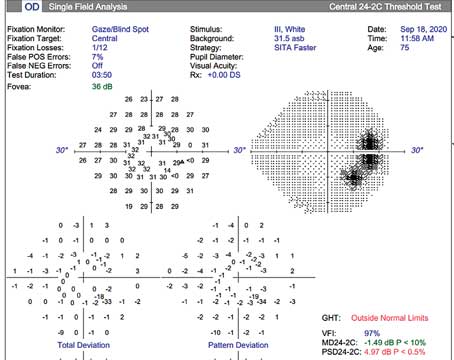
As glaucoma specialists, we’re all humbled by treating glaucoma and watching patients progress despite maintaining target pressure with drops, lasers, medications or regular monitoring with OCT and visual fields. When I see these patients, some of the questions that come to mind are: How can we find you sooner? Is there a technology we can use to identify fast progressors or those at the end of their disease? Is there anything we can offer these patients to enhance their lives despite vision loss from glaucoma?
As a clinician scientist, I’m constantly thinking about how the findings presented at our meetings might be implemented in the clinic. Here, I’ll review some exciting advances in our field from ARVO 2023: Emerging technologies for studying architecture, dynamics and function; improving deep learning and AI to diagnose glaucoma; and developing therapies to improve care after diagnosis and disease progression.
Emerging Technology
One of the technologies we’ve watched evolve in subsequent ARVO meetings is Detection of Apoptosing Retinal Cells (DARC), a method of visualizing apoptosis in real time. With this technology, we can detect glaucomatous cell damage at earlier stages in the disease and may one day be able to employ it as evidence of treatment efficacy.
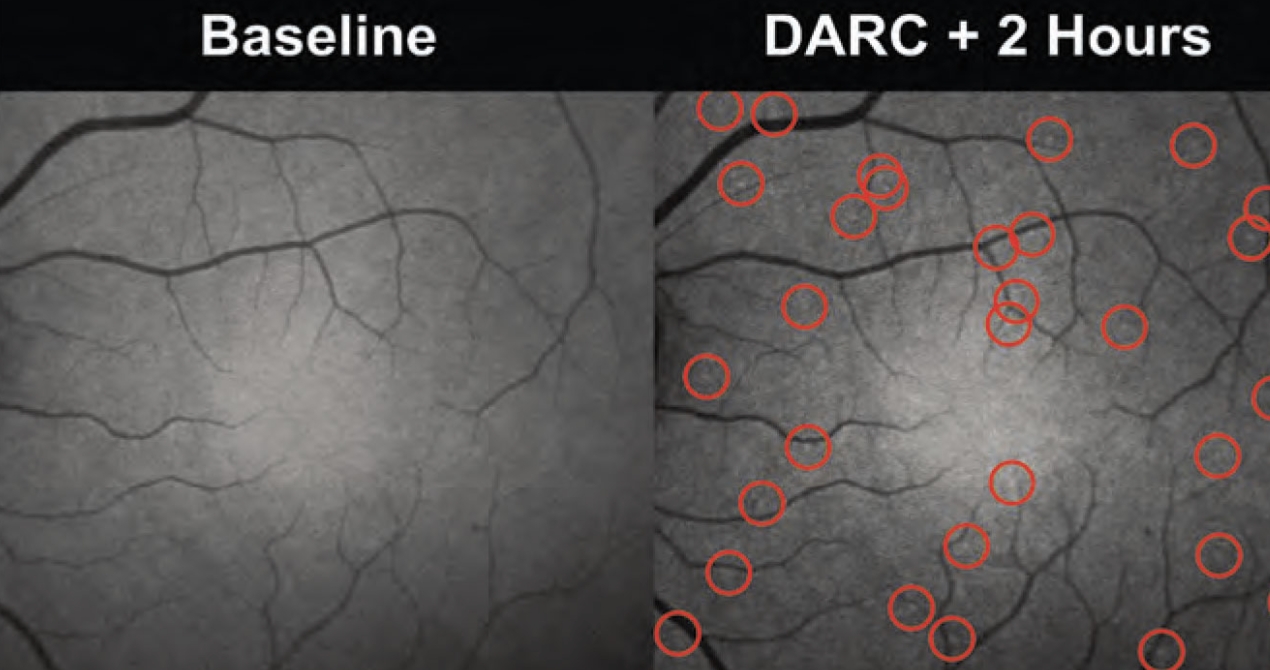 |
| Once apoptosing cells are made visible by DARC technology, they can be imaged and measured using confocal scanning laser ophthalmoscopy. Above, bright spots corresponding to cells externalizing phosphotidylserine are detectable in a patient's retina two hours after administration of the fluorescent Annexin V. The spots are identified with a convolutional neural network algorithm. (Courtesy Professor M. Francesca Cordeiro) |
DARC relies on the protein Annexin V as a sensitive probe to identify dying retinal ganglion cells with the assistance of a fluorescent dye. During apoptosis, which begins early in the disease process, the cell surface undergoes multiple changes. One of those changes is the transport of phosphotidylserine from the inside of the cell to the cell surface. When these receptors move from the inside to the outside of the cell, Annexin V is able to bind to the cell surface.
DARC is now able to be used in human diseases. Recently, Professor M. Francesca Cordeiro of Imperial College London and University College London in the United Kingdom, and colleagues demonstrated that distinct types of DARC spots could be observed in vivo and that an increased proportion of irregularly shaped DARC spots was associated with diseases that kill retinal ganglion cells, such as glaucoma, multiple sclerosis and demyelinating optic neuritis.
James Owler, of Novai, the company developing DARC, presented the team’s findings at ARVO. The study1 included 36 healthy adults (mean age: 45.97) and patients with glaucoma (n=20, mean age: 61.74) and multiple sclerosis optic neuritis (n=12, mean age: 44.47). The researchers applied hysteresis thresholding for each DARC spot to identify perimeters and extract a set of morphometrics: spot area; eccentricity; etc. Hierarchical clustering of these morphometrics showed distinct populations of DARC spots—C0: small regular; C1: irregular; C2: large regular. The most common type of DARC spot type in each eye differed significantly among the groups.
This study has massive implications. We now have the ability to use live imaging to study cellular architecture. By identifying which cells are dying and their dynamics, we could potentially demonstrate loss of function and use this technology to develop neuroprotective therapies in the future.
Deep Learning
This type of artificial intelligence uses representation-learning methods to extract patterns from raw data. In ophthalmology, deep learning algorithms are primarily used in diabetic retinopathy and retinopathy of prematurity, but the technology is also being investigated in glaucoma and age-related macular degeneration, in particular with the prediction of progression based on fundus images and OCT volume and thickness maps.
One of the challenges deep learning models in glaucoma face is imbalanced datasets used to train the programs, as many are skewed toward “glaucoma cases.” There haven’t been many large population datasets of patients who are glaucoma suspects, and many studies throw out glaucoma suspect samples. However, such imbalanced training models can lead to inaccurate estimation and generalization failure.
New research presented at ARVO by Ashkan Abbasi, PhD, of the Casey Eye Institute, found that studying glaucoma suspect eyes using a semi-supervised, resampling approach can lead to improvements in AI diagnostic performance that ameliorate imbalanced learning.2 He and his colleagues developed a baseline 3D convolutional neural network and trained it on a real-world glaucoma dataset, which is naturally skewed toward glaucoma cases. They applied three methods (reweighting samples, data resampling to form balanced batches and semi-supervised learning) to glaucoma suspect data and found statistically significant improvements in all metrics compared with traditional supervised training using weighted cross-entropy loss (95.24-percent mean accuracy, 97.42-percent F1 score and 95.64 percent AUCROC vs. 92.88 percent, 96.12 percent and 92.72 percent, respectively). The authors emphasized that making use of data with uncertain diagnosis can alleviate some imbalance in datasets, so long as combined semi-supervised and class-imbalanced learning strategies are used.
With this approach, the algorithm was also able to determine not only whether a patient had glaucoma or not, but who was a glaucoma suspect, who skewed more toward having pre-perimetric disease and who was a healthy control. This type of tool could potentially determine which glaucoma suspects might convert.
Improving Care
 |
| Visual function improvement was observed in patients after using an augmented reality device consisting of AR software and a smartphone paired with a Samsung Gear headset like the one above.3 (Courtesy Samsung) |
At the end-stages of glaucoma, there’s still hope for patients’ residual vision. Technologies such as augmented reality and virtual reality may help. AR overlays digital information onto a real-world environment while VR is a completely immersive, computer-generated environment. A prospective study presented at ARVO by Sarika Gopalakrishnan, PhD, FAAO, of the Envision Research Institute in Wichita, Kansas, found that visual function improvement was seen in patients with low vision (n=100) after using an AR device.3 At baseline, 21 percent of patients had central field loss, 35 percent had peripheral field loss and 44 percent had overall blurred vision. Common causes for moderate and severe visual impairment included myopic macular degeneration, cone dystrophy, retinitis pigmentosa and optic atrophy. After using the AR device, patients demonstrated significant improvements in distance vision (1.1 to 0.15 logMAR), near vision (0.6 to 0.3 logMAR) and two-week visual function score (VA LV VFQ-48; 0.35 to 1.90).
Retinal engineering and retinal ganglion cell replacement are two other promising avenues for glaucoma care. Retinal ganglion cell death is irreversible, but repopulation approaches have the potential to reverse optic neuropathy-associated vision loss, provided that the transplanted cells establish themselves.
Currently, the RReSTORe (Retinal ganglion cell Repopulation, Stem cell Transplantation, and Optic nerve Regeneration) Consortium, is attempting to overcome barriers to vision replacement in patients with optic neuropathies.4 The Consortium’s goals include defining and prioritizing the most critical challenges and questions related to retinal ganglion cell regeneration; brainstorming innovative tools and experimental approaches to meet these challenges; and fostering collaborative scientific research among diverse investigators.
RReSTORe Consortium program leader, Thomas V. Johnson, MD, PhD, of the Wilmer Eye Institute, Johns Hopkins School of Medicine, explained in his ARVO presentation5 that the group is currently developing techniques to differentiate retinal ganglion cells from human stem cells and how to enhance the transplantation of retinal ganglion cells into live retina. His group found that the internal limiting membrane was a barrier to retinal cell engrafting and is working to evaluate other cell integration approaches. The Consortium will hold a mini-symposium on the topic at this year’s ARVO 2024 in Seattle.
In summary, there are several exciting developments in glaucomatology that have the potential to alter the course of clinical treatment. Many of these discoveries in imaging, artificial intelligence, augmented reality and stem cell science have clear translational potential.
Dr. Ross is an assistant professor of ophthalmology and neuro-ophthalmology at the University of Pennsylvania, Scheie Eye Institute. In addition to clinical responsibilities, she leads a basic science laboratory with the goal of understanding combined molecular mechanisms of glaucoma. She also acts as a scientific consultant with Gyroscope Therapeutics and Noveome Therapeutics. She holds a patent for gene therapy for ocular disorders.
Dr. Singh is a professor of ophthalmology and chief of the Glaucoma Division at Stanford University School of Medicine. He is a consultant to Alcon, Allergan, Santen, Sight Sciences, Glaukos and Ivantis. Dr. Netland is Vernah Scott Moyston Professor and Chair at the University of Virginia in Charlottesville.
1. Owler J, Daws RE, Lotery P, et al. Abnormal retinal apoptosis morphometry in glaucoma and optic neuritis. Invest Ophthalmol Vis Sci 2023;64:8:4676.
2. Abbasi A, Antony BJ, Gowrisankaran S, et al. Can glaucoma suspect data help to improve the performance of glaucoma diagnosis? Transl Vis Sci Technol 2023;12:6.
3. Gopalakrishnan S. Role of head mounted augmented reality device in improving visual function of individuals with low vision. Invest Ophthalmol Vis Sci 2023;64:5522.
4. Johnson TV, Baranov P, Di Polo A, Ross AG, et al. The Retinal ganglion cell Repopulation, Stem cell Transplantation, and Optic nerve Regeneration Consortium. Ophthalmology Science 2023. [Epub August 24, 2023].
5. Johnson T. Retinal engineering and RGC replacement: Enhancing integration of transplanted RGCs. Invest Ophthalmol Vis Sci 2023;64:929.