Cataract surgery is a valuable adjunctive treatment for glaucoma. Alone, it’s been shown to reduce intraocular pressures in patients with glaucoma and ocular hypertension, and it opens up the possibility of combining cataract removal with a minimally invasive glaucoma surgical procedure in mild to moderate cases. However, selecting a lens implant and performing cataract surgery in glaucoma patients may be less straightforward than in those with healthy eyes.
Here, cataract surgeons and glaucoma specialists discuss considerations for these patients undergoing cataract extraction, share surgical pearls and tips for choosing lens implants and offer advice on postoperative steroid management.
Less (Disease) is More
In general, the less glaucoma a patient has, the better. “You’ll get a better outcome in milder cases because you’re not competing with other levels of impairment,” says Constance O. Okeke, MD, MSCE, of Virginia Eye Consultants in Norfolk. “So, patients who have ocular hypertension, glaucoma suspects or those with very mild, pre-perimetric glaucoma are going to be great candidates if they’re also well-controlled and if their visual fields have very minimal peripheral defects. These are the patients you should consider pushing the envelope to offer them multifocals or extended-depth-of-focus IOLs.”
“Poor candidates may include patients with advanced disease, any kind of central visual field defect, poorly controlled disease or at high risk for progression,” she says. “These patients may need a trabeculectomy or tube shunt and won’t do well with advanced-technology lenses because of the astigmatism these procedures can create.”
As with any cataract or refractive procedure, addressing ocular surface issues is important for achieving good measurements and good outcomes. However, if the patient is on a heavy medication load, it may be difficult to get their ocular surface under control, Dr. Okeke points out. “We have to do what we can to improve patients’ ocular surfaces, whether that’s by using artificial tears, medications such as Restasis or Xiidra, removing excess lid debris or treating the meibomian glands.
“I use a number of different treatments for meibomian gland rehabilitation such as iLux, LipiFlow and BlephEx,” she says. “I offer these in addition to recommending vitamins and warm compresses for long-term care. It’s important to address all aspects of ocular surface disease prior to surgery and then also let the patient know they may be dealing with some of these issues after the surgery as well.”
Set Expectations
For those who are candidates, Dr. Okeke says that setting expectations before surgery in a way that’s understandable by the patient is vital. “I always have a visual field that’s at least three months old or from that day to show the patient where they are with their glaucoma,” she says. “I explain to them in pictorial form what a normal visual field looks like, and then I show them what their visual field looks like. I explain to them where their blind spot is naturally and where the blind spots are from glaucoma.
“Then, I put my hand over the visual field and say, ‘A cataract is like a film on top of this. When I move the cataract away, what’s left are the areas that can be enhanced. So, a light gray area on the visual field is where you can expect to have enhanced vision. Any dark gray to black areas are permanently damaged spots from glaucoma.’ Patients need to have an understanding of where their glaucoma is so they can understand that after the surgery, when the cataract is gone, what’s left is their glaucoma,” she says. “Now, that’s the set expectation as opposed to having them wonder why they’re not seeing better and thinking the surgery was supposed to cure their glaucoma.”
For patients with moderate to advanced glaucoma, Dr. Okeke says a frank discussion about visual fields is also needed. “If they have central fixation loss, then it’s really important to let the patient know that you feel premium lenses may not be as beneficial to them and may actually make their vision worse, in terms of contrast sensitivity and vision quality. For patients who aren’t candidates for extended-depth-of-focus or multifocal lenses, adding a presbyopia drop as a secondary solution to a monofocal IOL set for distance may offer some advantage.”
 |
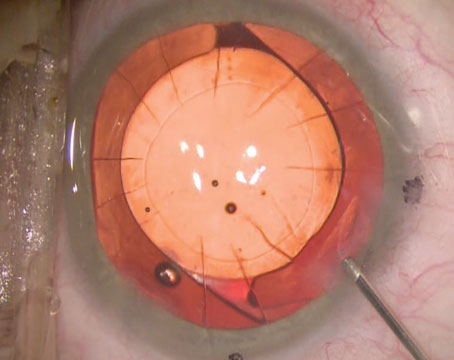
| Figure 1 A-D. (A) Patient is a candidate for a multifocal, an EDOF or a toric lens. (B) Patient is a candidate for an EDOF or toric lens. (C) Patient is a toric candidate. (D) Defer from advanced-technology IOLs. Photo: Constance O. Okeke, MD. |
Which Lenses Can You Offer?
When trying to choose a lens implant for a glaucoma patient, experts say they consider the patient’s age, the severity of their disease, the number of medications they’re on and the state of their ocular surface (Figure 1). “If I saw two patients with mild glaucoma—one younger and one older,” says Joshua Frenkel, MD, of Evergreen Eye Center in Auburn, Washington, “I may be more cautious in the younger patient, who has greater potential for future vision loss, than the older patient who’s farther along in age and perhaps less likely to progress.”
If patients want less dependence on glasses, there are trade-offs, Dr. Okeke points out. “Patients who have glaucoma already have reduced contrast sensitivity and may be sensitive to glare, so certain premium options may be off the table,” she says.
Monofocals are the safest choice for patients with preexisting ocular pathology since they don’t split light, but not all glaucoma patients are limited to the standard lens. Dr. Frenkel says, “Though multifocals can be used in patients with mild glaucoma, I tend to be a bit on the conservative side, in the sense that I’d probably recommend an extended-depth-of-focus lens, such as Vivity or Symfony, if a mild glaucoma patient desired a little more range of vision.
“For a moderate to severe patient, you’re certainly going to lean toward recommending a monofocal,” he continues. “Once the patient has a certain amount of loss of contrast sensitivity, they’ll be more likely to notice any drop-off in vision with a premium lens.”
He says Eyhance or the Light Adjustable Lens are appropriate options for glaucoma patients. “The LAL is a monofocal, but you can induce some extended depth of focus, though I wouldn’t induce a large amount in an advanced glaucoma patient,” he adds. “The LAL also does well with blended vision, so you can give patients something similar to the premium result they’re looking for with good accuracy for their glaucoma.”
Dr. Okeke cautions that monovision may not be a good option in certain glaucoma patients. “I tend to dissuade patients from monovision who have moderate to advanced glaucoma in one eye and very mild glaucoma in the other eye because they’ll be putting all the ‘weight’ of vision on one eye,” she says. “If you have significant glaucoma in one eye, it’s likely that there will be greater risk for progression in that eye, so at some point, they won’t be able to use that eye well for monovision function and they’ll be back in glasses.”
Toric lenses are another strong option for glaucoma patients who wish to have their astigmatism corrected. Since these IOLs don’t split light, they enable a glaucoma patient to see a specific focal point with their best vision, Dr. Okeke explains. “There are a number of premium toric options including the Tecnis toric, Eyhance, AcrySof IQ toric and EnVista toric,” she says. “Eyhance is a newer lens that has a continuous change in power from the outside in, which allows for both strong distance and enhanced intermediate vision.”
One thing to consider with toric lenses is that glaucomatous eyes often have small pupils. “You may need to use pupil expanders in these eyes,” Dr. Okeke says. “Afterwards, when removing the pupil expander, the pupil can come down, and this can sometimes limit your view of the toric markings, so you have to be able to manipulate the eye to ensure the lens is appropriately aligned.
“Also, be cognizant of zonular weakness,” she adds. “You may have a patient with pseudoexfoliation who shows no signs of zonular dehiscence at the time of surgery, but it’s still a good idea to prevent that from happening and use a capsular tension ring for extra stabilization.
“I don’t combine toric IOLs with trabeculectomies or tube shunts because of the inconsistency of the outcomes and the potential development of astigmatism afterwards,” Dr. Okeke notes. “I’d give the patient at least six months or more to have a stable refraction and then do the cataract surgery, with the expectation of a good outcome if they have a good field of central vision.”
Douglas K. Grayson, MD, of Omni Eye Services in the New York Metro Area, points out that postoperative glaucoma bleb patients will benefit from a toric lens if their bleb-induced astigmatic error is stable. “These patients have to be followed for a while,” he says. “If the bleb is overgrowing onto the cornea, you may one day have to revise it by physically minimizing its intrusion. Then, whether you want to use a toric lens or not depends on how true the toricity induced in the cornea is.”
He says multifocal and extended-depth-of-focus IOLs can be used successfully in glaucoma patients, with certain caveats. “We’ve been successfully using multifocal lenses in glaucoma patients—mild, moderate and borderline-severe—depending upon where the defect is,” he explains. “Now, if a patient has advanced field loss with a central island, they’ve had so much loss of contrast sensitivity already from their glaucoma that using a multifocal lens wouldn’t be a good idea. In patients with superior arcuate defects and clear inferior hemifields, however, we’ve had success using Symfony and PanOptix.
“The hedge for most of my team is to use something like Symfony rather than PanOptix because the EDOF lens tends to be a little more forgiving of power fluctuations,” he continues. “Whether you’re off by half a diopter plus or minus, patients still seem to do very well, as opposed to the trifocal where precision calculation is particularly important.”
He adds that implanting a PanOptix lens in a patient with a filtering bleb and a low pressure can be problematic. “A patient with a filtering bleb and a pressure of 6 mmHg will have their axial length affected,” he says. “If they have cataract surgery and their bleb tends to fail, the pressure could go back up to 25 mmHg and that would change the size and contour of their eye and alter the refraction. We’ve been successful using multifocals in patients who’ve had filtering operations, so we don’t think that’s a contraindication, but in more severe cases, it’s an individual discussion and judgment call with the patient.”
Multifocal lenses can lead to OCT artifacts, and the reduced contrast sensitivity can decrease one’s ability to monitor visual fields, Dr. Okeke notes. “It’s important to keep this in mind,” she says. “However, it seems that patients who have mild, pre-perimetric glaucoma and glaucoma suspects can do well with multifocals. If reduced visual field encroaches anywhere close to fixation, multifocals aren’t ideal.”
She says that Vivity is a good option for certain glaucoma patients. “The lack of diffractive rings means there’s less visual disturbance and fewer halos,” she explains. “The fact that Vivity doesn’t split light helps with the issue of contrast sensitivity. It’s enabled more glaucoma patients to be premium IOL candidates.”
Dr. Grayson says that some normal-tension glaucoma patients can also receive premium lenses. “Normal-tension glaucoma’s effects tend to be near fixation and these patients tend to be younger and need lower pressures,” he says. “In terms of doing the cataract surgery, they’re still going to have a MIGS. Depending upon how severe their visual field loss is, I’d still do a multifocal lens if they had even some damage around fixation, as long as it wasn’t too extensive.”
Dr. Frenkel says that depending on the level of visual field changes, he’d treat normal-tension glaucoma patients similarly to those with primary open-angle glaucoma. “Anecdotally, (n= a few), I’ve noticed that some normal-tension glaucoma patients who have cecocentral scotoma tend to have more significant glare,” he notes. “So, for some of them, I’d consider a lens that has a very low amount of glare such as the LI60AO from Bausch + Lomb, optic-captured in the sulcus.
“I once had a normal-tension glaucoma patient who complained of some glare before,” he continues. “We thought it was due to cataract, but after we took his cataract out, his glare intensified. He had a monofocal and a great result, but we wound up exchanging it for a three-piece lens that we put in the sulcus with optic capture to reduce glare, which did help some. So, it seems some patients with visual field deficits close to fixation may be more sensitive to glare.”
 |
|
Figure 2. A Hydrus and a first-generation iStent in the angle. Dr. Frenkel demonstrates a Hydrus implantation in the video below. |
MIGS and Cataract Surgery
Experts say it’s more or less become standard practice to perform a MIGS procedure in someone who’s on multiple glaucoma medications and is undergoing cataract surgery, in order to try to restore vision, minimize medication burden and control intraocular pressures.
“With a few exceptions, I’d consider doing a combined procedure in almost any patient who has glaucoma with an angle that can be treated and a cataract that’s significant,” Dr. Frenkel says. “You can combine many of the MIGS procedures with cataract surgery, including goniotomy procedures such as Trabectome and the Kahook Dual Blade; gonioscopy-assisted transluminal trabeculectomy; stents such as iStent, iStent Infinite and Hydrus (Figure 2); canaloplasty such as ABiC with iTrack or Visco360; subconjunctival such as Xen; and the combined Omni.”
(To view a video of Dr. Okeke performing femtosecond laser combined cataract and MIGS surgery, go to https://youtu.be/evvQ6ZrJtVo.)
Dr. Grayson’s MIGS procedure of choice is goniotomy using a Kahook Dual Blade (Figure 3). “The procedure doesn’t involve implanting any material that can be obscured by peripheral anterior synechiae, which is especially useful in the chronic angle closure patients who will always tend to form PAS at a higher rate,” he says. “I find that removing the anterior wall of Schlemm’s canal is a safe, effective and minimally traumatic approach. Stents, on the other hand, may be difficult to implant in a chronic angle-closure patient due to synechiae, and canaloplasty induces more trauma to the trabecular meshwork which may result in a higher rate of hyphema and regression to higher pressures.”
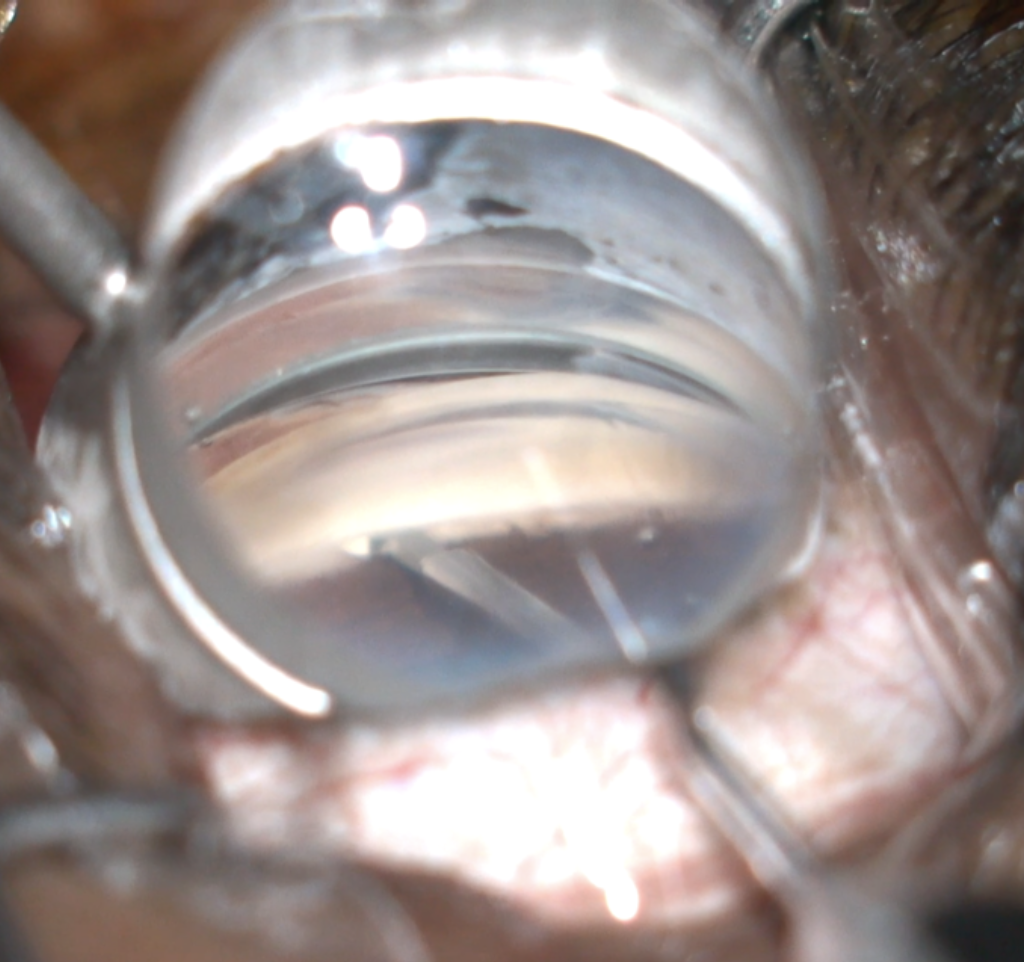 |
Figure 3A. Douglas K. Grayson, MD, performs the goniotomy after capsulotomy and before phacoemulsification. He says, “Visibility is best with a clear cornea. If there’s any hyphema caused by the goniotomy, it won’t obscure the creation of the capsulorhexis. The hyphema usually resolves by the end of the case.” Here, the goniotomy blade is used to unroof the anterior wall of the trabecular meshwork with minimal trauma to the canal of Schlemm. A single forehand pass with the KDB Glide engaging the trabecular meshwork, maintaining mild anterior traction and continuing for three to four clock hours results in a clean excisional goniotomy. Photo: Douglas Grayson, MD. |
Dr. Grayson says he considers Xen procedures somewhere between trabeculectomy and MIGS procedures. “Any of the Xen procedures are done in combination with mitomycin,” he explains. “If you don’t get mitomycin, the Xen is destined to fail. And once you start doing cataract surgery and giving mitomycin subconjunctivally, you’re really entering the realm of true glaucoma-specialist surgery and that’s not typically something that the comprehensive ophthalmologist wants to deal with. Mitomycin has a higher risk of inducing hypotony. That’s why I don’t really consider Xen as a MIGS. To me, the goniotomy and stent groups have a risk profile that’s roughly zero, whereas with Xen, the risk profile is somewhat higher than zero and thus becomes a true glaucoma procedure.”
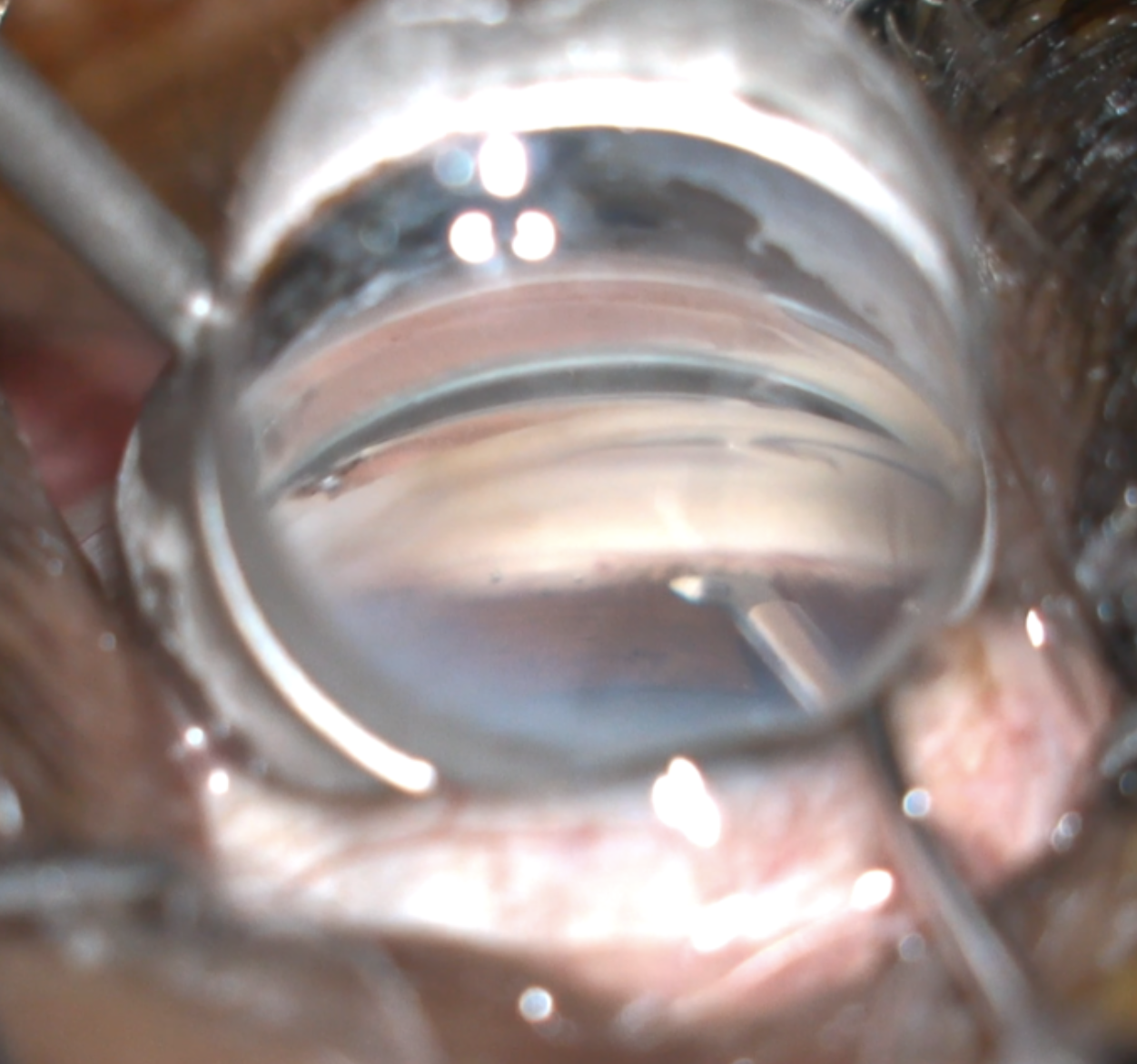 |
| Figure 3B. Continuing from Figure 3A, the blunt edge of the KDB Glide can be used for goniosynechialysis and to further open the canal of Schlemm to access anterior chamber aqueous after the anterior wall of trabecular meshwork is removed. Photo: Douglas Grayson, MD. |
When should a comprehensive ophthalmologist think about referring the case? Dr. Grayson says that if a patient has severe glaucoma with pressures that aren’t adequately controlled, “certainly the comfort level of doing trabeculectomy or Xen is up to that individual comprehensive ophthalmologist. If they perform a combined MIGS (e.g., goniotomy) and cataract surgery, then it’s important for the patient to be aware that there’s a real chance their glaucoma could still go out of control, in which case they’d be referred to a glaucoma specialist. It’s important for comprehensive ophthalmologists to have relationships with glaucoma specialists if they’re doing a lot of MIGS procedures.”
Dr. Okeke notes that if you’re considering adding a MIGS procedure, ensure your patient has at least 180 degrees of viable angle tissue to perform the MIGS and has controlled IOP. “An eye with an open angle, mild to moderate glaucoma and with one to three medications is an ideal candidate for MIGS,” she says.
What about using a femtosecond laser to perform the cataract surgery? Experts say using femto for cataract surgery in glaucoma patients is an excellent option, particularly for addressing astigmatism. “It’s good for low levels of astigmatism correction and for premium lenses, which benefit from the consistent capsulorhexis size and location a femtosecond laser can create,” Dr. Frenkel says. “Some doctors aren’t fans of FLACS in general and, theoretically, the IOP does rise for 15 to 25 seconds, but the pressure isn’t going to be much higher than it is at times during cataract surgery, so I don’t believe the risk is much higher. The benefits of femto in glaucoma patients are similar to those in non-glaucoma patients.”
“It’s a matter of how much phaco energy you need to break up the cataract, and there’s always less energy needed with femto, so I really have no contraindication to using femto in glaucoma patients,” Dr. Grayson says, who uses the Catalys laser. “Femto also offers a more stable capsulotomy. In patients with pseudoexfoliation and zonular compromise where there’s a question of lens stability, it’s beneficial to have a perfectly circular capsulotomy cut by femto in case you need to use capsular hooks to support the capsule during the phaco procedure. It’s a nice added benefit of femto, because very frequently, capsular hooks are needed to stabilize these patients.”
When it comes to cataract surgery, there are several potential anatomical considerations in glaucomatous eyes. “Glaucoma patients often have small pupils, as a result of certain drops they may be using such as pilocarpine, or they may have uveitis as a component of their glaucoma, creating synechiae formation,” says Dr. Okeke.
“Anterior capsule contraction and posterior capsule opacification are also potential concerns, as well as zonular instability in certain subtypes of glaucoma such as pseudoexfoliation glaucoma,” she says. “In these eyes, you’ll have to consider the lens position decades out from when the procedure was done. In a combined cataract and trabeculectomy procedure, there may be issues related to postoperative astigmatism creating a refractive error, which will need correction.”
“Cataract surgery works well in patients with pseudoexfoliation glaucoma because the infusion for doing the surgery scatters the pseudoexfoliation material obstructing the trabecular meshwork, usually resulting in a lower pressure,” notes Dr. Grayson. “We see a similar effect in pigmentary-dispersion glaucoma. However, in this subtype, patients often have elevated pressures again, whereas the narrower angle or angle-closure patients usually have longer, sustained reductions.”
In certain patients with angle-closure glaucoma, cataract extraction may be warranted even if the cataract isn’t visually significant. “The EAGLE study confirmed that some patients had a significant lens-induced secondary glaucoma mechanism from a cataract,” Dr. Grayson explains. “Cataracts grow thicker, and in someone who’s predisposed to narrow angles, the progression of a cataract can actually obstruct the angle. And so, if someone had elevated pressure, a narrow angle and a cataract, cataract surgery alone would lower the pressure.
“Our threshold to take out a cataract went much lower after the EAGLE study came out in 2016,” he continues. “A patient may or may not be visually symptomatic from cataract, but if, for example, they’re unresponsive to medications or to iridotomy and clearly have a lens-induced secondary angle-closure mechanism, you may then have an indication to do a lensectomy in someone who’s not truly visually symptomatic from the cataract.”
Importantly, he adds that the patients included in the EAGLE study had difficulty using their medications or had pressures that were uncontrolled. “Simply having narrow angles and a cataract isn’t an appropriate criterion for lensectomy,” he says. “I believe that this is a subgroup in which an iridotomy wouldn’t be beneficial. It was shown not to benefit the patient, in addition to being less cost-effective than clear lens extraction.”
Pearls
Here are some pearls for successful cataract surgery in glaucoma patients:
• Consider performing the MIGS procedure at the beginning of the case. “Most people tend to do their MIGS procedure at the end of the case,” Dr. Frenkel says. “For several years, I did it at the end of the case but then at the suggestion of someone, I tried doing the MIGS at the beginning. I found I liked that because the cataract was still in there, maintaining good chamber stability. The amount of bleeding is very minimal after most of these procedures, so it doesn’t really obscure the view. I also use less viscoelastic when I do the MIGS at the beginning because I’ve already filled the chamber with Viscoat at the start of the case.
“I prefer to do the MIGS procedure at the beginning of the cataract surgery now, but it can be done either way,” he continues. “It’s really a personal preference. For younger surgeons in training, it can be nice because it ensures them a clear view and good opportunity to do the MIGS procedure. Also, if they’re just starting out and have a hard time getting [the MIGS done] at the beginning, it gives them a second shot at the end of the case as well when they can try again.”
• Clear PAS using an iris stretching technique. Chronic angle closure patients frequently have PAS that need to be cleared. Dr. Grayson says he prefers a stretching technique for the iris, but if that’s insufficient, then he turns to iris hooks. “I don’t use Malyugin rings because I feel that they compromise maneuverability in the eye, especially in a hyperope with a shallow chamber with chronic angle closure,” he says. “Using iris hooks is a precise way to get pupillary dilation and have more maneuverability in the eye. Plus, if you do need more capsular support, you already have iris hooks in the eye. Those hooks can be moved slightly to engage the capsular bag to give you more stability. If you have a Malyugin ring in place, it can be difficult to get all the additional hooks over the ring to then support the capsular bag. Sometimes you may even take out the ring and put in a clip, which is a lot of maneuvers inside the eye.”
• Add goniosynechialysis to cataract surgery in angle-closure patients. Performing goniosynechialysis in conjunction with cataract surgery is a valuable adjunct in the chronic angle-closure patient, according to Dr. Grayson. “Very frequently, the pressures will come down and stay down and much of the synechiae won’t re-form if you clear out the angle and allow better access of the trabecular meshwork to the aqueous,” he explains. “Occasionally, I’ll do an additional Miostat infusion to keep the pupil down after goniosynechialysis for a few days to allow better aqueous access to the trabecular meshwork.”
• Try supracapsular-tilt nuclear disassembly in pseudoexfoliation patients. “If a pseudoexfoliation patient has zonular issues, you have to be much more careful disassembling the nucleus,” Dr. Grayson says. “I prefer a supracapsular tilt procedure where the nucleus is tilted approximately one-third of the iris plane and the remainder is in the capsular bag. However, this tilt maneuver sometimes places stress on the zonules, which may be compromised, so if I feel there’s too much mobility, I’d defer to a quadrantic cracking technique in the capsular bag, which is going to be a little bit less traumatic to the zonules.
• Leave the eye firm in a patient with a previous trabeculectomy. These patients may be prone to postoperative hypotony. “If the trabeculectomy is working, you could consider leaving the eye a little firmer, so the pressure doesn’t get too low,” Dr. Frenkel advises. “I usually recommend leaving the eye very firm at the end of the case to reduce the risk of hyphema. Leaving the firm helps tamponade any bleeding or at least reduces the risk of it. Obviously, you don’t want to leave the eye so firm that the pressure makes it so the patient can’t see anything. There’s a test where you ask the patient if they can see the lights, and if they can’t then their pressure is too high.
“Find a balance between leaving the eye firm enough that it’s going to reduce the risk of bleeding but don’t leave it so high that the patient has really high pressure overnight. You may also consider tapering steroids more slowly if you’re worried about the inflammation causing trab or tube failure, though this should be done on a case-by-case basis.”
• Consider shortening a tube shunt if it’s an issue. Dr. Okeke says, “If the tube is in contact with the cornea or cataract, you can shorten it using intraocular scissors and Utrata forceps to remove it from the eye; a Sinskey hook through a paracentesis will stabilize the tube as you perform this maneuver. Be sure to use Viscoat to protect the endothelium and reapply as needed during the procedure.”
• Use a higher steroid regimen in a filtering bleb patient. Patients with filtering blebs usually require more aggressive postop steroid treatment to control inflammation. “If you’re operating on a cataract in a patient who already has a filtering bleb, you want to ensure you’re using a higher postop steroid regimen, such as every two hours, preferably with something like Durezol. Pred Forte and its generics aren’t quite as potent. We’ll also combine a goniotomy in a patient who’s had a filtering bleb if they’re on additional medications if their filtering bleb isn’t functioning at 100-percent efficiency. So, for someone who’s on two medications along with a filtering bleb, we’ll do cataract along with goniotomy.”
Postoperative Steroids
Monitoring patients’ pressures is important. For certain patients, it may be necessary to check them more frequently in the postoperative period to make sure that their pressure hasn’t spiked. “Sometimes, after a goniotomy, we’ll keep the patients on pilocarpine for a month or so since pilocarpine drops reduce the risk of PAS developing,” Dr. Frenkel explains. “I also counsel patients to continue their glaucoma medication while in the immediate postop period, because of the risk of high pressure the day after surgery and the risk of steroid response within the postoperative period. Many glaucoma patients tend to be steroid responders. I tend to say, “Keep on your medications while you’re on your steroids, and then we’ll reassess after that.’ ”
Dr. Grayson says that patients who already have glaucoma or patients who are predisposed to developing glaucoma may exhibit steroid response, where using steroids, especially a potent one like Durezol, could cause an elevation of pressure. “These patients need steroids after their cataract surgery to decrease inflammation, so you have to follow them closely,” he says. “For example, if a patient had a combined cataract-goniotomy procedure, they’ll probably end up on Durezol or Pred Forte four times a day. Instead of seeing that patient at the three-week postop, you may want to see them one day and then one or two weeks postop to make sure that they don’t have a pressure spike to 30 mmHg.
“It just requires more attentiveness,” he says. “You may have to then stop the steroid, switch to a non-steroidal, and then switch to a less potent steroid to regulate the pressure again—which is one of the reasons I don’t use intracameral postop steroid injections. Should that patient have a steroid response, and they’ve got intracameral steroids, those aren’t going anywhere. You may have a very hard time controlling their pressure because your only choices—especially with a glaucoma patient who’s already on glaucoma medications—would be something like Diamox. These aren’t patients in whom you can just add timolol and their pressure will go down. Odds are they’re already on one or two glaucoma medications. So, I avoid intracameral steroid injections in pretty much all my patients but especially in those glaucoma patients.”
Post-goniotomy patients with small hyphemas may also need more aggressive steroid treatment. Dr. Grayson notes that in any minimally invasive glaucoma procedure, there’s a risk of bleeding from either the device or the trauma to the eye during the procedure. He says that even a small amount of blood in the anterior chamber can cause a significant decrease in vision and an elevated pressure.
“Generally speaking, more steroids help clear that blood and squash the inflammation faster,” he says. “If there’s a hyphema from goniotomy, patients will be concerned because they’re not seeing that well, but if you want to increase their steroids, you have to follow these patients a little bit more closely, maybe every few days. We can’t let them just coast with a one-day and then a three-week postoperative visit until you know exactly what their intraocular pressure status is. So, they’ve got to be monitored. They’ve got to be watched to make sure there isn’t a steroid response. Especially with hyphema, which could in a matter of days lead to a spike to 40 mmHg, which could cause a significant amount of damage very fast. It’s important to be more on top of those patients.”
Dr. Okeke discloses consultant, researcher and/or speaker relationships with Alcon, Allergan, New World Medical, Sight Sciences, Santen, Nova Eye Medical and Glaukos. Drs. Frenkel and Grayson have no related financial disclosures.