Researchers and clinicians continue to break new ground in glaucoma management. This year, ARVO studies have made inroads into nonstandard approaches to detecting glaucoma (and demonstrated the limits of some familiar approaches); tested new approaches to drug delivery, ciliary body coagulation, and nutritional supplementation; provide more data showing the value of combining structure and function to monitor the disease; and provide new insights into risk factors, surgery and selective laser trabeculoplasty.
Detecting Signs
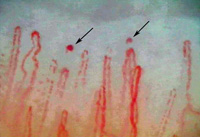
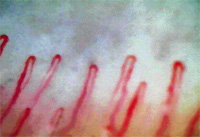
Researchers at two Chicago universities and a clinic in Denver have found that low-tension glaucoma patients have microhemorrhages detectable in capillaroscopic scans of their fingers, seldom found in normal subjects, suggesting the presence of a systemic vascular dysfunction.
| ||||||
The study authors note that the cause of the microhemorrhages remains undetermined. Possible explanatory factors would include capillary growth regulation, breakdown of support matrix, blood flow velocity and neurogenic dysfunction. They conclude that POAG has systemic features characteristic of connective tissue diseases.1881
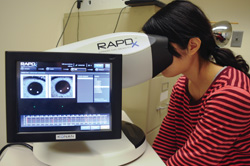
Researchers at Wills Eye Institute and Jefferson Medical College in Philadelphia conducted a study to compare the ability of the RAPDx pupillograph (Konan) and the swinging flashlight method (both with and without magnification) to detect afferent pupillary defects, indicative of asymmetric glaucomatous damage.
|
• Apparent APDs were detected in 15 patients using SFM (26.3 percent); in 35 patients using MA-SFM (61.4 percent); and in 35 patients using the RAPDx device (61.4 percent).
• “Corroborated” APDs were found in nine patients using SFM (15.8 percent); in 21 patients using MA-SFM (36.8 percent); and in 22 patients using RAPDx (38.6 percent).
• Clinically detected asymmetry in disc damage was missed in 28 patients using SFM (49.1 percent); in 12 patients using MA-SFM (21.1 percent); and in 12 patients using the RAPDx (21.1 percent).
The authors conclude that the RAPDx pupillograph and MA-SFM method are both useful for the detection of asymmetric glaucomatous damage, and both detect apparent and “corroborated” APDs more often than the non-magnified SFM.4811
Researchers at the Wilmer Eye Institute and Johns Hopkins University in Baltimore conducted a prospective, case-control study to assess the relationships between the pupillary light reflex, as measured by a pupillometer, and visual field defects and retinal nerve fiber layer thickness. They analyzed pupillary responses to various stimulus patterns in 157 patients with glaucoma (mean age 67 ±11, 50 percent female) and 71 controls (mean age 60 ±10, 69 percent female). They compared the responses between the two eyes and to superonasal and inferonasal stimuli within each eye, and calculated the absolute PLR value for each individual eye. The data showed:
• Glaucoma patients had a more asymmetric pupil response between the two eyes (p<0.001), and between the superonasal and inferonasal fields within the same eye (p=0.014).
• Glaucoma patients had a smaller amplitude, slower velocity and longer latency of pupil response than controls (all p<0.001).
• For every difference of 0.3 log units in between-eye asymmetry of PLR there was an average 2.3-dB difference in visual field mean deviation (R2=0.62, p<0.001) and a 3.7-µm difference in RNFL thickness between the two eyes (R2=0.34, p<0.001).
• Greater visual field damage and a thinner RNFL were associated with a smaller response amplitude, slower velocity and longer time to peak constriction and dilation (all p<0.001 after adjusting for age and gender).
• Asymmetry of PLR between superonasal and inferonasal stimulation within a given eye was not associated with differences in visual field or RVFL in those locations.
The authors conclude that carefully measured pupillary light reflex is strongly correlated with visual field functional testing and measurements of RNFL thickness, indicating that quantitative pupillography may have a role in the diagnosis and management of optic neuropathies.2261
A study conducted at the Prasad Eye Institute in Hyderabad, India, used spectral domain optical coherence tomography to evaluate the optic nerve head, RNFL and ganglion cell complex in 261 eyes of 197 recently referred glaucoma suspects, to see how well current SD-OCT technology and algorithms could diagnose preperimetric glaucoma. (Visual fields were normal in all eyes.)
Two glaucoma experts masked to the patient’s data independently classified the optic nerves into glaucoma and non-glaucoma groups based on digital optic disc photographs. Sixty-eight eyes of 60 patients were classified as glaucomatous; 193 eyes of 137 subjects were classified as controls.
Although age and visual field characteristics were comparable between the groups, all SD-OCT parameters were significantly different. Inferior neuroretinal rim area (area under the receiver operating characteristic curve: 0.708), inferior quadrant RNFL thickness (AUC: 0.713) and global loss volume (AUC: 0.702) had the best AUCs for differentiating preperimetric glaucoma from control eyes. Sensitivities at a fixed specificity of 95 percent for all these parameters were below 27 percent. Likelihood ratios of the outside-normal-limits category of the RNFL and GCC parameters were between 2.2 and 5.9; for the within-normal-limits category they were between 0.6 and 0.8; for the borderline category they were between 1 and 2.
The authors conclude that the ability of the ONH, RNFL and GCC parameters of SD-OCT to diagnose preperimetric glaucoma is poor, indicating a need for improvements in the current analysis algorithms.4828
New Treatment Approaches
Surgeons at nine glaucoma centers in France participated in a prospective, non-comparative interventional clinical study to test the safety and efficacy of a new high-intensity focused ultrasound procedure called Ultrasonic Circular Cyclo Coagulation.
Forty-two eyes of 42 patients with primary open-angle glaucoma were treated. Subjects had an IOP>21 mmHg, an average of 1.65 failed previous surgeries and were using an average of 3.2 medications. Eighteen patients (Group 1) received four-second exposures for each shot; 24 patients (Group 2) received six-second exposures. Complete ophthalmic examinations were performed before the procedure and at one day, one week, and one, two , three, six and 12 months after. Primary outcomes were surgical success (defined as an IOP reduction from baseline ≥20 percent and IOP>5 mmHg) at the last follow-up visit, and vision-threatening complications. Secondary outcomes were the mean IOP at each follow-up visit compared to baseline, medication use, complications and re-interventions.
The data showed that mean IOP was significantly reduced in both groups (p<0.05). Group 1 dropped from 28.6 ±4.7 mmHg to 16.1 ±2.8 mmHg at last follow-up; Group 2 dropped from 28.1 ±8.6 mmHg to 16.7 ±4.4 mmHg. An IOP reduction >20 percent was achieved in 12 of 18 eyes in Group 1 (67 percent) and 17 of 24 eyes in Group 2 (71 percent). Four patients had to be retreated. No major intra- or postoperative complications occurred. The authors conclude that Ultrasonic Circular Cyclo Coagulation appears to be an effective and well-tolerated method to reduce IOP in patients with POAG.4775
Because previous studies had found a decrease of the magnesium content in aqueous humor, anterior sclera and tear fluid of patients with various stages of POAG, a group of researchers in Moscow conducted a study to determine whether a magnesium-containing supplement might have a beneficial effect on IOP, visual fields and retinal nerve fiber layer thickness.
In the study, 22 POAG patients (mean age 61.1 ±2.5 years; r: 42 to 72) received hypotensive therapy supplemented by Magnerot (Wörwag Pharma, Germany) daily for six weeks: two tablespoons three times a day for one week, then one or two tablespoons two to three times a day for five more weeks. As a control, 16 POAG patients (63.7 ±2.7 years) received hypotensive therapy without Magnerot. The group assessed the optic nerve before treatment and at one, two, three and five months post-treatment. In addition to perimetry and retinal tomography, the ORA (Reichert) was used to measure corneal-compensated IOPcc, IOP equivalent to Goldmann (IOPG) and corneal hysteresis.
The treatment group showed almost twice as many improved visual field results as the control group. IOPG dropped by 3.3 ±0.4 mmHg after adding the magnesium supplement; IOPcc dropped by 4.1 ±0.3 mmHg (both p<0.05). Perimetry showed a significant increase in total functional visual field (from 426.5 ±7.8 to 452.5 ±8.8 degrees, p<0.05), mainly in patients with moderate glaucoma, and analysis of the MD index showed reduction in the total depression of sensitivity from an average of -5.8 dB to -3.9 dB (p>0.5). Furthermore, the average thickness of the RNFL tended to grow in patients in the early stages of glaucoma (from 0.25 ±0.02 mm to 0.27 ±0.01 mm) and moderate stage (0.19 ±0.03 mm to 0.21 ±0.02 mm) following treatment.
The authors believe these results suggest that a magnesium-containing supplement may have a stabilizing effect on the course of glaucoma, and recommend further study.759
|
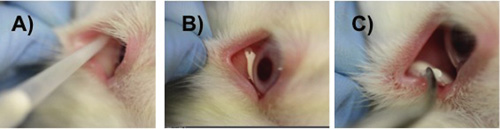
To test the formulation in vivo, a single drop was administered to the inferior fornix of New Zealand white rabbits. IOP was measured periodically and histology was used to assess the biocompatibility of the system. Testing showed that the gel/microparticle drop could be easily administered, and that it formed a stable, opaque gel (See images, above). The gel eye drop was easily removed, leaving no evidence of gel or microparticles. Cytotoxicity testing demonstrated no significant effect on conjunctival cell viability.
The authors believe this new system will provide a simple way for patients to achieve adequate IOP reduction without the issues surrounding injections or compliance with daily eye drop instillation.4294
Structure & Function
Researchers at Bascom Palmer Eye Institute in Miami have demonstrated that in glaucoma suspects, pattern electroretinogram may reveal loss of function years before an equivalent loss is seen in retinal nerve fiber layer thickness loss, as measured by OCT.
Researchers compared progressive losses measured by pattern electroretinogram and those indicated by retinal nerve fiber layer thickness loss over time in 201 eyes of 107 glaucoma suspects. Subjects were monitored with PERG and OCT every six months for at least four years. Longitudinal PERG amplitudes and peripapillary RNFL thicknesses were normalized based on the difference between the values in normal subjects and advanced stages of the disease. The researchers calculated the time required for PERG and OCT to lose 10 percent of their baseline value.
The data showed that in eyes that had an abnormal PERG amplitude at baseline (50 to 90 percent below normal, n=99), pooled PERG amplitude slopes took 1.9 to 2.5 years to lose 10 percent of their initial amplitude. In contrast, measured RNFL thickness took 9.9 to 10.4 years to show the same level of loss (p<0.05). Thus, it appears that it may take eight years longer for retinal nerve fiber layer thickness to reveal this level of loss.796
| |||||||||||||||||||||||||||||
Subjects were followed for an average of 74.8 months; all had normal standard automated perimetry at baseline. Time domain OCT was used to assess RNFL thickness. Data models were adjusted for age, IOP and central corneal thickness. Conversion to glaucoma was defined as repeatable abnormal SAP or progressive optic disc changes during follow-up.
Fifty-eight eyes (16.8 percent) converted to glaucoma. Mean baseline CSFI was 14.1 percent in those who developed glaucoma, and 0.2 percent in those who did not. In the multivariable model, higher CSFI was predictive of conversion (adjusted hazard ratio=2.08 per 10 percent higher; 95 percent CI: 1.70 to 2.56; p<0.001). The model that included CSFI had better predictive ability than models including standard visual field parameters and OCT average RNFL thickness. (See table, facing page).
The authors conclude that baseline CSFI values were predictive of conversion to glaucoma and performed significantly better than conventional approaches for risk stratification.1887
Researchers in Pittsburgh and Boston followed 60 eyes of 31 subjects (four healthy, 34 glaucoma suspect and 22 glaucoma) for an average of 7.4 ±2.3 years to evaluate glaucoma progression in terms of the relationship between structure and function (using visual field mean deviation and OCT RNFL thickness) over the long term. Visual field progression was defined as a decline in MD ≥2 dB from baseline; for OCT, progression was defined as RNFL thinning ≥20 µm. Subjects had a median of 12 visual field and 27 OCT scans. The data showed:
• A total of 41 eyes (68.3 percent) progressed; 46.7 percent according to OCT criteria; 10 percent according to visual field criteria; and 11.7 percent according to both criteria.
• Of the seven eyes that progressed by both criteria, three progressed in terms of structure and function simultaneously; in three eyes OCT showed progression before visual field; and in one eye the visual field showed progression before OCT.
• Using the same progression criteria after four, five, six and seven years of follow-up, there was a gradual increase in the number of progressors with all criteria. The agreement between both devices increased from 6 percent to 14 percent.
• Fifty-three percent of eyes that were defined as OCT rapid progressors were also labeled as visual field rapid progressors.
The authors note that although they expected to find improved agreement between structure and function over extended follow-up, that increase was marginal. Thus even over the long term there may be limited detectable correspondence between structural and functional progression.1898
Glaucoma Surgery
Researchers at the North Shore LIJ Department of Ophthalmology in New Hyde Park, N.Y., conducted a retrospective study of 195 Descemet’s Stripping Automated Endothelial Keratoplasty grafts performed between January 2007 and August 2012, to assess the incidence of medical or surgical escalation of IOP management after DSAEK in patients with or without pre-existing glaucoma, and whether such escalation was associated with graft failure.
Seventy-one grafts were performed in eyes that had required surgical or medical management of elevated IOP prior to DSAEK; 22 were performed in eyes that had previous glaucoma surgery (including trabeculectomy and/or tube implant). One graft was performed concomitantly with a tube implant. The study found:
• Eleven eyes with preexisting glaucoma required a glaucoma valve implant within 24 months after DSAEK; only one eye without preexisting glaucoma did (p<0.0003).
• Thirty-one eyes with preexisting glaucoma that received grafts (46.5 percent) required drops for IOP management after DSAEK; only 23 eyes without pre-existing glaucoma that received grafts (18.5 percent) required drops (p<0.0002).
• Graft failure occurred in 17 out of 71 eyes with pre-existing glaucoma (23.9 percent); seven of these had glaucoma surgeries prior to DSAEK, and three of them had had a tube revision performed concomitantly with DSAEK. Four of the seven required rebubbling and one had hypotony postoperatively. Five required subsequent glaucoma tubes after DSAEK, and three of those five had intra- or postoperative complications during tube placement (including choroidals in two eyes and malignant glaucoma in one eye).
• In contrast, of the 124 eyes without glaucoma, only six DSAEKs failed, all without adjuvant IOP management following DSAEK. Five of the 124 (4 percent) required rebubbling, and of these, two went on to failure.
The authors conclude that adjuvant medical or surgical therapy is more likely to be needed after DSAEK in patients with a pre-existing history of glaucoma and that prior glaucoma surgery may be a risk factor for failure of DSAEK grafts.3093
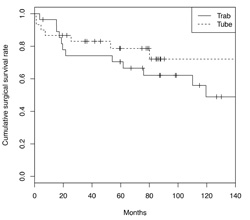
Researchers at Washington University in St. Louis retrospectively reviewed the charts of 58 eyes of 42 patients (mean age 55 years) with a diagnosis of uveitic or steroid-induced glaucoma, in order to compare the surgical outcomes of tube shunts versus trabeculectomy (with antimetabolites) in patients with glaucoma associated with ocular inflammation.
|
In this group there were 30 cases of tube shunt implantation (53 percent), 16 Ahmed, 14 Baerveldt; and 28 cases of trabeculectomy with mitomycin (47 percent), two receiving Ex-Press shunts. The most common etiologies of ocular inflammation were idiopathic (33 eyes, 55.9 percent), followed by sarcoidosis (12 eyes, 20.3 percent).
After controlling for covariates, tube shunts trended towards a 44.4 percent lower risk of surgical failure (HR: 0.56 95 percent CI: 0.21 to 1.49). Shunts also had a lower sustained risk of time to failure compared to the trabeculectomy group.4767
Risk Factors
A study at the University of Washington, Seattle and Duke Eye Center in Durham, N.C., partly retrospective and partly prospective, found that longer axial length is a risk factor for glaucoma, regardless of refractive status.
Retrospective data was collected from patients who had undergone biometry (IOLMaster) prior to elective cataract surgery at the University of Washington or Harborview Medical Center from 2009 to 2011; prospective data was collected from patients at the University of Washington Eye Institute Glaucoma Clinic who agreed to have ocular biometry. (They excluded patients with secondary causes of glaucoma and those classified as glaucoma suspects.) Of 901 eyes of 901 subjects, 281 had glaucoma; 620 did not. Average age was 67.16 ±12.38; corneal curvature was 43.87 ±1.67 D; axial length was 24.38 ±1.76 mm.
The data showed:
• Eyes with glaucoma had longer axial lengths (24.88 ±1.91 mm vs. 24.16 ±1.65 mm, p<0.001) and flatter corneas (43.62 ±1.73 D vs. 43.98 ±1.64 D, p=0.002) than eyes without glaucoma.
• In a multivariate analysis, age and axial length were significant independent risk factors for glaucoma.
• For any given refractive error, sub-jects with glaucoma had longer axial lengths than controls (0.218 ±0.882 mm vs. -0.099 ±0.82 mm, p<0.001).
The authors note that this suggests that increased axial length might be the pathophysiologic mechanism underlying the association between myopia and OAG, rather than strictly refractive error.3524
A study at the University of California, San Francisco and Stanford University was conducted to investigate possible associations between myopia and glaucoma risk in 115 young, healthy adults (mean age, 24.63; 61.74 percent female). The participants were categorized into two groups: 56 subjects with emmetropia or mild myopia (0.99 to -2.99 D) comprised Group A; 59 subjects with moderate to severe myopia (>-3.00 D) made up Group B. Anyone who had previously undergone LASIK was excluded.
Subjects underwent a comprehensive ophthalmic examination including visual acuity, refraction, slit lamp exam, ophthalmoscopy, IOP measurement, A-scan biometry, automated perimetry and optic nerve and anterior segment optical coherence tomography. Outcome variables included IOP, RNFL thickness, visual field mean deviation and pattern deviation. In addition to comparing the mean value of each outcome variable, a multivariate linear regression model was used to examine independent associations between spherical equivalents and outcome variables.
The data showed that mean IOP in group A was significantly lower than in Group B (13.13 mmHg vs. 14.68 mmHg, p=0.0064); mean RNFL thickness was greater in Group A (110 µm vs. 104.24 µm, p=0.0016); and mean visual field mean deviation and pattern deviation were significantly more suspicious for abnormality in Group A (-0.34 dB vs. -1.29 dB, p<.0001; and 1.46 dB vs. 1.71 dB, p= 0.02).
After adjusting for age, gender and ethnicity, the relationship between spherical equivalent and each of the outcome variables remained statistically significant.
The authors conclude that the association between greater myopia and higher IOP, thinner RNFL thickness and more suspicious visual field mean deviation and pattern deviation may represent greater glaucoma risk among young myopes, consistent with previous findings of increased risk in older myopic populations.3499
SLT Update
A retrospective study involving 79 eyes of 56 patients, conducted at the Massachusetts Eye and Ear Infirmary and Tufts University School of Medicine, sought to determine the repeatability of selective laser trabeculoplasty in patients with pseudoexfoliation glaucoma. (One researcher has a commercial relationship with Lumenis.) The subjects (24 males, 32 females) were PXFG patients who had undergone initial and/or repeat SLT between January 2001 and March 2012. (Anyone with prior laser or incisional surgery was excluded.)
The authors compared IOP, percent of IOP reduction and number of glaucoma medications before and after the first and subsequent laser treatments. The data showed:
• Of the 79 eyes that underwent an initial SLT, 21 eyes (27 percent) underwent a second SLT; five eyes (6.3 percent) underwent a third SLT.
• Of the eyes that did not have a second or third SLT, 43 (59 percent) had controlled IOP, while 15 (19 percent) underwent surgery.
• Of the eyes that had a second SLT but not a third, 11 (out of 21) had controlled IOP; five (21 percent) underwent surgery.
• Of eyes that had a third SLT, two (out of five) had controlled IOP; the other three underwent surgery.
• Time between SLT #1 and #2 was 29 ±24 months. Time between SLTs #2 and #3 was 17 ±11 months.
• Percent of IOP reduction was 39 ±18 percent after SLT #1, 33 ±14 percent after SLT #2, and 36 ± 19 percent after #3. In all three procedures, the reduction was statistically significant.
• There was no significant change in the mean number of medications before and after SLT #1 or SLT #3. However, the mean number of medications significantly increased after SLT #2 (from 2.19 to 2.67 meds, p<0.05).
The authors conclude that SLT was effective in reducing IOP in both initial and repeat treatments in eyes with pseudoexfoliation glaucoma. The percentage of eyes maintaining IOP control without additional SLT (about 50 percent) and those requiring surgery (about 20 percent) were similar after SLT #1 and #2. Although a small group, 60 percent of eyes after SLT #3 required surgery.1857 REVIEW
Dr. Netland is a professor and chairman of the department of ophthalmology at the University of Virginia; Dr. Singh is a professor of ophthalmology and director of the Glaucoma Service at Stanford University School of Medicine.