Jetrea is approved in the United States for the treatment of symptomatic vitreomacular adhesion, an age-related progressive, sight-threatening condition that may lead to visual distortion, decreased visual acuity and central blindness.
ThromboGenics, which is commercializing Jetrea in the United States, plans to launch the already-diluted formulation of Jetrea in the first half of 2017. More information is available at thrombogenics.com.
Efficient In-Office Procedures
Reliance Medical Products and Haag-Streit USA announced the new I-OPS In-Office Procedure System at the American Society of Cataract and Refractive Surgery in May. Developed in collaboration with Christopher Riemann, MD, of the Cincinnati Eye Institute, I-OPS is an ergonomically optimized instrument delivery system that allows physicians to perform injections and other minor in-office procedures much more efficiently. I-OPS eliminates wasted movements, which saves time and improves patient flow, as well as patient safety and comfort, the company says.
Physicians can now perform a multitude of procedures without the need for an assistant or the need to turn around and reach for supplies, increasing productivity and efficiency. By eliminating the need for an assistant during procedures, the physician’s staff is also able to focus on documentation and other productive tasks.
The modular I-OPS System features a durable tray with space for up to nine interchangeable inserts. The ability to organize and set up procedures beforehand brings consistency and standardization to procedures. Physicians can keep a hand, and their attention, on the patient, reducing patient stress levels and increasing their comfort.
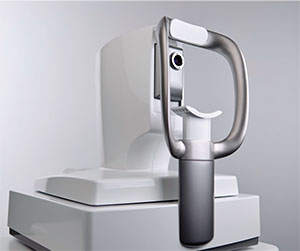 |
The I-OPS tray and accessory kit come with two syringe holders, deep and shallow cups and one each of a dropper bottle holder, twist bottle holder and gel pack holder. If the physician prefers to customize his system, inserts are available for purchase in any combination a la carte.
The basic I-OPS arm fits almost any manufacturer’s stand pole and is easy to move to swing out of the way of patients. It also comes with Reliance Medical Products standard one-year warranty. For information, visit haag-streit-usa.com/IOPS, or view the I-OPS video on YouTube.
AngioVue Retina from Optovue
Optovue announced the immediate availability of AngioVue Retina, a proprietary imaging system that provides retina specialists with a non-invasive, dyeless way to quickly visualize blood flow in the retina.
AngioVue Retina is configured with essential optical coherence tomography angiography and OCT features designed specifically for retinal practices to allow adoption of OCT and OCTA into the clinical workflow with minimal disruption.
Optovue’s AngioVue Retina provides retinal specialists with the ability to quickly visualize the presence or absence of retinal vessels and assess new information about the microvasculature with extraordinary detail. This information may be integrated with other diagnostic imaging results to form a complete picture of a patient’s disease state and their treatment options.
The company also announced its new DualTrac Motion Correction for use with both the AngioVue and AngioVue Retina systems, a two-level approach to correcting motion artifacts resulting from patient movement. The first level provides real-time correction for rapid eye movements, blinking or eye drifting. The second level occurs during imaging post-processing and corrects smaller levels of motion distortion. This combined approach results in robust motion correction for patients who have trouble directing their focus on a central point during an eye exam, which is essential for high quality OCTA imaging. For information, visit optovue.com.
Moria Adds to Punch Portfolio
Moria has extended its portfolio in purely endothelial graft preparation with new Single-Use Donor Vacuum Guarded Punches to safely assist corneal surgeons and/or eye-bank practitioners in the preparation of purely endothelial graft following the SCUBA (“no touch”) technique: the Guarded Punch.
Its 350-µm guarded blade is designed to make a non-penetrating trephination of the Descemet’s membrane all around 360° without any hinge, so delamination becomes an easy process.
Guarded punches are currently available in six diameters: 9.5- and 10.0-mm: for a peripheral scoring; 7.5-, 7.75-, 8.0-, 8.5-mm to finalize the purely endothelial graft size.
With the new guarded punches, just one press scores Descemet’s membrane, creating a clean, 360-degree stromal incision to easily find a cleavage plane for the consecutive delamination. REVIEW



