GLAUCOMA THERAPY
Thea Launches Iyuzeh
Thea has launched preservative-free Iyuzeh in the United States. The company says the drug is the first and only preservative-free latanoprost option for patients with primary open-angle glaucoma and ocular hypertension in the nation. According to the company, this will help avoid patients experiencing the moderate to severe signs and symptoms of ocular surface disease associated preservatives.
The company says that, in multiple trials across the United States and Europe, Iyuzeh demonstrated consistent IOP-lowering effects and tolerability. It lowered IOP by 3 to 8 mmHg in patients with OAG or OHT with a mean baseline IOP of 19 to 24 mmHg, compared to the BAK-preserved Xalatan’s 4 to 8 mmHg.
The recommended dose of Iyuzeh is one drop in the affected eye(s) once daily in the evening. The company says reduction of the IOP begins three to four hours after administration, with the maximum effect achieved after eight to 12 hours, lasting at least 24 hours. The most frequently reported ocular adverse events in the clinical trials included conjunctival hyperemia and eye irritation.
For more information about Iyuzeh, visit theapharmainc.com.
Generic Alphagan-P Now Available
Apotex has released its latest ophthalmic product in the United States: brimonidine tartrate ophthalmic solution, 0.1%.
Brimonidine is an alpha-adrenergic receptor agonist indicated for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
For information, visit www.apotex.com/products/us/downloads/pre/brim_opso_0.1_ins.pdf.
CORNEAL SURGERY
 |
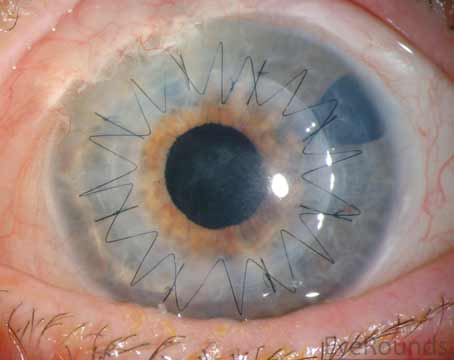
A Corneal Cover-Up
NovaBay Pharmaceuticals recently announced the launch of Avenova Allograft in the United States. The company says this prescription product is the only optic allograft manufactured using BioStem Technologies proprietary process and is intended for use as a protective covering during the repair of ocular surfaces. BioStem will be responsible for manufacturing, packaging, shipping and regulatory compliance.
The Avenova Allograft provides a protective environment or covering for repair of the cornea and conjunctiva, helping the ocular surface to return to a healthier state, the company says. BioStem says the graft uses what the company calls its patented six-step BioRetain process that preserves the natural integrity of the placental tissue. The product consists only of the amnion layer of the placental membrane and measures between 20 to 50 µm thick, which the company says makes it ideal for delicate ophthalmic applications. Avenova Allograft is available in 8 mm, 10 mm and 12 mm diameter sizes.
NovaBay’s commercial launch of Avenova Allograft includes outreach programs aimed at educating eye-care specialists on both the clinical benefits of the product, as well as the process for Medicare reimbursement. Clinics can order the product directly on Avenova.com.
EXAM ASSISTANCE
Bring That Pupil Down
Ocuphire Pharma and Viatris recently annouced the FDA approval of their mydriasis-reversal drug Ryzumvi (phentolamine ophthalmic solution, 0.75%). The drug is approved for the treatment of pharmacologically-induced mydriasis produced by adrenergic agonists (e.g., phenylephrine) or parasympatholytic (e.g., tropicamide) agents, which are commonly used during eye exams for dilation.
Ryzumvi was evaluated in the comprehensive MIRA clinical trial program involving more than 600 subjects. In the MIRA-2 and MIRA-3 Phase III trials, a total of 553 subjects aged 12 to 80 years, who had mydriasis induced by instillation of phenylephrine or tropicamide or a combination of hydroxyamphetamine hydrobromide and tropicamide (Paremyd) were randomized. Two drops (study eye) or one drop (fellow eye) of Ryzumvi or placebo (vehicle) were administered one hour after instillation of the mydriatic agent. The percentage of subjects with study eyes returning to ≤0.2 mm from baseline pupil diameter was statistically significantly greater (p<0.01) at all time points measured from 60 minutes through 24 hours in the Ryzumvi group compared with the placebo (vehicle) group across both of the MIRA-2 and MIRA-3 trials, says the company. The efficacy of Ryzumvi was similar for all age ranges including pediatric subjects aged 3 to 17 years. Pediatric subjects aged 12 to 17 years (n=27) were treated in MIRA-2 and MIRA-3 and pediatric subjects, aged 3 to 11 years (n=11) were treated in MIRA-4.
The most common ocular adverse reactions reported in >5 percent of subjects were instillation site discomfort including pain, stinging and burning (16 percent) and conjunctival hyperemia (12 percent). The product is expected to launch in the first half of 2024, the company says.
For more information, visit ryzumvi.com.