 |
|
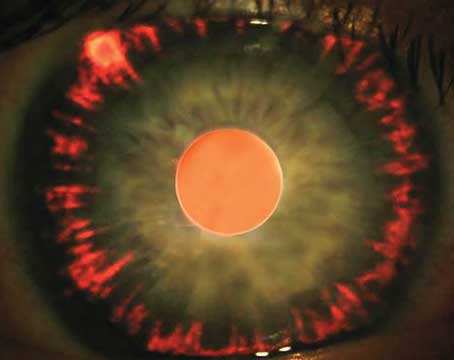
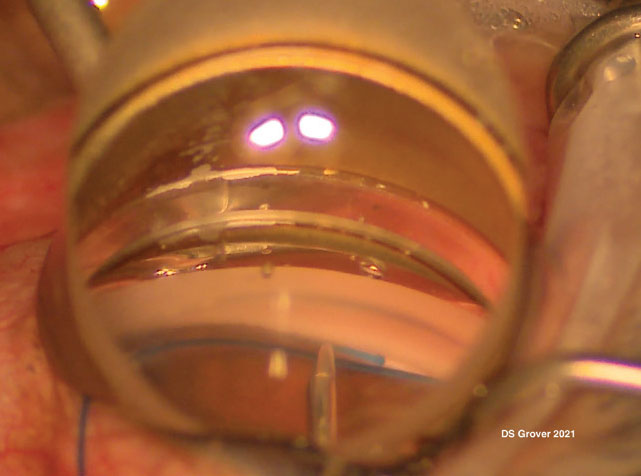
A 5-0 prolene-suture GATT. The blunted-tip suture has passed 360 degrees around the canal and will soon be retrieved and externalized to create a circumferential 360-degree ab interno trabeculotomy. All images courtesy of Davinder S. Grover, MD. |
With the popularity of minimally invasive glaucoma surgeries continuing to increase—and new MIGS options in the pipeline—more surgeons are adding one or two MIGS procedures to their toolbox every year. Here, surgeons with extensive experience performing these surgeries answer questions often asked by cataract surgeons thinking about going down this path.
1. Why offer MIGS?
Obviously a key reason to offer MIGS to an appropriate patient is the ability to lower IOP further than cataract surgery alone. However, surgeons point out several other advantages.
Vikas Chopra, MD, medical director of the Doheny Eye Centers Pasadena and a professor of ophthalmology at the David Geffen School of Medicine at UCLA, believes that every general ophthalmologist should have one or more MIGS in their armamentarium. “The majority of cataract surgeries in the United States are performed by comprehensive ophthalmologists, and many of their patients have mild to moderate glaucoma—the ideal population for MIGS,” he notes. “One of the main advantages of MIGS is reduction in medication burden. This greatly enhances a patient’s quality of life, and it’s well-documented that adherence with glaucoma medications is a major issue, with consequences that can include glaucoma progression. In fact, we’re increasingly seeing patients who’ve done research on the internet and want specific MIGS procedures done in conjunction with their cataract surgery.”
Davinder S. Grover, MD, MPH, an attending surgeon and clinician at Glaucoma Associates of Texas in Dallas, points out that in addition to having smaller pupils and possibly some zonular weakness, patients who have both cataract and glaucoma are at a slightly greater risk of having a postop pressure spike. “In addition to lowering the patient’s overall pressure and decreasing their dependence on drops, most MIGS procedures help to mitigate the potential for that postop pressure spike,” he notes.
What about the increased surgical risk associated with adding a second procedure to cataract surgery? “Any time you add an additional step to surgery there’s a very small chance of something going wrong,” notes Arsham Sheybani, MD, an associate professor of ophthalmology and visual sciences at the School of Medicine at Washington University in St. Louis. “However, in the hands of an experienced surgeon, the risk of a minor complication is probably less than 5 percent. In terms of something serious like a cyclodialysis cleft, I’d say the risk is less than 1 percent. If the patient and the doctor decide that adding MIGS isn’t worth it—maybe the doctor doesn’t feel comfortable doing MIGS—then so be it. But I do think anyone doing cataract surgery on a patient who has glaucoma or is on glaucoma medications needs to inform the patient that these options exist.”
Dr. Sheybani points out that with such a low risk associated with adding MIGS to cataract surgery, and potential upsides like reducing the need for medications and reducing the likelihood of needing further surgery in the future, doctors who aren’t already offering MIGS should be considering it. “It’s certainly something that any cataract surgeon should be able to master,” he notes. “I’ve worked with residents and fellows who’ve only done 10 or 20 cataract surgeries, and they get the hang of working in the angle despite much less phaco experience than a comprehensive ophthalmologist.”
“Incorporating a MIGS procedure doesn’t add significantly to the risks of the surgery, but it adds a lot of potential benefits,” Dr. Grover concludes. “After all, you’re already in the eye to remove the cataract. You could argue that if a patient only has very mild glaucoma, the cataract surgery alone might be sufficient; the iStent and Hydrus trials showed convincingly that cataract surgery does lower IOP a little bit. But I’d say that if a patient has mild to moderate glaucoma and is using multiple drops, it’s a disservice to the patient not to do something more to address the glaucoma.”
2. What skills will I need as a MIGS surgeon?
“If you’re thinking about adding one of the MIGS angle surgeries to your armamentarium, the first thing you need to do is understand angle anatomy,” notes Dr. Grover. “Also, you need to be familiar with gonioscopy—not just clinical gonioscopy, but intraoperative gonioscopy.”
“One of the best ways to get started is to do gonioscopy in clinic,” says Dr. Sheybani. “A lot of us who aren’t glaucoma specialists aren’t doing gonioscopy very often. As a result, we’re not accustomed to visualizing the angle structures; sometimes the zones blend together. So the first big benefit of doing gonioscopy in clinic is that you’ll start building your base understanding of what’s normal and what’s not. Once you get accustomed to doing gonioscopy in clinic, the next step is to start doing gonioscopy in the OR during some of your routine cases.”
“After you put the lens in the bag, before you wash out any viscoelastic, tilt the head and scope and put a gonioprism on the eye and look at the angle anatomy,” suggests Dr. Grover. “Make sure you can identify all of the landmarks. Then, put an instrument into the eye. Note the tactile feedback, so you understand what it feels like to have a tool working in the angle. This can take a little while to get used to.”
“For many of the MIGS procedures, mastery of intraoperative gonioscopy is critical for success,” agrees Dr. Chopra. “It’s important to practice getting the best possible view of the angle. Practicing will help you in multiple ways. First, you’ll learn to use the goniolens in your nondominant hand while managing the angle-based surgery with your dominant hand. Among other things, this will help you avoid creating corneal folds due to compression. Second, you’ll learn how much to tilt the patient’s head, versus how much to tilt the operating microscope.
“Third,” he continues, “you’ll get accustomed to filling the anterior chamber with the right amount of OVD. Overfilling can compress Schlemm’s canal, making it difficult to cannulate or surgically open during a trabecular bypass procedure, while underfilling can allow the iris to bow forward and obscure a clear view of the iridocorneal angle. Fourth, you’ll learn to avoid nicking the perilimbal vessels, which can cause bleeding and clouding up of the corneal tear film, making the view through the gonioprism challenging.”
Dr. Chopra adds that it’s important to do your homework. “I’d strongly encourage ophthalmologists considering adding a MIGS surgery to their repertoire to watch surgical videos of the procedure to help learn the technique,” he says. “Work with industry reps to do personalized wet labs, and sign up for surgical wet-lab training at meetings like ASCRS and AAO. Talk to colleagues and glaucoma specialists to pick up learning tips and surgical pearls. Perhaps most important, to have a successful procedure you need to understand the surgical anatomy—especially in the iridocorneal angle. That’s why achieving expertise in clinical and surgical gonioscopy is essential.”
3. Which MIGS option should I learn first?
 |
|
A goniotomy with the KDB Glide. Note the white stripe on the left side of the angle, demonstrating the treated angle structures with the exposed back wall of Schlemm’s canal visible. |
Dr. Grover says that once you’ve grown accustomed to the basic angle maneuvers and refreshed your familiarity with angle anatomy, you can try some type of angle surgery. “Pick one MIGS surgery and work with it; get comfortable with the technique,” he suggests. “Become familiar with the way it changes things at the postop follow-up visit. Options with a slightly easier skill set would include a goniotomy or an iStent. Goniotomy is easy to start with; once you understand angle anatomy it’s relatively straightforward and doesn’t require a huge amount of manipulation in the eye.
“At first, I’d start with milder cases of glaucoma where you don’t need a tremendous amount of pressure-lowering,” he continues. “In those cases a goniotomy or iStent will be of benefit, and those procedures are relatively safe. Once you’re comfortable with those, you can talk about trying options like the Hydrus, or performing a GATT. The second MIGS option you choose should be based on the type of patients you generally treat, and how much pressure lowering is likely to be required.”
“What you offer,” Dr. Chopra adds, “should be based on your comfort level with learning the procedure, understanding how to manage its intraoperative and postoperative complications, and whether you have enough surgical volume to improve your experience beyond the learning-curve phase.”
“I’m biased toward performing stenting procedures in general,” says Dr. Sheybani. “However, I think if you’re trying MIGS for the first time, doing something that doesn’t leave a device in the eye might be a good way to go; that way, if you make an error you won’t leave something permanent in the eye in the wrong spot. That’s why I think goniotomy is a good way to start learning how to work in the angle.”
Dr. Grover adds that most of the MIGS procedures aren’t that hard to learn. “I do all of the MIGS procedures, and I didn’t learn any of them in fellowship,” he says. “I learned by doing, watching, teaching and collaborating with my partners. I never stop learning and trying new techniques in the OR. I find that those who stop learning in the OR have a more difficult time incorporating newer techniques and gaining new knowledge.”
4. Should I offer more than one MIGS procedure?
“Having the ability to offer at least two different approaches—even if they’re both angle-based MIGS—is important,” says Dr. Chopra. “That makes it possible to customize surgery for each particular situation.”
Dr. Sheybani agrees that it’s good to offer more than one MIGS procedure, but notes that there’s no need to offer them all. “If you’re trying to treat a wide array of patients, it’s good to become proficient at one stenting option, because that will give you the ability to treat patients who might, for example, not be able to stop taking blood-thinning medications,” he explains. “Then, it’s good to have a stripping or cutting option, just because that gives you the ability to treat a wider angle. It also means you’ll have a stand-alone option that can be done separately from cataract surgery—with the caveat that if you’re doing stand-alone MIGS, you probably need to get proficient at treating at least 180 degrees of the angle. The bottom line is, if you master one stripping option and one stenting option, you’ll be able to cover a wide array of patients.
“Generally,” Dr. Sheybani concludes, “if you’re looking for safety and the patient is already controlled on medications, the disease is stable, the optic nerve and visual fields aren’t getting worse and the patient needs cataract surgery, the stenting options are the way to go. If you’re looking to reduce the number of medications or achieve significant pressure reduction at the time of cataract surgery, then you should really start considering either a larger stent or a larger stripping procedure.”
5. What about options that don’t involve angle surgery?
The MIGS angle surgeries are clearly the mainstay MIGS procedures for many surgeons, but there are other procedures that some surgeons (though not all) consider to be MIGS procedures. Three of the most notable are Allergan’s XEN gel stent, a flexible ab interno collagen implant draining aqueous fluid into the subconjunctival space through a scleral channel; endoscopic cyclophotocoagulation (ECP), which uses a laser endoscope to visualize and ablate the ciliary body epithelium; and noninvasive micropulse transscleral cyclophotocoagulation.
“Options like the XEN are for surgeons treating more moderate-to-advanced glaucoma,” notes Dr. Grover. “I actually consider XEN to be in a different category. It involves an entirely different skill set.”
Dr. Sheybani says he also doesn’t think of XEN as being a true MIGS procedure. “It’s kind of a hybrid between MIGS and traditional surgeries,” he says. “It’s also kind of a hybrid between a tube shunt and trabeculectomy surgery. You could call it a MIBS—minimally invasive bleb surgery. It can potentially get your pressure down a bit lower, so there’s a broader patient population that can be treated. But it’s not a MIGS surgery that uses the physiologic outflow pathway.”
Dr. Grover says that he doesn’t perform ECP very often, but sees it as a reasonable option in some situations. “ECP involves a different skill set, although it doesn’t have a huge learning curve,” he notes. “Mostly, I avoid using ECP as the sole treatment. The eye has a faucet and a drain; it makes aqueous and drains aqueous. If your drain is clogged and you’re not doing anything to enhance outflow, the sink is going to overflow whether the faucet is turned on completely or halfway. Typically, I find that ECP works for me when outflow is already established, or doing ECP in combination with an outflow-enhancing procedure. I don’t believe ECP done by itself provides a significant benefit in most cases, unless you have an intact outflow pathway.
“Some surgeons do combine an angle surgery with ECP—a combination inflow and outflow procedure,” he notes. “I don’t do that, but I don’t think there’s anything wrong with it, per se. The reason I choose to avoid it is that ECP increases the rate of inflammation, and we don’t really know how inflammation affects wound healing in the angle. However, some surgeons do combine angle surgery and ECP, and they report relatively good results.”
Dr. Sheybani says that, like the XEN, he doesn’t think of ECP as a true MIGS procedure. “Many of us use it more in angle closure and plateau iris cases,” he explains. “A patient with a functioning tube shunt who needs a little bit more pressure reduction could be another good choice. And I do appreciate the endoscopic aspect for viewing structures during anterior segment surgeries if there’s a lens dislocation, or if the patient has a problem that requires viewing the ciliary body. But it’s a totally different ball game from most MIGS as far as the structure you’re treating.”
Dr. Chopra says his experience with micropulse transscleral cyclophotocoagulation has been positive. “This procedure has been quite effective and well-tolerated in patients for whom a non-incisional surgery makes sense, either as a primary or adjunctive procedure,” he explains.
Dr. Grover believes that micropulse technology has a lot of potential, but is still somewhat problematic. “The issue is that people are still trying to figure out the settings that will produce the most predictable outcomes,” he explains. “I don’t think this is the fault of the technology itself; we need to better understand the parameters that are needed to treat different patients. Many groups, including ours, are working on this.”
6. What about the suprachoroidal space?
 |
|
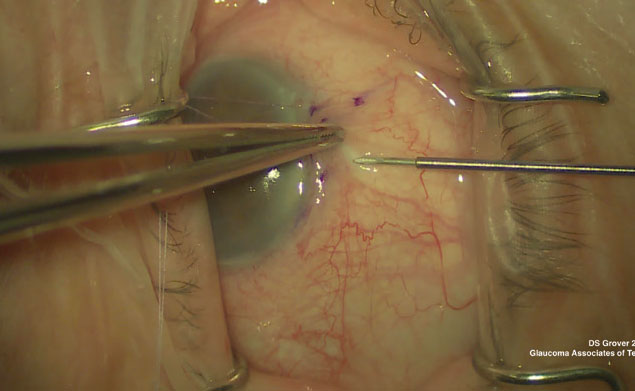
Intraoperative photo demonstrating the ab externo closed conjunctival XEN implantation technique. |
Shunting aqueous into the suprachoroidal space is another approach to lowering pressure that’s shown promise. Surgeons briefly had access to the CyPass stent, designed to do this, but it was withdrawn from the market after some patients experienced corneal endothelial cell loss. However, new options designed to tap into the suprachoroidal space, including the iStent Supra, are now in the pipeline.
Dr. Grover believes that such an option could be very useful. “It’s probably not necessary for patients with mild glaucoma, but it could be essential for patients with refractory glaucoma and a lot of scarred conjunctiva,” he says. “I believe the concern here is the risk-benefit ratio. I suspect that the CyPass ended up being withdrawn because they were targeting patients with mild glaucoma who were not likely to go blind, and the risks—assuming that the endothelial cell loss was actually real—turned out to be too great for the benefits a mild glaucoma patient might gain. On the other hand, if a patient has serious disease and will go blind if their glaucoma isn’t addressed, then considering a slightly more invasive procedure that might cause mild endothelial cell damage is potentially justifiable.
“I think being able to access the suprachoroidal space will be a very valuable means of treatment once we have an approved device,” he concludes. “This approach has a lot of potential, and I think it will be one of the next frontiers.”
Dr. Sheybani’s outlook is a little more cautious. “I believe a shunt into the suprachoroidal space could be a good option for a refractory patient,” he says. “But until we figure out how to prevent scarring that leads to sudden failure, how to prevent the hypotony that rarely occurs, and how to prevent the effusion that can cause shallowing of the anterior chamber, it’s not something I’d add to cataract surgery for a patient with mild disease. I think it would be reasonable to consider for a patient who might need something more than a trabecular-based surgery, someone who isn’t a good candidate for subconjunctival surgery. And it might be good for someone who has failed prior procedures. But given the traditional view of cataract surgery plus a MIGS procedure to address mild glaucoma, I wouldn’t jump to the suprachoroidal space, at least using the current technologies that are coming down the pike.”
7. Which MIGS should I offer to which patients?
Although he acknowledges that a choice of MIGS procedure should be customized for each individual patient, Dr. Chopra believes it’s a good idea to try to enhance the patient’s own outflow system before trying to bypass it. “I tend to recommend trabecular meshwork bypass procedures as first-line, especially in patients with mild to moderate glaucoma and/or a desire to reduce their topical medication burden,” he says.
“For other patients with more advanced disease who may require a very low intraocular pressure, I routinely offer the XEN gel stent,” he continues. “The XEN generally achieves low IOPs approaching those achieved with traditional glaucoma procedures like trabeculectomy or tube shunts—although it may leave the patient with a greater medication burden [than those procedures].
“Occasionally you may encounter an older, monocular patient who may not be a good surgical candidate due to systemic or ocular health issues,” he adds. “In that case it would make sense to consider starting with a non-incisional procedure like micropulse transscleral cyclophotocoagulation.”
Dr. Grover notes that when combining MIGS with cataract surgery, the characteristics of the cataract shouldn’t make too much difference in terms of which MIGS you choose. “In general, the angle anatomy needs to be normal, appropriate and visible,” he says. “If a patient has a very small eye or narrow angle, that can complicate things.”
8. Which patients shouldn’t be offered MIGS?
Dr. Sheybani says there are cases in which he might not add a MIGS procedure to cataract surgery. “If the patient is an ocular hypertensive with a 0.4 disc, a TMax pressure of 22 or 23 mmHg and no disc or field changes—but someone has started them on a prostaglandin—I’d say cataract surgery alone for that patient is perfectly fine,” he explains. “But in that case I’d wonder whether the patient really needs the prostaglandin.
“The data from the Ocular Hypertension Treatment Study suggests that when there’s low suspicion of glaucoma but elevated IOP, the end result is the same even if you wait to start medications,” he continues. “If the patient may not really need the drop, then you probably don’t need a MIGS procedure either. But if the patient has disc changes and is on a medication, even if the pressure is controlled, then a MIGS procedure is very reasonable. Or if you think that someone with a healthy disc is at high risk of progressing, that’s another good MIGS candidate.”
Dr. Chopra agrees that not every patient should have a MIGS procedure. “The decision about adding MIGS has to be individualized, but it should certainly be dictated by the patient’s level of glaucoma, as well as the patient’s ability to tolerate topical anti-glaucoma medications,” he says. “In any case, proper preoperative screening with in-office gonioscopy, and a detailed ocular exam to determine feasibility of surgery based on ocular anatomy, are essential for properly matching a particular patient to a specific procedure.
“A patient shouldn’t have MIGS just because it’s the newest addendum to cataract surgery,” he adds. “In the randomized, controlled trials for FDA approval of all the trabecular bypass procedures, cataract surgery without MIGS was also effective in achieving lower intraocular pressures in a majority of patients. So if the patient isn’t an appropriate candidate for a MIGS procedure, or the ophthalmologist isn’t comfortable with a procedure, the patient should just undergo the cataract surgery.”
9. What should be done before a MIGS surgery?
Surgeons offer these suggestions:
• Make sure MIGS is mentioned in your pre-cataract-surgery informed consent. “When you’re doing cataract surgery on a patient with a diagnosis of glaucoma, even if the pressure is controlled—especially if the patient is on medication treatment—MIGS should at least be mentioned in the informed consent,” says Dr. Sheybani. “The patient should be made aware that there are procedures with a favorable safety profile that can be done at the same time as cataract surgery that could potentially reduce the need for medications after surgery and decrease the need for later glaucoma surgery. You have to tell the patient about all the major options that exist—including toric lenses and multifocals—whether you offer them or not. Then, it’s up to you and the patient to decide whether to add a MIGS procedure.”
• Don’t offer a procedure you seldom perform. “Ophthalmologists have an ethical obligation to offer only those MIGS procedures for which they’re ‘certified,’ ones they’re comfortable performing,” says Dr. Chopra. “If you’re only performing these procedures once in a while, it’ll be challenging to develop both a skill set that allows successful surgery and an understanding of how to manage intraoperative and postoperative complications.”
• Check the pigmentation in the angle before surgery. Dr. Sheybani points out that one of the few ways you can get into trouble with MIGS angle procedures is if you’re working at the ciliary body band and you’re counting on pigmentation to guide you. “In some angles, the pigmentation is very light,” he points out. “If that’s the case, it’s possible to misidentify the angle structures and create a cleft; that could lead to a lot of bleeding and possibly hypotony. It’s rare for that to happen, but that’s the big danger.
“This is one reason you need to examine the angle before surgery,” he says. “If the pigmentation is really light, Trypan blue can be used to stain the trabecular meshwork nicely and give you clear landmarks to guide you. If you’re considering doing a MIGS procedure and the pigmentation is very light, write a note in the chart to remind yourself to use Trypan.”
• Avoid MIGS if inflammation is present. “If the eye is calm and quiet with a history of uveitis, sometimes MIGS works well,” notes Dr. Grover. “But if there’s active inflammation or neovascular glaucoma, I’d categorically avoid MIGS.”
10. What can I do to help ensure better outcomes?
These strategies can help ensure optimal MIGS results:
• Avoid pushing down with the gonioprism. Dr. Sheybani says this is mostly an issue with surgeons who’ve done very little gonioscopy—especially residents and fellows. “When you’re holding the gonioprism in your hand, there’s a tendency to want to push down,” he explains. “That can cause a lot of striae. The gonioprism should be coming on and off the cornea; if it has a light-touch meniscus that comes and goes, then you know you’re doing it right.”
• Don’t push into the angle too hard during goniotomy. “Many surgeons do this,” says Dr. Sheybani. “Pushing too hard can create striae, and you can hit the back wall. You need to use a light touch. You should think of it as kind of working back toward yourself. I had a fellow who said, ‘It’s almost like you’re scooping things out of the canal.’ That’s a nice way to describe it. You really shouldn’t be pushing in.”
• Early in the surgery, don’t have the patient look left or right while you’re hovering in the angle with your device. “If the patient looks the wrong way, you could hit tissue and cause a problem,” Dr. Sheybani explains.
• During surgery, make sure the patient’s head is turned far enough away from you. “Surgeons sometimes complain they couldn’t get a good view of the angle,” says Dr. Sheybani. “The number one reason is that they didn’t turn the head far enough away from them.”
Dr. Sheybani has been a consultant for Allergan, Alcon, Santen, Katena and Ivantis. Dr. Grover is a consultant for Allergan, New World Medical, Olleyes, Reichert and Santen. Dr. Chopra reports no relevant financial ties to anything discussed in this article.