Two recent studies looked at ways ophthalmologists might be able to screen for disease more effectively.
In the first study, researchers from Washington University in St. Louis and Blue Sky Neurology of Denver investigated whether or not retinal changes might allow clinicians to catch preclinical Alzheimer’s disease.
The case-control study analyzed 30 individuals from Washington University’s Alzheimer’s research center. All participants were tested to ensure they were cognitively normal. Individuals with prior ophthalmic disease, media opacity, diabetes or uncontrolled hypertension were excluded. Fourteen participants had biomarkers positive for Alzheimer’s and, because of that, a diagnosis of preclinical disease. Sixteen patients without biomarkers acted as a control group.
“We were interested in looking at very early disease in which there’s no evidence of any cognitive decline,” says study co-author Raj Apte, MD, PhD. “However, we knew they had Alzheimer’s because they had [biomarkers] tau or amyloid-b. We felt that if we studied the test in later disease when there’s already cognitive decline, what’s the value of another test in that instance?
“We were interested in looking at the retina because, in more advanced neurodegenerative diseases, there’s been some evidence of retinal thinning in the past, both in the neuro and vascular components,” Dr. Apte adds. “However, no study has shown this involvement early in the disease—in other words, where there’s biomarker positivity but no cognitive decline. So, we were looking at the thickness of the neuronal layers—the inner and outer layers of retina—and because we were using OCTA, we were looking at the vascular plexuses, as well.”
Though a number of parameters were evaluated, the one that proved significant was the foveal avascular zone: The FAZ was increased in the biomarker-positive group compared to the control group (mean: 0.364 ±0.095 vs 0.275 ±0.060 mm2; p=0.002). Also, the mean inner foveal thickness was decreased in the biomarker-positive group (66.0 ±9.9 vs 75.4 ±10.6 µm; p=0.03).
“We removed a number of possible confounders—for instance, patients with diabetic retinopathy get an increase in their FAZ also—so this gave us relatively pristine retinas to analyze,” Dr. Apte explains. “However, even though the enlargement in the FAZ was the strongest correlator, it’s not ready for prime time. In other words, you can’t take a group of people and have a metric above or below which you’re comfortable saying anything definitive about disease diagnosis.”
The other study that sought to learn more about screening methods evaluated several published studies of 11 artificial intelligence, deep-learning-based diabetic retinopathy screening algorithms to determine the algorithms’ accuracy.
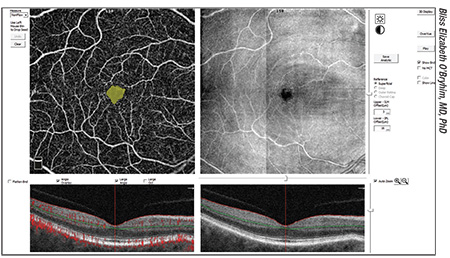 |
| Researchers say that enlargement of the foveal avascular zone as imaged on OCTA may be a sign of preclinical Alzheimer’s disease. |
In order to qualify for the comparison, a study in question had to provide a DR grading scale, a human grader as a reference standard and a deep-learning performance score.
Eight of the 11 studies reported sensitivity and specificity of 80.28 to 100 percent and 84 to 99 percent, respectively. Two studies reported accuracies of 78.7 and 81 percent. One study provided an area under the receiver operating curve of 0.955.
Study co-author Jakob Grauslund, MD, PhD, head of research and clinical professor of ophthalmology at Denmark’s Odense University Hospital, says the latest algorithms were challenging to analyze, but appear to be improvements over past approaches.
“We found that it’s very difficult to compare these studies against each other,” he says. “You had different methodologies in the studies, and they were performed using different subjects from different ethnic groups that resulted in different pigmentations of the retina that, again, yielded different results. So we couldn’t perform a true meta-analysis. But, taking those factors into account, when compared to earlier forms of automated grading in DR, these were very strong results.
“I was surprised, though, that almost all of the studies only used a single retinal field in the DR evaluation,” Dr. Grauslund continues. “As a clinician, that’s not the gold standard. I expected more would use ETDRS seven-field studies or at least two retinal fields, because that would make it easier to implement their findings in the clinic. Also, a lot of the studies only included images of very high quality, which might be the reason they got very good results. But, if you want to use them in a real-world screening facility, you need your system to be able to recognize poor-quality images. Another surprise was that few studies included screening for DME, which you need to do if you want to use a system in a real-world clinic.” He adds that most of the algorithms had difficulty distinguishing between patients with no retinopathy and those with only mild, non-proliferative disease. This might impact the exam intervals for patients who actually don’t need frequent follow-up.
“This kind of screening is much better than previous versions, but we’re not there yet,” Dr. Grauslund says. REVIEW
1. Bliss E, Apte R, Kung N, et al. Association of preclinical Alzheimer disease with optical coherence tomographic angio-graphy findings. Ophthalmol 2018;136:11:1242-1248.
2. Nielsen K, Lautrup M, Andersen J, et al. Deep learning–based algorithms in screening of diabetic retinopathy: A systematic review of diagnostic performance. Ophthalmology Retina 2018;1-11 [epub ahead of print].



