|
"This is regeneration or replacement of cells, so it's not a prosthesis. And even after four years, the regeneration is still ongoing."
–Professor May Griffith |
Much of the coverage of research in cornea in recent years has
centered on developing alternatives to penetrating keratoplasty. Despite
the success of PK in curing corneal blindness and the widespread
adoption of less invasive, partial thickness procedures such as DSEK,
DSAEK and DMEK, a major challenge remains—the shortage of donor corneas.
Additionally, the success rates of transplant procedures drop off
significantly in the developing world. And any donor tissue brings with
it attendant challenges of dealing with immunological issues.
An international group of researchers centered in Sweden now
has four years of follow-up in its efforts to develop recombinant, human
collagen corneas.
Centered at Linköping
University in Linköping, Sweden, the group is taking a regenerative
medicine approach to repairing diseased corneas. They are testing
implants made from Type III recombinant human collagen (RHC),
synthesized in yeast and chemically cross-linked, and molded into a
biosynthetic cornea. The cell-free implants induce the patient’s own
epithelial cells to grow over the implant, while stromal cells migrate
into
the implant and anchor it to the eye. In December, they reported the four-year follow-up of their Phase I study.1
Professor May Griffith, of the Integrative Regenerative Medicine Centre, Department of Clinical and Experimental Medicine, at Linköping University is one of the researchers. “This is still a work in progress, but we’ve found some very interesting things,” says Prof. Griffith. “One that was very interesting to us is that we had less inflammation than in donor corneas. We did not have dendritic cells inside the corneas. With donor corneas we found dendritic cells.”
The reported data is on 10 patients (eight male, two female; age 18 to 75 at surgery; nine with keratoconus and one with central scarring). The 10 patients were grafted with the biosynthetic implants by anterior lamellar keratoplasty, with the implants being retained by overlying sutures. A second group of nine patients with similar pathologies (six male, three female; age 40 to 79); with keratoconus (five cases), endothelial decompensation (two), a deep central scar (one) and pseudophakic bullous keratopathy (one) were grafted with human donor allograft corneas by full-thickness penetrating keratoplasty, stabilized with peripherally located running sutures.
Key four-year results included:
• The implants were all well-integrated within the corneas of all 10 patients. The mechanical strength of the implants was significantly lower than that of the average human cornea and the implants were softer, resulting in the need for overlying rather than interrupted sutures for retention.
• By anterior segment optical coherence tomography, the shape, thickness and border areas of the implanted corneas remained constant from year one to year four. Central corneal thickness was not significantly changed from year one to year four in either the biosynthetic implant group or the human cornea transplant group. CCT at four years was 358 ±101 μm in patients with biosynthetic implants; 576 ±50 μm in patients with human donor corneas, and 534 ±30 μm in healthy corneas. The biosynthetic corneas were significantly thinner than healthy corneas, and the human donor corneas were significantly thicker than healthy corneas.
|
• In a key finding, over the four-year postoperative period, no episodes of rejection were observed in the biosynthetic implanted corneas. In this group, the sutures were removed at 6.5 weeks (range: four to seven weeks), and prophylactic immunosuppressive steroids were stopped. The nine patients implanted with donor corneas received steroids for 12 months and sutures were removed after a mean of 13 months; one patient in this group had a rejection, which is in keeping with statistical averages in the first year postop.
Dendritic cells, which regulate the immunogenicity of an organ and determine whether a graft is tolerated or rejected,were present in the center of the human donor corneas but not in the biosynthetic implants, or healthy corneas. Total dendritic cell density in the human donor cornea group was significantly greater than in the biosynthetic group.
• Regeneration. At four years, the regenerated epithelium remained stratified and displayed cell layers that were similar to human donor corneas and healthy controls. The initially cell-free implants were populated by stromal cells that had grown into the implants, but cell-free areas remained. Human donor corneas had fewer stromal cells than healthy corneas, and additionally had small particulate bodies and linear structures indicative of apoptosis; healthy controls had a dense, even distribution of stromal cells. The nerves from the plexus lying under the epithelium in the human donor cornea group had reached the central corneal region to varying degrees. However, they were generally sparse, highly branched and abnormally tortuous. In corneas that received bio-synthetic implants, the regenerated nerves followed straighter, parallel paths as described for healthy corneas, although the nerve fibers in healthy corneas were thicker and more densely packed.
Central corneal touch sensitivity was assessed by contact esthesiometry in the patients’ operated eyes and their contralateral unoperated eyes, which served as controls. Touch sensitivity in corneas implanted with human donor and biosynthetic tissue was significantly reduced relative to unoperated eyes, which consistently exhibited normal touch sensitivity. Touch sensitivity in corneas with cantly better than in human donor corneas.
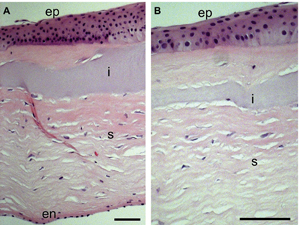
Histology of a regenerated neo-cornea showed a normal, healthy corneal architecture, with a stratified nonkeratinized epithelium, lamellarly arranged stroma and a layer of endothelium. There was a cell-free region in the center of the stroma, which represented the part of the implant that had not yet been remodeled. In areas where the remodeling was more advanced, the implant had blended seamlessly into the stroma. The authors say the histology, coupled with the in vivo confocal images, supports the contention that active regeneration was still ongoing at four years postop.
Distance-corrected visual acuity was achieved in the biosynthetic group by use of custom-fitted hard contact lenses to regularize an uneven corneal surface. Patients tolerated these lenses after surgery but not before. In the human donor group, distance-corrected VA was measured with spectacles. At four years, distance-corrected VA was 20/54 in the biosynthetic group, and 20/36 in the human donor groups. In terms of vision improvement from the preoperative level, the bio-synthetic group had a mean gain of 5.6 Snellen lines, while the human donor group had a mean gain of 9.9 Snellen lines at four years postop.
The next step, says Prof. Griffith, is another trial but with a fortified material. “We have a more robust, reinforced material that we’re trying for higher-risk transplants, such as chemical burns. What we had before was just a single network of recombinant human collagen. It’s now two networks and that makes it significantly stronger.”
It may be some time before this is mainstream, but Prof. Griffith and her coworkers feel its impact could be significant. “This is regeneration or replacement of cells, so it’s not a prosthesis. And even after four years, the regeneration is still ongoing.”
There are other issues to address as well. “The suturability can be improved,” says Prof Griffith. “And we still have the question of whether or not to have an endothelium. Some people have removed the full thickness, some have left Descemet’s. So that’s a question we have not answered.”
REVIEW
1. Fagerholm P, Lagali N, Ong J, Merrett K, et al. Stable corneal regeneration four years after implantation of a cell-free recombinant human collagen scaffold. Biomaterials 2014 Mar;35(8):2420-7. doi: 10.1016/j.biomaterials.2013.11.079. Epub 2013 Dec 25.