clinic hours. A smart-light guide and interactive display screen means most patients can utilize the iCare Home2 on their own, the device’s maker adds. iCare Home2’s new design enables IOP measurements to be taken while the patient is supine, reclined or sitting, iCare says. For more information, visit icare-world.com/us/product/icare-home2.
Dry-Eye Therapy
 |
A New Option for MGD Sufferers
Physicians now have an additional tool in their in-office dry-eye armamentarium. Alcon’s Systane iLux2 is a meibomian gland dysfunction thermal pulsation system that uses new imaging technology to capture infrared photos and high-definition videos of the meibomian glands. The company says physicians can use the iLux2 to tailor MGD treatment to individual patients in eight- to 12-minute sessions and customize heat and compression along the treatment zone. Additionally, iLux2 stores meibomian gland images for disease tracking and comparison over time. The company points out that by enabling patients to see their MGD for themselves in high-definition video, iLux2 may bolster patient relationships and treatment credibility. For information, visit SystaneiLux.com.
Lens-based Surgery
 |
The EVO Visian ICL Debuts
In March, the Staar EVO Visian ICL was granted FDA approval for the correction of myopia and myopia with astigmatism. Staar says the EVO Visian ICL offers a lens-based alternative for the correction/reduction of refractive error in patients who currently use glasses or contact lenses for distance vision correction.
The EVO Visian is implanted in the posterior chamber directly behind the iris and in front of the natural crystalline lens. Due to a new design feature, no peripheral iridotomy is necessary, as was the case with previous lens iterations. The implantable collamer lens has already been in use outside the United States with good outcomes, the company says. One advantage Staar points out is that the EVO lens doesn’t cause dry eye.
The EVO Visian ICL is available for the correction or reduction of myopic astigmatism in patients with spherical equivalents ranging from -3 to -20 D, with astigmatism from 1 D to 4 D at the spectacle plane; with an anterior chamber depth of at least 3 mm when measured from the corneal endothelium to the anterior surface of the crystalline lens; and a stable refractive history (defined as not varying more than 0.5 D for one year prior to implantation). Staar adds that it will soon be training and certifying surgeons for EVO use. For more information, visit discovericl.com.
New Clareon Line of IOLs Available
Alcon recently launched its Clareon family of IOLs. The company says that Clareon uses the company’s newest, most advanced IOL material, to “deliver consistent visual outcomes and exceptional clarity.” Alcon says this clarity is born from a glistening-free IOL material that has very low levels of haze and subsurface nanoglistenings.
Clareon Monofocal, Clareon PanOptix, Clareon PanOptix Toric, Clareon Vivity and Clareon Vivity Toric IOLs are now available in the United States. The lenses are inserted using the reusable Clareon Monarch IV Delivery System. The company says that the Clareon Monofocal is also available in the next-generation automated, single-use delivery system, AutonoMe. Read more at alcon.com/media-release/alcon-strengthens-leadership-iol-innovation-launch-clareon-portfolio-us.
Imaging & Diagnostics
 |
Push-button Images
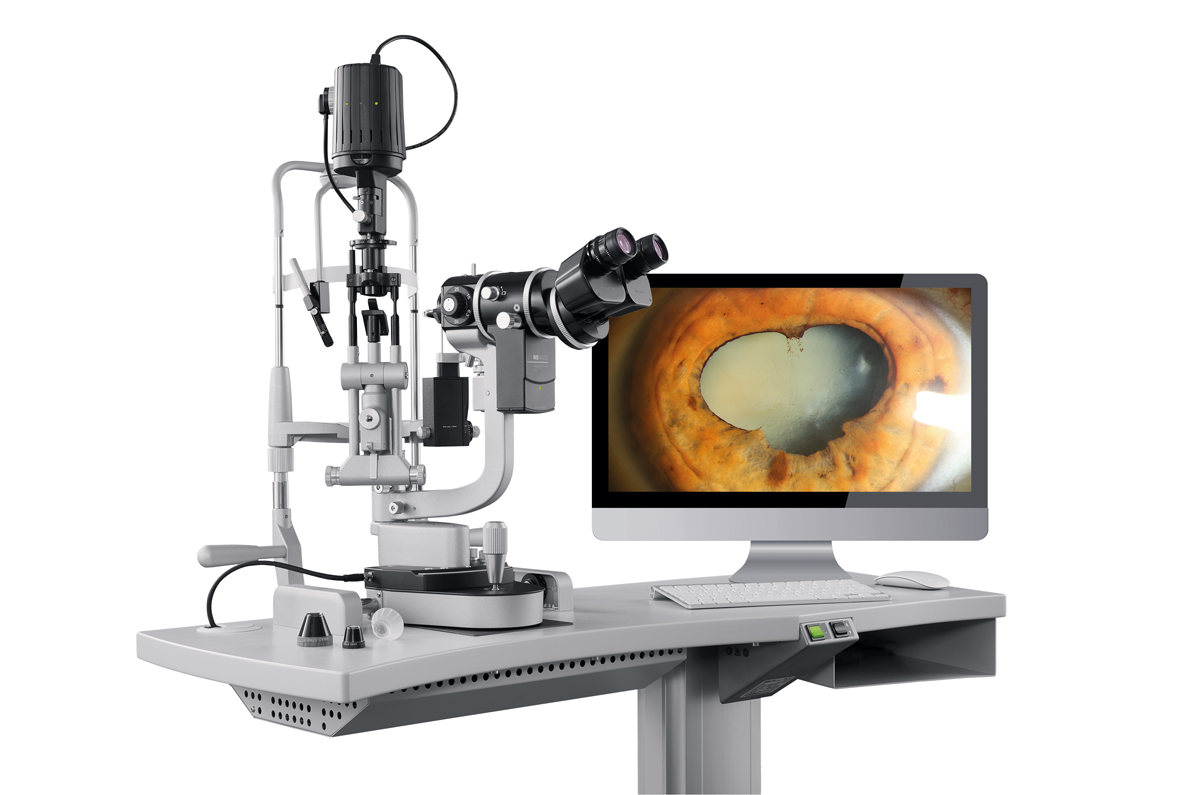
Haag-Streit says its new slit lamp Imaging Module 910 offers ophthalmologists the ability to quickly capture high-quality images. The company says the module doesn’t require any software and is ready to go by just turning a knob, so it won’t slow down exams. Images are captured by pressing the camera trigger button.
The Imaging Module 910 includes a camera sensor and smart features such as a performant auto-exposure mode and automatic aperture control to ensure good illumination in all images. Its image-selection algorithm chooses the best image in order to cut down on time. There are two modes to choose from: standalone and EyeSuite. In the standalone mode, images are stored directly in the practice’s EMR system. In the EyeSuite mode, image editing tools and features are available. For information, visit haag-streit.com/haag-streit-usa/products/haag-streit-diagnostics/digital-imaging/imaging-module-910/.
Contacts
 |
A Contact Lens that Fights Allergy
Johnson & Johnson Vision says its newest contact lens innovation offers patients both vision correction and relief from ocular allergic itch. The FDA-approved Acuvue Theravision contact lenses are the first and only medication-releasing contact lenses for preventing ocular itch due to allergic conjunctivitis, the company says. Each contact lens contains 19 mcg of the antihistamine ketotifen. Johnson & Johnson Vision says these daily disposable contact lenses are suitable for patients with 1 D or less of astigmatism.
In the Phase III clinical studies, Acuvue Theravision demonstrated a clinically and statistically significant reduction in ocular itch beginning at three minutes after insertion. The effect lasted up to 12 hours. The company notes that these lenses aren’t suitable for those with red or irritated eyes. For information, visit acuvuetheravision.com.
Surgical Training
 |
Go VR Before the OR
Alcon’s Fidelis Virtual Reality Ophthalmic Surgical Simulator is a portable VR tool for cataract surgeons-in-training. Its virtual operating room provides haptic feedback to simulate the tactile experience of cataract surgery, the company says. Alcon adds that the real-time simulator can be used from any location around the world, increasing access to surgical training in regions with limited resources. Participants can also join virtual instruction and training sessions.
The portable Fidelis includes a VR headset, two haptic engines, an integrated Centurion footswitch, a realistic OR environment with Alcon equipment, remote connectivity for multiple users and instructors within the same virtual OR, and real-time feedback and performance tracking in order to improve technique.
Alcon says the first Fidelis VR simulators will be used in the company’s phaco department program and will enter teaching and residency programs later this year. The Fidelis will also provide VR ocular anatomy and physiology education for other eye-care professionals. For information, visit alconexperienceacademy.com/index-aea.aspx.
Retinal Therapy
 |
Xipere Takes a New Approach to Treatment
Bausch + Lomb and Clearside Biomedical recently announced the U.S. commercial launch of Xipere, a triamcinolone acetonide injectable suspension that was FDA approved in October 2021 for suprachoroidal use for the treatment of macular edema associated with uveitis. The therapy is administered using Clearside’s suprachoroidal space Microinjector. In the clinical studies, the most common ocular side effects were increased intraocular pressure and eye pain, and the most common non-ocular side effect was headache.
Bausch + Lomb explains that the suprachoroidal space expands upon injection, which enables targeted drug delivery to the posterior structures. The company also points out that since the suprachoroidal administration technique differs from traditional intraocular administration, it’s currently offering Xipere training. For information, visit xipere.com. For training, visit xipere.com/hcp/xipere-training.
Vision Therapy
Don’t Fear the Light
Do you have light-sensitive patients suffering from migraine or other conditions? Eschenbach says its new Acunis FL-41 filters are designed for patients with light sensitivity due to migraines, traumatic brain injury and other eye conditions.
 |
The company explains that Acunis FL-41 lenses provide more visual comfort for these patients by reducing the transmission of certain light wavelengths that are responsible for glare, unlike traditional sunglasses that attenuate all light wavelengths evenly.
The company has three lens tints available: light, medium and dark, which feature 25-, 50-, and 75-percent light absorption, respectively. There are four frames of varying styles to choose from, made from either stainless steel or acetate. For information, visit eschenbach.com/products/fl-41-filters.asp.



