As every ophthalmologist knows, medications—especially glaucoma medications—are worth-less if the patient doesn’t use them. Unfortunately, many studies (including some done by our group, which includes Dolly S. Chang, MD, Travis C. Frazier, MD, and David S. Friedman, MD) have confirmed that glaucoma patients often fail to adhere to their prescribed medication regimen.1-3 One study found that 45 percent of glaucoma patients use their drops less than 75 percent of the time.1
Of course, it’s possible to intervene in an attempt to encourage adherence. Our group has tried some fairly complex interventions that required staff time and effort, such as providing patient education in the clinic and calling patients to check up on them and remind them to use their medications. These strategies work, as we demonstrated in one of our recent studies involving automated dosing reminders; in that study, automated calls or texts linked to the patient’s personal health record increased adherence by 16 percent.4 The problem is that no practice can afford to do this with every glaucoma patient.
In fact, it’s not really necessary to expend that extra time and effort with every patient, because many patients take their drops as prescribed. For that reason, it makes sense to look for ways to identify those patients who are most likely to have poor adherence. Having that information would allow a practice to take the necessary steps to encourage adherence—but only with the patients most at risk of failing to take their meds appropriately.
 |
 |
Having already done research relating to adherence, we set out to find a simpler solution to the problem of patient adherence. With that in mind, our most recent study consisted of two parts: first, looking for a simple way to identify patients at risk of nonadherence; and second, finding a practical way to intervene when a patient is at risk.
As every clinician knows, we can’t tell much about adherence from the intraocular pressure we measure in the office, partly because people tend to use their drops more in line with their treatment protocol right before they see the doctor. So, the adequate pressure control we see in the office could be explained by the patient only using the drops correctly in the days leading up to the visit. (Some of the data from the Travatan dosing aid study confirmed that; patient adherence was higher in the weeks leading up to a visit, and right after a visit.1)
Our concern was that prior methods for identifying these patients were simply too complex to use in the clinic on a regular basis. Options such as sending everyone home with an electronic monitoring device are impractical, outside of a study. So, we put a high priority on creating something that would be usable in the average practice.
With that in mind, we decided to try to create a questionnaire—hopefully a very simple one—that could be given to patients in clinic and would provide the doctor with a good idea of which patients are most at risk of being nonadherent.
This study included 323 glaucoma patients undergoing once-daily prostaglandin therapy. We asked the subjects a group of questions that we thought might provide a clue regarding which of them were most likely to fail to adhere to treatment. That list included questions about everything we thought might be connected to adherence: overall health; depression; how people take their drops; and so forth. Our goal was to find the smallest number of questions we could use that would make a reasonable prediction about future adherence.
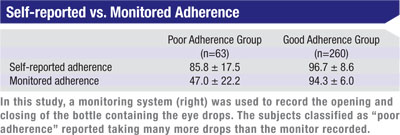
We monitored subjects electronically for three months, starting the monitoring two weeks after the baseline visit and concluding it two weeks before the follow-up visit, to minimize any effect from the artificially high adherence rates usu-ally seen immediately before and after clinic visits. We defined poor adherence as taking less than 75 percent of doses during the monitor-ing period. Overall, subjects took 85 ±22 percent of their drops correctly, with 20 percent of subjects showing poor adherence.
It was interesting to see which questions failed to correlate, in a multi-variate analysis, with the risk of being nonadherent. For example, we asked whether the patient knew individuals who are blind, or had gone blind from glaucoma. We asked questions about general health, thinking that people who were generally more ill might be less willing or able to get the eye drops in. We included some questions designed to indicate the patient’s level of glaucoma knowledge. None of these correlated with level of adherence.
Another question that didn’t play a role was: “Are you concerned that using glaucoma medications every day may cause long-term side effects?” We thought that if people were more concerned about side effects, they might be less likely to take the meds, but there was no correlation. We asked subjects to respond to the statement: “I don’t like the idea of using glaucoma drops.” That wasn’t predictive either. We also asked about the length of time they’d been using glaucoma drops, thinking that people who were newly diagnosed might be less focused on their disease. And, we tried using a short depression scale, on the premise that being some-what depressed might influence adherence. (Such a scale had turned out to be somewhat significant as an indicator of nonadherence in prior studies.) In our study, none of these fac-tors carried any significance as an indicator of nonadherence risk when combined in a multivariable model.
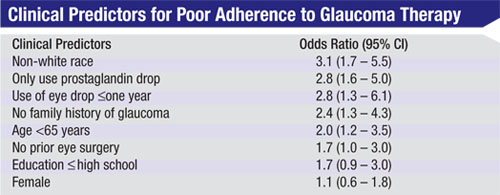 |
Ultimately, when we correlated the questionnaire responses with the study data showing actual adherence, we did find five questions that are highly predictive of which people are likely to not follow their treatment protocol:
- Is the patient of European de-scent, or a member of some other ethnic group? Those of European descent were much more likely to adhere to the treatment regimen.
- Does the patient have a family history of glaucoma? Not having any history of glaucoma in the family correlated with an increased risk of nonadherence, regardless of race.
Clearly, these two questions are likely to be answered during the history. Beyond that, patients can be directly asked three additional questions:
- On some days, do you forget to take one of your doses of glaucoma medications? If the answer is yes, the likelihood of non-adherence is greater.
- What percentage of your drops in the past month did you take correctly? Any answer lower than 95 percent turned out to be a risk factor.
- Can you name the medications you’re taking? If the patient can’t name them, that’s another risk factor.
Having a non-European heritage was a consistent risk factor in the last two studies we did—even when we controlled for things we thought might be related to that, such as socioeconomic status or educational background. This suggests that there’s some other factor we’re not measuring that makes those individuals more likely to be nonadherent. We haven’t found any indication of what that factor might be, but if we could identify it, it could help to determine the most effective intervention for that group. It’s an issue we need to understand better.
As far as the importance of a family history of glaucoma, I suspect that this factor influences how seriously the patient takes his eye disease. If you have a family member with the disease, it’s possible that he or she has done poorly, which would make an impression on you; you’d have first-hand experience that glaucoma is a serious disease requiring treatment. (Of course, these patients have probably also seen the family member use the drops, which could possibly affect their attitude about using drops themselves.) People without any family history of glaucoma may be less motivated at baseline to focus on a disease that otherwise has no symptoms and requires you to put in annoying eye drops every day.
How Well Does It Work?
How accurate is a prediction based on this set of questions? Our data found that these questions accurately identified about 69 percent of the people who ended up being nonadherent. Expressed a different way, the odds of someone being nonadherent if he is positive on this screening test is 14 times the odds of nonadherence if you randomly picked a patient. Furthermore, this system only had a 14 percent false-positive rate, so it didn’t place many adherent patients into the “risky” group.
| It's likely that you could ask additional questions and make an even more precise prediction about adherence, but the greater the number of questions you have to ask, the less practical this becomes. |
Given this reality, it’s possible that those who lie, or simply over-estimate their adherence, could fail the screening and produce a false negative. However, our correlations automatically take that into account. In fact, that may explain why a self-report of anything less than 95 percent adherence was a significant predictor of nonadherence.
At the practical level, a big ad-vantage of this set of questions is that it’s short. These questions could be presented in the form of a written questionnaire, something you’d give patients when they check into your clinic, or you could simply ask the questions that were not already answered by the history during the exam. In essence, they represent a trade-off between practicality and completeness. It’s likely that you could ask additional questions and make an even more precise prediction about adherence, but the greater the number of questions you have to ask, the less practical this becomes. We ended up feeling that this set of questions achieves a good balance between predictive accuracy and practicality.
We’re currently in the process of validating this using data from our previous studies; we hope to confirm that it would have been equally effective in those groups as well. We’ll soon be publishing those results, in hopes that others can take advantage of this approach.
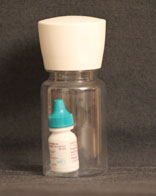
It’s worth noting that one limitation of our study is that we couldn’t determine whether a given patient actually got the drop onto her eye. This study was not set up to answer that question. In a prior study we used the Travatan dosing aid device, which was specific to Travatan; in this study we tried to be more general, so we put the patient’s bottle of drops into a clear medicine bottle so they could see what was in it (See photo, p. 113). The bottle had an electronic cap that allowed us to determine whether the patient opened the bottle. However, we had no way of knowing whether the patient actually succeeded in get-ting the drop onto the eye once the bottle was open.
This is a reflection of the reality that adherence is a chain of events: The doctor has to recognize the disease and prescribe the medication; the patient has to get the medication; the patient has to remember to use it; and the patient has to actually get it onto the eye. Any point at which that chain breaks down can result in the patient not getting the medicine. Our study only looked at whether the person had the bottle of drops and was attempting to use it.
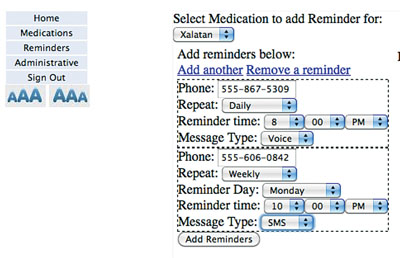 |
| A sample screen from the custom HealthVault application used by patients to specify the time at which they would like to receive the reminders (text or voice) to take their eye drops. |
The second part of our study was focused on finding a simple and effective intervention to address the potential for nonadherence. Clearly, few practices can afford to devote large amounts of staff time to educating glaucoma patients, which is not reimbursed. At the same time, several studies have already demonstrated that intervention can make a difference in adherence.5
For example, automated reminders that indicate it’s time to use the medication, sent by either voice or text messaging, produce a significant increase in adherence. We’ve also shown in our own studies that more extensive education, the use of personal phone calls and an alarm on the bottle are approaches that work well, but those arrangements are more complicated.
All of these strategies are interesting, and they may all end up being useful; it may turn out that different nonadherent individuals will need different types of interventions to get them to use their medications correctly.
Ironically, we haven’t implemented our nonadherence-identifying questionnaire system in our clinic yet, primarily because we don’t have an effective system for intervention in place. The interventions we’ve used in our studies have shown promise, but none of them are ready to be used on a larger scale in the clinic. So, we’re holding off implementing the questionnaire in our clinic until we’re in a position to really help the patients who are identified as being at risk.
On the Road to an Answer
When it comes to treating glaucoma, adherence is a very serious problem. Our hope is that our work will eventually help to address the problem by producing a fast and easy way to identify those patients most likely to fail to take their medications, as well as a practical way to help those patients once they’re identified. That will move us two steps down the road to a truly effective glaucoma solution.
Dr. Boland is an assistant professor in the Glaucoma Center of Excellence at the Wilmer Eye Institute of the Johns Hopkins University School of Medicine in Baltimore.
1. Okeke CO, Quigley HA, Jampel HD, Ying GS, Plyler RJ, Jiang Y, Friedman DS. Adherence with topical glaucoma medication monitored electronically the Travatan Dosing Aid study. Ophthalmology 2009:116:2:191-199.
2. Nordstrom BL, Friedman DS, Mozaffari,E, Quigley HA, Walker AM. Persistence and adherence with topical glaucoma therapy. Am J Ophthalmol 2005;140:4:598-606.
3. Friedman DS, Quigley HA, Gelb L, Tan J, Margolis J, Shah SN, Kim EE, Zimmerman T, Hahn SR. Using pharmacy claims data to study adherence to glaucoma medications: Methodology and findings of the Glaucoma Adherence and Persistency Study (GAPS). Invest Ophthalmol Vis Sci 2007;48:11:5052-5057.
4. Okeke CO, Quigley HA, Jampel HD, Ying GS, Plyler RJ, Jiang Y, Friedman DS. Interventions Improve Poor Adherence with Once Daily Glaucoma Medications in Electronically Monitored Patients.
Ophthalmology 2009;116:12:2286-93.
5. Haynes RB, Ackloo E, Sahota N, McDonald HP, Yao X. Interventions for enhancing medication adherence. Cochrane Database Syst Rev 2008;Issue 2. Art. No.: CD000011. DOI: 10.1002/14651858.CD000011.pub3



