The Scope of the Problem
Netherlands ophthalmologist Frank Kerkhoff and his colleagues found the incidence of rhegmatogenous retinal detachment in patients with uveitis to be approximately 3 percent.1 This study included patients with viral retinitis, however, which, like syphilitic uveitis, has a relatively high rate of detachment.2 However, the
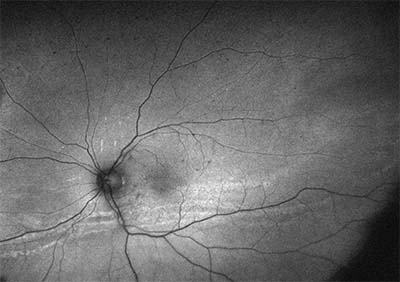 |
| Figure 1. Fundus autofluorescence of the left eye of a patient with chronic panuveitis showing a low-lying retinal detachment with multiple demarcation lines. |
Is It Rhegmatogenous?
Although this might appear to be a simple question to answer, patients with uveitis, especially with intermediate and posterior uveitis, often develop multiple pinpoint retinal breaks due to peripheral vitreoretinal traction. When these occur in association with cataract and vitreous haze or debris, visualization of small breaks can be difficult. Assessment of the location of the detachment is helpful in searching for retinal breaks, as is careful examination with a three-mirror lens. Retinal detachments with small breaks in these patients are often inferior and low-lying, and may demonstrate multiple demarcation lines (See Figures 1 and 2).
The presence of shifting fluid can indicate that the detachment isn’t rhegmatogenous, but this can be misleading if the fluid is longstanding and viscous. In general, non-rhegmatogenous retinal detachment is much less common when the uveitis is controlled. Hence, a new detachment is most likely to be rhegmatogenous in a patient on chronic therapy for intermediate or posterior uveitis that’s not currently active. A trial of one to two weeks of prednisone 1 mg/kg orally can be helpful to assess whether the fluid improves, with improvement suggesting a serous component to the detachment. This is also useful in getting the uveitis quieter in preparation for surgery. In patients with persistent fluid from serous non-rhegmatogenous detachment, surgical intervention with scleral buckling, vitrectomy and drainage of subretinal fluid can still result in resolution of the detachment.3
Perioperative Management
Even if a patient has a history of noninfectious uveitis, no recent activity and is not on chronic immunosuppression, it’s still advisable to treat him with at least 30 mg oral prednisone daily for a few days prior to surgery. The dose can be adjusted based on the severity of the previous inflammation as well as accompanying conditions such as diabetes. This can be continued postoperatively for a few days, then tapered. Intraoperatively, administer 125 mg of intravenous methyl-prednisolone. If the patient isn’t a known intraocular pressure steroid responder, 20 to 40 mg of sub-Tenon’s triamcinolone can be given at the end of the surgery, which will last for several months. Of course, the risk of steroid-induced ocular hypertension must be weighed against the risk of increased postoperative inflammation. The same regimen can be applied to patients with chronic inflammation on chronic immunosuppressive therapy who don’t currently have inflammation.
It’s been suggested that an eye should be quiet for at least three months prior to cataract surgery. However, we can’t wait three months if there’s a retinal detachment. Patients with uveitis and RRD have active inflammation 46 percent of the time.1 If there’s significant active inflammation preoperatively, a serious attempt to control the inflammation with systemic and periocular corticosteroids should be made. This puts the clinician in a difficult position: Surgery may need to be delayed for a few weeks to avoid operating on an inflamed eye, but this risks progression of the retinal detachment. Both choices increase the risk of PVR and the risk of a poor visual outcome. Eyes with uveitis often have pre-existing visual compromise and this also needs to be considered when choosing to delay surgery.
In the case of infectious uveitis and RRD, control of the infection is a high priority, but the post-infection inflammation may persist for weeks after the infection has resolved. In addition to the human herpes viruses, toxoplasmosis and syphilis are associated with RRD. The latter two organisms, however, are less likely to be associated with the widespread retinal necrosis that follows infection by herpes-class viruses, and thus have a lower risk of RRD.
Surgical Options
Although vitrectomy and laser, combined with scleral buckling, will usually be the preferred surgical procedure, ancillary issues may complicate the overall surgical plan. Corneal and lenticular opacities often make visualization difficult. If a significant cataract is present, the choice of placing an intraocular lens versus performing a lensectomy warrants careful consideration. This is further complicated if there are significant synechiae. If the eye is relatively quiet, a simultaneous lens implant is often the preferred choice, as lensectomy precludes the future use of an intravitreal dexamethasone implant. However, visualization is paramount, and a lensectomy is an acceptable choice, especially in an eye with residual active inflammation.
For pinpoint breaks in the retina that can’t be visualized intraoperatively, or through which subretinal fluid cannot be drained due to the small size of the retinal break, fluid should be drained through a drainage retinotomy. Endolaser should be applied around the retinotomy site, as well as in the area of the retinal detachment to cover any tiny breaks that may be missed in the periphery.
The choice of retinal tamponade is influenced by several factors. Silicone oil requires a second surgery for removal and will interfere with the function of a fluocinolone implant, if present. The use of intravitreal dexamethasone may also be suboptimal; it may not release drug properly if it remains initially within the silicone oil bubble rather than within the liquid vitreous. Intravitreal triamcinolone is difficult to use with silicone oil since it can obscure the postoperative view. In addition, the concern about the oil interface acting as a matrix for inflammatory membranes is an unanswered question. In patients with postviral retinal detachments, silicone oil is the preferred tamponade. In other patients, intraocular gas may be a better choice if the eye can be repaired in a single operation and there is no preoperative PVR.
Tractional components of retinal detachment, especially inferiorly in patients with pars planitis, need to be addressed by both scleral buckling and vitrectomy. This part of the surgery is complicated, since patients with RRD and uveitis are younger, may not have a
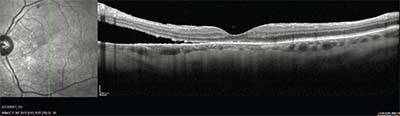 |
| Figure 2. Spectral domain optical coherence tomography from the patient in Figure 1, showing inferior parafoveal retinal detachment. |
Some patients with uveitis also have an associated scleritis. The scleral thinning makes scleral buckle placement difficult, if not impossible. As a result, the surgeon is often forced to alter the surgical plan to accommodate areas of scleral thinning that limit the placement of trocars.
Postoperative Inflammation
There are no systemic immunosuppressants that become fully effective within a few weeks, which means that the initial management of postoperative inflammation involves corticosteroids. Maximal use of oral, periocular, intravitreal and topical steroids may be required, as inflammatory mediators will increase the likelihood of PVR. Local steroid use such as the dexamethasone implant hasn’t been shown to improve the prognosis of PVR; however, PVR will be accelerated by the presence of inflammatory cytokines. If the patient’s uveitis wasn’t adequately controlled preoperatively, it’s important to initiate a long-term plan for uveitic control. This is especially important if additional surgeries are likely. In patients with infectious uveitis such as toxoplasmosis or cytomegalovirus, however, corticosteroids can lead to recurrent infection, which will further complicate the management of the inflammation. In these patients, it is essential to use sufficient antimicrobial agents throughout the postoperative course.
In summary, the management of rhegmatogenous retinal detachment in eyes with intraocular inflammation requires control of the inflammation before, during and after surgery, as well as understanding that the postoperative course and outcomes will be strongly influenced by the associated inflammatory process.
Dr. Palestine is a professor of ophthalmology, and Drs. Pecen and Siringo are assistant professors of ophthalmology, at the University of Colorado School of Medicine in Aurora. Dr. Palestine may be contacted at alan.palestine@ucdenver.edu.
1. Kerkhoff FT, Lamberts QJ, van den Biesen PR, Rothova A. Rhegmatogenous retinal detachment and uveitis. Ophthalmology 2003;110:2:427-31.
2. Haug SJ, Takakura A, Jumper JM, Heiden D, McDonald HR, Johnson RN, Fu AD, Lujan BJ, Cunningham ET Jr. Rhegmatogenous retinal detachment in patients with acute syphilitic panuveitis. Ocul Immunol Inflamm 2016;24:1:69-763.
3. Galor A, Lowder CY, Kaiser PK, Perez VL, Sears JE. Surgical drainage of chronic serous retinal detachment associated with uveitis. Retina 2008;28:2:282-8.



