|
Strategies that would help diagnose other diseases may be less helpful when the problem is glaucoma. For example, most physicians are trained to take a careful history as an aid to making a correct diagnosis. Occasionally, a patient at risk for glaucoma may be able to contribute some useful information, such as a strong family history that suggests increased risk. This is a rarity; even the most knowledgeable patients may be unaware that other family members are affected. Although some systemic conditions such as migraine, Raynaud’s phenomenon or low systemic blood pressure, African or Latino ancestry, older age or myopia might suggest a higher risk for glaucoma, the history is generally not as helpful as we might wish.
In most cases, detecting glaucoma requires an eye examination. Early in the course of the disease, this means looking at the optic nerve for signs of glaucomatous disease, such as damage to the disc rim or disc hemorrhages. (Fifty percent of the progression found in the Ocular Hypertension Treatment Study was discovered during the optic nerve exam.) Of course, even that isn’t foolproof; sometimes it’s not possible to be certain that the nerve is suffering early damage.
Given these realities, the best way to ensure that glaucoma and progression don’t go undetected is for the clinician to have a list of things at the top of his mind when faced with a patient. Here, I’d like to offer such a list, divided into six clinical pearls of “what not to miss:” 1) Try to make the best diagnosis. 2) Assess for major glaucoma risk factors. 3) Use all the tools at your disposal to detect progression. 4) Don’t underestimate a patient’s life expectancy. 5) Don’t assume that glaucomatous damage will only be peripheral. 6) Don’t miss a good screening opportunity.
Make the Right Diagnosis
A simple screening, such as taking the patient’s intraocular pressure, may uncover some patients with early primary open-angle glaucoma. But without a more complete exam, other forms of the disease will go undetected:
• Normal-tension glaucoma. Fifty percent of patients who have glaucoma may present with a normal pressure at the time of diagnosis. Many physicians apply the term “low-tension glaucoma” to these patients.
|
• Angle-closure glaucoma. Missing angle-closure glaucoma can have serious consequences, in part because you may end up treating the patient as if he has open-angle glaucoma. The vast majority of narrow-angle patients can be cured by laser iridotomy, but if you misdiagnose and give the patient medications instead, you may fail to prevent progression and make the disease more difficult to manage.
Unfortunately, early in the course of angle closure the IOP may be normal, with an area of iris-trabecular contact that’s easy to miss. The way to ensure that angle closure doesn’t escape your notice is to always do careful gonioscopy. Gonioscopy using an indentation gonioprism with an artificial tear interface is very easy to do and takes less than 15 seconds once you’re in the habit of doing it. (Dynamic gonioscopy, in which you indent the cornea, allows the clinician to differentiate synechial angle closure and appositional angle closure).
Virtually every patient should have gonioscopy at least once in a lifetime, preferably the first time you meet the patient. Even if the patient doesn’t have angle-closure glaucoma at the time of the exam, gonioscopy will tell you whether he has an anatomically narrow angle, putting him at greater risk. (Part of our job, after all, is to risk-stratify patients and determine how quickly they should return for the next exam.) If you discover that a patient has an anatomically narrow angle, you may want that person to come back in a year, so you can make sure the angle hasn’t narrowed further and turned into angle closure. On the other hand, if the patient has a wide-open angle, she may not need gonioscopy again for five or more years. In either case, gonioscopy should be done again at some point because the angle can change over time.
One alternative to gonioscopy is using an imaging technology such as optical coherence tomography to visualize the angle. However, OCT has limitations in this context. You can’t use indentation to see its effect on the angle; it takes more time than gonioscopy; and it’s a very expensive piece of equipment. I find gonioscopy to be quick, easy and effective.
• Exfoliation glaucoma. Another sign you don’t want to miss is exfoliation. Most of these patients will have normal pressure when you first meet them, but they are at high risk for the disease; patients with exfoliation progress more rapidly, have higher pressures and also have the greatest risk of complications during cataract surgery. It’s a critically important glaucoma diagnosis.
|
If you find that exfoliation is present, the patient should be examined every year for glaucoma, as the risk is high.
Watch for Risk Factors
Signs of elevated risk can be divided into two categories: those requiring continuous surveillance and those only needing a one-time assessment. Pachymetry—measuring corneal thickness—is a once-in-a-lifetime measurement. (Not every patient requires pachymetry, but every glaucoma patient and suspect does.) Of course, the thinner the cornea, the greater the risk. You should also check at least once for beta-zone peripapillary atrophy. Although this can change over time, usually you either have it or you don’t. If you have it, you’re at greater risk of glaucoma progression.
Other conditions require continuous surveillance. Disc hemorrhages are an ongoing concern; they are a very strong risk factor for progression. You need to look for them at regular intervals because they come and go; they may last anywhere from two weeks to four months. So, if the patient is an established glaucoma patient or suspect, you want to look at the disc at every visit. Dilation isn’t necessary; you can look at the disc using a 78- or 90-D lens to check for hemorrhages. It’s very quick, taking only about 10 seconds. Of course, if you find a hemorrhage you may want to follow the patient more carefully and/or advance treatment.
Don’t Miss Progression
Don’t Miss Progression
One of the best ways to avoid missing clinically meaningful progression is to take advantage of the technology that’s available today. Many of the tools we use contain software that can help identify subtle changes that have occurred between exams. We’re far less likely to catch these changes looking at numbers and printouts by ourselves. So, take advantage of the progression analysis software in your visual field, OCT and other devices.
The Life Expectancy Factor
When managing a disease like glaucoma that takes a long time to unfold, the amount of time left in a patient’s life is an important consideration. A slow rate of progression isn’t likely to lead to blindness in a patient who is 80 years old, but a patient progressing slowly at age 40 has a serious problem; he may live another 50 years. Dealing with this is a challenging aspect of glaucoma management that every clinician struggles with. How do you make a decision for a 40-year-old that might impact him 50 years later?
| ||||||
Obviously, we can’t predict how long any given patient is going to live, so we have to assume the patient may live longer than the average. This is also a consideration when deciding whether to pursue glaucoma surgery; you have to think about the ramifications down the road.
Check for Central Damage
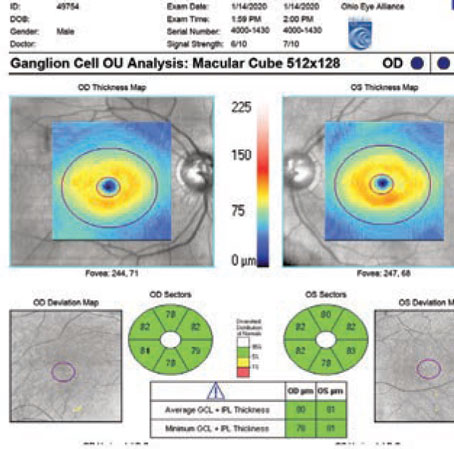
A fifth factor that can cause us to miss a diagnosis—or progression—is that we tend to think of glaucoma as a peripheral disease. Actually, glaucoma causes diffuse ganglion cell loss across the entire retina, and many patients have noticeable central loss; a patient with glaucoma can develop scotomas and other problems in the paracentral region. Loss of macular ganglion cells can lead to diminished contrast or reading ability. These problems will have visual impacts that are meaningful to the patient; in fact, they’re the kind of problems that are associated with falls and fractures.
If a glaucoma patient does have a visual complaint, it’s usually the result of central damage; peripheral loss doesn’t usually draw a patient’s attention unless it is bilateral and severe. Patients don’t come in and say, “I have a nasal step.” They come in saying “I’m having trouble reading the newspaper.” If you hear a complaint like that, you should assume central function is becoming impaired.
Currently, clinicians managing glaucoma seldom check the central region. Our group has been studying glaucoma’s effect on this region for the past 10 years, and we advocate testing every patient with a 10-degree visual field at some point, whether or not it looks like the patient has loss on the 24-2 test. We also recommend getting a baseline 10-degree visual field. If it’s abnormal, you’re aware of it. If it’s not, then at least you have a baseline for future comparison. Five years down the road you might want to repeat it to see if the disease is progressing centrally rather than peripherally.
Of course, you don’t have to do every test on the first day you encounter a patient; you may be seeing that patient for 30 years, so it’s perfectly reasonable to do some of the baseline tests over a period of one or two years. (Furthermore, if you attempt to run every possible test in one visit, the patient will likely wind up frustrated and unhappy.)
For example, suppose you have a new patient who is a glaucoma suspect. You get two visual fields, one at the first encounter and one after six or nine months. If the second field shows no change, you could get a 10-degree visual field the next time the patient comes in. The visit after that, you can go back to the 24-2. You haven’t lost anything by switching to the 10-degree test, and you haven’t made the patient come back for an extra visit. But you’ve gotten a base-line of the central field that might be important in the future.
A key part of monitoring what’s happening in the patient’s central vision is to simply listen to the patient. As noted above, when damage does extend to the central field it begins to affect vision in ways that patients notice. A patient may complain of difficulty reading or say that things look washed out. An astute patient may specify that his contrast sensitivity has decreased. Most ophthalmologists would assume this indicates the beginning of a cataract, but it could also indicate macular disease—or glaucoma. If a patient with known glaucoma tells you he’s having increased difficulty reading, but everything else looks the same, that’s probably a sign of progression even if the visual field hasn’t changed. In that situation you should definitely get a 10-degree visual field.
Screen Individuals at Risk
The last item on my list is to make sure you don’t miss a good screening opportunity. Whenever you know that someone may have an elevated risk of glaucoma, it’s worth doing whatever you can to see that that person is examined.
For example, we know that siblings of a POAG patient are at fairly high risk of having the disease—even higher than a child of the patient.1 Likewise, African Americans are more likely to be affected than Caucasians. So, it’s worth encouraging your glaucoma patients to have their siblings come in for an exam. If you examine five siblings of a glaucoma patient, odds are very good that you’ll find at least one individual with glaucoma. (That’s compared to 1 or 2 percent of the general population.) It is perhaps most important that African-American patients have their family members examined.
We could identify a lot of undetected and undiagnosed glaucoma just by doing this, and that could go a long way toward reducing unnecessary vision loss.
Being on the Lookout
Today, all of us are seeing more patients and we’re more pressed for time. Unfortunately, that just increases the odds that we’ll miss something potentially important. Almost every ophthalmologist dilates his patients at the first visit and periodically after that, and looking at the optic nerve is a part of a regular eye exam. But if the clinician is just thinking about taking a cataract out, he may very well miss signs of glaucoma. Likewise, if you’re focused on a patient’s complaint, you might not think to check for signs of glaucomatous damage. But every exam is an opportunity to catch the warning signs that glaucoma is present and the patient is at risk.
To make sure we don’t miss those signs, we need to be focused and use optimum detection and management strategies. Keeping the six points mentioned here in mind when examining a patient can help avoid a mis-diagnosis and make it easier to do an accurate risk assessment, stratifying patients as to who needs to come back soon and who needs more extensive testing. And that will mean less unnecessary vision loss. REVIEW
Dr. Liebmann is a clinical professor of ophthalmology at New York University School of Medicine, an adjunct professor of clinical ophthalmology at New York Medical College in Valhalla, N.Y., and director of glaucoma services at Manhattan Eye, Ear, and Throat Hospital and New York University Medical Center.
1. Tielsch JM, Katz J, Sommer A, Quigley HA, Javitt JC. Family history and risk of primary open angle glaucoma. The Baltimore Eye Survey. Arch Ophthalmol 1994;112:1:69-73.