As the prevalence of dry eye continues to grow, so does the need for effective therapies. There are a number of new therapeutic approaches in development for the management of this condition that could help improve outcomes and quality of life for these patients. Here, we’ll take a closer look at these agents and where they currently stand in the developmental process.
Reproxalap (Aldeyra)
This novel, small-molecule reactive aldehyde species (RASP) inhibitor lessens ocular inflammation in dry-eye disease via a unique mechanism of action. RASP are pro-inflammatory molecules that are elevated in ocular and systemic inflammatory disease. Reproxalap mitigates inflammation by binding the free aldehydes, which leads to a reduction in RASP levels.
The company recently released findings from a 12-month safety clinical trial of reproxalap that included 447 dry-eye patients—299 received reproxalap and 148 were treated with vehicle. Treatment-related serious adverse events in ocular safety (the primary endpoint) weren’t observed.
Similar ocular safety events were reported across both treatment groups. A post-hoc analysis showed that reproxalap was superior to vehicle in improvement from baseline in distance visual acuity.
Of the many agents currently in development, reproxalap is one that stands out, according to Robert Latkany, MD, an ophthalmologist who practices in New York City. “Ocular surface disease is heavily influenced by inflammation. While steroids can help a lot of these patients, they also carry significant side effects,” he explains.
“This is an alternative that comes with minimal side effects,” says Dr. Latkany. “Also, this is one of the first drugs that I’m aware of that’s attacking not only dry eye, but also allergic conjunctivitis. Many ocular surface disease patients don’t just have dry eye, so the ability to address both dry eye and allergies is very exciting.”
Based on data from five clinical trials, including the phase III TRANQUILITY-2 study (NCT05062330), the U.S. Food and Drug Administration accepted the New Drug Application for topical ocular reproxalap in February. The assigned Prescription Drug User Fee Act (PDUFA) date is November 23, 2023.
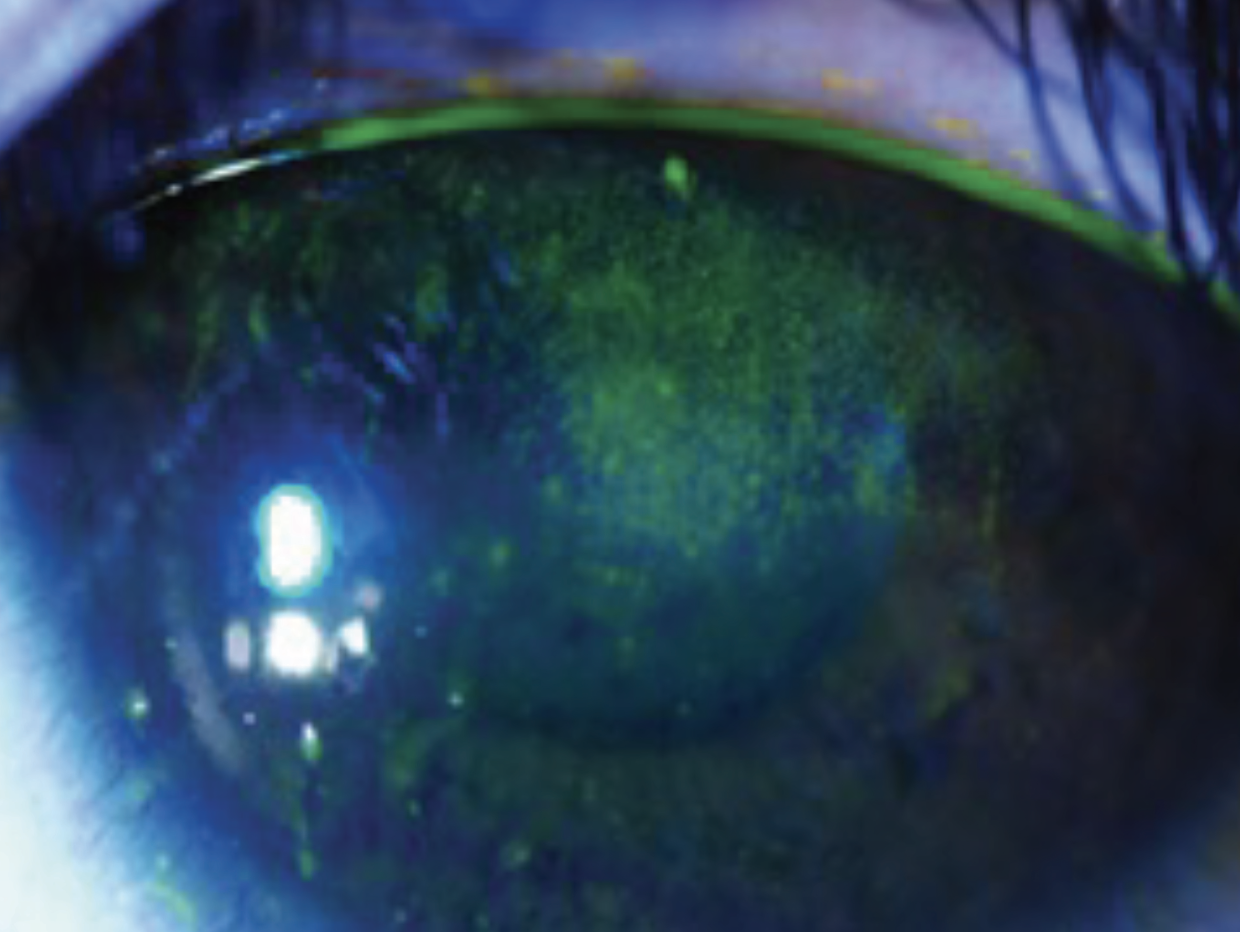 |
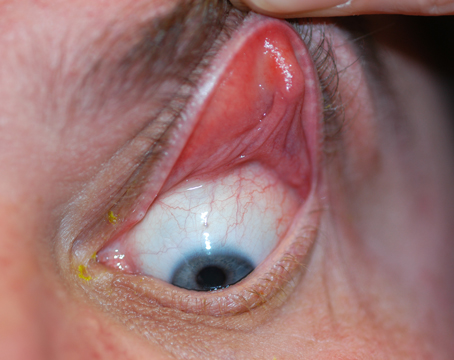
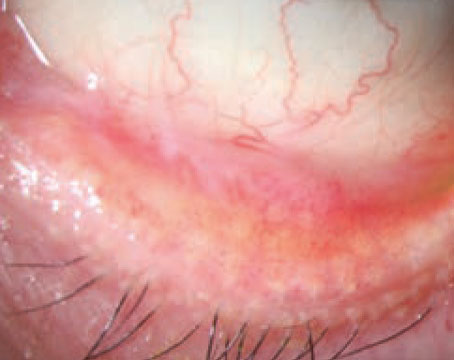
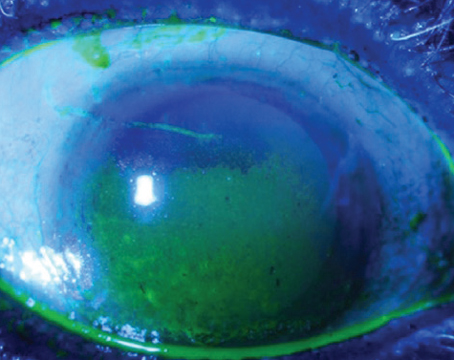
| Future therapies for dry eye aim to reduce potential damage to epithelial cells in an effort to preserve patients’ visual function. (Courtesy Esen Akpek, MD) |
CyclASol (Novaliq)
CyclASol—a topical anti-inflammatory and immunomodulating ophthalmic drug solution—contains 0.1% cyclosporine A in EyeSol, which is a water-free technology designed by the company.
EyeSol “increases residual time on the ocular surface and enables a high bio-availability in the target tissues to unfold the full potential of cyclosporine A and fast onset of action within two weeks,” according to Novaliq.
Data from the Phase III ESSENCE-2 trial were recently published in JAMA Ophthalmology.1 In this study, 834 patients were randomly assigned to one of two treatment groups: cyclosporine or vehicle. The primary endpoints were changes from baseline in total corneal fluorescein staining and in dryness score at day 29.
“Even though the vehicle itself is a very good dry-eye treatment, there was separation between the vehicle and active group,” says first author Esen Akpek, MD, a professor of ophthalmology and rheumatology at Johns Hopkins University School of Medicine, and director of the Ocular Surface Disease and Dry Eye Clinic at the Wilmer Eye Institute in Baltimore.
The trial showed that treatment with CyclASol “results in early therapeutic effects on the ocular surface compared with vehicle,” Dr. Akpek reports. “The responder analyses suggest that the effect is clinically meaningful in 71.6 percent of participants in the cyclosporine group.”
The study authors observed improvements in total and central corneal staining score after only two weeks of treatment, with persistent efficacy through day 29. “Timing of improvement is a relevant and valuable finding,” according to Dr. Akpek, especially for patients who are undergoing cataract or refractive surgery.
The FDA has accepted the New Drug Application for CyclASol and set a PDUFA target action date of June 8, 2023.
B+L, Novaliq Eye Drop Approved For Dry Eye After several years of clinical trials and anticipation, Bausch + Lomb and Novaliq just announced the FDA approval of a new eye drop for evaporative dry eye. Sold under the name Miebo (perfluorohexyloctane ophthalmic solution; formerly known as NOV03), the drop is indicated to treat the signs and symptoms of dry-eye disease. The solution is also the first on the market to directly target tear evaporation—a leading cause of DED. Miebo achieved primary endpoints and demonstrated good tolerability in all three trial phases, the company says. The FDA approval of Miebo was based on results from two 57-day, multicenter, randomized, double-masked, saline-controlled studies, GOBI and MOJAVE, which enrolled a total of 1,217 patients with a history of DED and clinical signs of meibomian gland dysfunction. In the GOBI and MOJAVE Phase III studies, Miebo met both primary sign and symptom efficacy endpoints. The two primary endpoints were change from baseline at week eight (day 57 ± 2) in total corneal fluorescein staining (tCFS) and eye dryness Visual Analog Scale (VAS) score. Patients experienced relief of symptoms as early as day 15 and through day 57 with statistically significant reduction in VAS eye dryness score favoring Miebo observed in both studies, the company says. B+L adds that, at days 15 and day 57, a significant reduction in tCFS favoring Miebo was observed in both studies. The most common adverse reactions were blurred vision (1.3 to 3 percent) and eye redness (1 to 3 percent). With MGD contributing to the majority of dry-eye cases, Miebo may offer this patient population an alternative therapeutic option to other types of drops and in-office procedures, the company says. |
TP-03 (Tarsus)
Clinical trials are currently underway examining TP-03 (lotilaner ophthalmic solution, 0.25%)—a novel treatment for Demodex blepharitis. Lotilaner is an anti-parasitic agent that paralyzes and eradicates Demodex mites by selectively inhibiting the GABA-Cl channels. “Demodex is a highly underdiagnosed and underappreciated condition,” says Dr. Akpek, while noting that treatment can be frustrating. “In-office procedures are typically the most effective options. However, this can be expensive or inconvenient for patients. I am very excited about new options that could offer additional treatment avenues.”
Data from the Phase III Saturn-2 study were released by the company and presented during the American Academy of Ophthalmology 2022 Annual Meeting (Session ID PO058). The primary endpoint (complete collarette cure) as well as all secondary endpoints (mite eradication, erythema cure, erythema/complete collarette composite cure) were met, and TP-03 had a favorable safety profile.
In this randomized, controlled, double-masked study, 412 patients with Demodex blepharitis were enrolled. Study participants self-administered one drop of TP-03 twice per day in each eye for six weeks. They received no treatment for blepharitis, during the trial or 14 days prior to enrollment.
Fifty-six percent of patients on TP-03 achieved complete collarette cure versus 13 percent in the vehicle group. Additionally, mite eradication was achieved in 52 percent of patients on TP-03 compared to 14 percent on vehicle. The safety profile was consistent to the Phase IIb/III Saturn-1 trial, demonstrating that TP-03 was well-tolerated.
In November 2022, the FDA approved the New Drug Application for TP-03. The target action date for the PDUFA is August 25, 2023.
RGN-259 (RegeneRx)
This Tβ4-based sterile and preservative-free eye drop was developed for a number of ophthalmic indications, including dry-eye disease. Topline results from ARISE-3, which included 700 patients, were released in 2021. The company reported that the trial didn’t meet its primary outcomes; however, the results showed statistically significant improvement in ocular grittiness at one and two weeks post-treatment.
The drops continued to demonstrate safety in this patient population, consistent with previous clinical trials. There were no signs of adverse events with only mild to moderate events observed in both arms. The most common ophthalmic adverse event was mild ocular pain upon instillation.
Towards the end of 2022, RegeneRx submitted a request for the Special Protocol Assessment (SPA) to the FDA for an in-depth discussion and assessment of the clinical protocol for a fourth Phase III clinical trial (ARISE-4). This study is expected begin in 2023.
AR-15512 (Aerie/Alcon Pharmaceuticals)
Another drug under development is the investigational eye drop AR-15512, a transient receptor potential melastatin 8 (TRPM8) agonist.
Findings from the Phase IIb COMET-1 study, which were published in 2022, showed that AR-15512 demonstrated statistically significant improvements in dry-eye disease signs, symptoms and disease-related quality of life.3 Although predefined co-primary study endpoints were not met, AR-15512 is a promising approach, according to Dr. Latkany, who emphasizes the need for further research.
The first patient has been dosed in the Phase III registrational COMET-3 study—the second of three trials, according to Aerie Pharmaceuticals. The COMET-4 safety study (NCT05493111) was initiated in November 2022.
The company, which was acquired by Alcon in November 2022, plans to complete the AR-15512 registrational program in 2023 and, if clinically successful, will file a New Drug Application with the FDA in 2024.
Visomitin (Mitotech)
Visomitin—a cardiolipin peroxidation inhibitor—is currently being studied as a possible treatment for a variety of indications, including dry-eye disease. This agent uses a novel, multimodal action that targets inflammation, corneal/conjunctival damage, tear deficiency, and gland tissue regeneration.
Findings from the Phase IIb/III VISTA-1 and Phase III VISTA-2 studies were presented during the 2022 Association for Research in Vision and Ophthalmology meeting. Visomitin (SkQ1 ophthalmic solution) showed statistically significant effects on clearing of corneal staining in both studies, which, according to the study authors, is a “highly clinically relevant result.”
These findings support clearing of corneal fluorescein staining as the primary endpoint for the upcoming VISTA-3 study.
Lacripep (Tear Solutions)
Developed to treat symptoms of dry eye and primary Sjögren’s Syndrome, Lacripep is derived from the lacritin protein—a first-in-class topical therapy that preserves lacritin’s bioactivity.
Findings from a first-in-human study of Lacripep among patients with primary Sjögren’s syndrome showed clinically significant improvements in specific signs and symptoms.4 Study participants started to experience a positive impact after two weeks of treatment.
This research established Lacripep’s safety and tolerability as well as its ability to significantly improve clinically relevant signs and symptoms of dry-eye disease, according to Tear Solutions. Further investigation is ongoing to determine appropriate dosing and concentration.
AZR-MD-001 (Azura Ophthalmics)
AZR-MD-001—a keratolytic ointment—has been developed for the treatment of meibomian gland dysfunction and evaporative dry-eye disease. A multicenter, double-masked, vehicle-controlled, parallel group, Phase IIb trial evaluated the safety and efficacy of AZR-MD-001 in 245 patients with meibomian gland dysfunction.
Patients administered AZR-MD-001 twice weekly directly to the meibomian glands in the lower eyelid at bedtime. Co-primary efficacy endpoints included the number of glands secreting meibum and patient-reported symptoms.
Azura Ophthalmics reported that AZR-MD-001 0.5% achieved statistically significant differences in both signs and symptoms at month three when compared to vehicle. Data also showed significant improvements in MGYLS and OSDI scores.
The company plans to initiate a second pivotal multicenter clinical trial of AZR-MD-001 0.5%.
GLK-301 (Glaukos)
This investigational drug candidate uses Glaukos’ iLution platform’s cream-based drug formulations, which are applied to the outer surface of the lid for dropless transdermal delivery of pharmaceutically active compounds. The active ingredient in GLK-301 is pilocarpine.
The company recently announced topline results from its Phase IIa, first-in-human clinical trial. This multicenter, randomized, double-masked, placebo-controlled study enrolled 218 dry-eye patients to examine the safety and efficacy of three different dose levels of GLK-301.
GLK-301 demonstrated an improvement in tear breakup time and a corresponding reduction in blurred vision. Based on these Phase IIa outcomes, Glaukos announced plans to advance GLK-301 into a Phase IIb clinical trial.
ST-100 (Stuart Therapeutics)
ST-100 is an eyedrop formulation based on Stuart’s Collagen Mimetic Peptide platform, PolyCol. In 2022, the company released findings from its Phase II study which included 160 dry-eye patients who received either 20 µg/ml or 50 µg/ml of ST-100 twice daily, or placebo.
Findings showed that the drug met the Schirmer’s Responder Rate endpoint at 28 days. This was defined as a statistically significant difference in the percentage of patients achieving a 10 mm or greater increase in Schirmer’s test scores.
The company also reported that the agent demonstrated significant results in several symptoms as well as ocular surface staining scores as early as treatment day seven. ST-100 was well tolerated among study participants.
In July 2022, Stuart Therapeutics announced that the FDA accepted the company’s plan to conduct a Phase III trial for ST-100 and approved the proposed endpoints—a single primary endpoint (Schirmer’s Responder Rate) and a series of secondary endpoints.
Tanfanercept (Harbour BioMed)
A TNF-α inhibitor, tanfanercept (HBM9036) is under investigation as a treatment for moderate-to-severe dry eye. Harbour BioMed completed its first interim analysis from their Phase III clinical trial. The trial plans to enroll 674 patients and two interim analyses will be conducted.
As of December 28, 2021, 187 patients had been evaluated for key efficacy endpoints assessment, according to the company. Interim data was reviewed by the Independent Data Monitoring Committee. They observed an efficacy trend and favorable safety, recommending that the study continue according to the current protocol.
SURF-100 and SURF-200 (Surface Ophthalmics)
Two therapeutic drops for dry eye are under development using Surface Ophthalmics’ Klarity diluent as the vehicle. SURF-100 (mycophenolate sodium and betamethasone sodium phosphate) targets chronic dry-eye disease while SURF-200 (betamethasone) is designed to treat acute/episodic dry eye. Phase II clinical trials are currently underway for both products. As of May 2021, the study for SURF-100 reached 50-percent enrollment. It consists of multiple arms and will enroll approximately 300 patients. SURF-200 is being studied in two different low-concentration formulations. The trial will enroll between 120 and 140 patients, and the first patient was dosed in early 2021.
Oxervate (Dompe)
A topical solution 0.002% of cenegermin-bkbj, Oxervate is approved for the treatment of neurotrophic keratitis. Studies are currently underway to evaluate the use of this agent in patients with severe Sjögren’s-related dry-eye disease.
The NGF0121 study (NCT05133180), which enrolled 104 subjects, aims to assess the efficacy and safety of Oxervate in this patient population. The co-primary endpoints are Schirmer’s test and the Symptom Assessment Questionnaire in Dry Eye (SANDE) questionnaire.
The NGF0221 study (NCT05136170) enrolled 85 patients who are currently being treated for severe Sjögren’s-related dry-eye disease with topical cyclosporine A. The primary endpoint is Schirmer’s test and change from baseline in SANDE global score.
With a plethora of new agents in the pipeline, the potential implications for dry-eye management are significant. “Approaching dry-eye disease can be difficult because there are so many factors involved,” notes Dr. Latkany. “However, we continue to learn and there are a lot of new things brewing, which I’m very excited about."
Dr. Latkany and Dr. Akpek report no relevant financial disclosures.
1. Akpek EK, Wirta DL, Downing JE, et al. Efficacy and safety of a water-free topical cyclosporine, 0.1%, solution for the treatment of moderate to severe dry eye disease: The ESSENCE-2 randomized clinical trial. JAMA Ophthalmol. Published online April 6, 2023. doi:10.1001/jamaophthalmol.2023.0709.
2. Tauber J, Berdy GJ, Wirta DL, et al. NOV03 for dry eye disease associated with meibomian gland dysfunction: Results of the randomized Phase 3 GOBI study. Ophthalmology 2023;130:5:516-524.
3. Wirta DL, Senchyna M, Lewis AE, et al. A randomized, vehicle-controlled, Phase 2b study of two concentrations of the TRPM8 receptor agonist AR-15512 in the treatment of dry eye disease (COMET-1). Ocul Surf 2022;26:166-173.
4. Tauber J, Laurie GW, Parsons EC, Lacripep Study Group, et al. Lacripep for the treatment of primary Sjögren’s-associated ocular surface disease: Results of the first-in-human study. Cornea 2022 Aug 3;10.1097/ICO.0000000000003091.Online ahead of print.