It’s no secret that having patients monitor their own eye-disease-related parameters is on the horizon. It’s also no secret that COVID-19 and the resulting restrictions in office visits caused a significant acceleration of that trend.
To learn more about the current status of this shift, we spoke to an ophthalmologist who is well-versed in this technology to find out how close we are to having home monitoring be an everyday reality.
Shifting the Focus
Lama A. Al-Aswad, MD, MPH, who practices at NYU Langone Health in New York City, is a professor of ophthalmology, a professor of population health, and the director of teleophthalmology, artificial intelligence and innovations at the NYU Grossman School of Medicine. She worked on the possibility of patient remote screening and monitoring for a number of years prior to COVID, but says the opportunity to really move forward with home monitoring came when the pandemic hit. “We were already experimenting with things like the iCare Home tonometer,” she says. (The iCare Home is a rebound tonometer that patients can use on their own eyes without the need for a topical anesthetic. To learn more, see “IOP Around the Clock: How Close Are We?” in the June 2018 issue of Review.) “We wanted to better understand how well a device like that works in real-world use, and whether it has a place in our practice.
“As part of my research I was sending the iCare device home with patients, free of charge—especially in cases where I was suspicious about the status of the patient’s glaucoma,” she continues. “I’d usually ask patients to check their pressure six times a day, which produced a lot of information, some of which was surprising. There were times when I was shocked by a patient’s data when I got the device back. Some had multiple IOP elevations at times of day when they weren’t in my office.
 |
| Home monitoring of IOP and visual fields can be done using devices like these. Proponents say the data has been shown to be clinically useful, and that most patients have been enthusiastic about the experience. |
“Then COVID happened, and everything changed,” she says. “In our practice, with the infrastructure available at NYU, we launched virtual patient visits using videoconferencing; we were up and running within two weeks of the government easing the restrictions on telemedicine reimbursement. We did more than 1,500 virtual visits over the next four months. This quickly increased the urgency of the question: How can we do a better job of evaluating vision remotely? I created a checklist for our practice listing the visual parameters we needed to be evaluating and how we might go about performing these visits.”
Checking Acuity at Home
Dr. Al-Aswad says the first two glaucoma-related factors they wanted to focus on assessing were visual acuity and IOP. “Initially, some people thought that sending a visual acuity printout, along the lines of the Amsler grid, might be a workable way to check visual acuity,” she says. “The problem is that you don’t know what the patient is doing with it. We decided that a digital app that could be used on a computer or smartphone would be a better approach.
“We quickly discovered that the existing apps weren’t ideal,” she continues. “First of all, we wanted an app that we could connect to our electronic health record system. Second, we noticed a challenge with these apps: the patient’s ability to zoom in and enlarge an image. If the patient can zoom in on your image, you’re going to have a hard time getting useful information about the patient’s visual acuity. For that reason, we decided to develop an app of our own. We began by creating a quick-and-dirty app in our lab called ‘Snap-eye,’ that doesn’t allow patients to alter the size of the test images. Once we had that in hand, we reached out to our institution and showed them what we’d done. We asked to create a more robust build, and connect it to EPIC, our electronic health record system.
“Ultimately,” she explains, “we created two versions of the app: one that can connect to our EHR, and a free-standing version that’s available for download at no charge at the Google Play store and the Apple App Store. We thought it might be something other ophthalmologists could use, especially during COVID. Our in-house version of the app allows patients to check their vision while waiting in the virtual waiting room during a virtual visit. The resulting data directly populates to our EHR.
“Since developing the app, we’ve validated it with 120 patients,” Dr. Al-Aswad notes. “Our study showed that it’s very accurate in patients with good vision, but somewhat less accurate when evaluating patients with worse vision—a common problem with apps designed for this purpose. We’re currently getting ready to publish that data. In the meantime, we’re working on other apps for similar purposes.”
IOP and Visual Fields
Regarding home-checking of IOP, Dr. Al-Aswad notes that COVID made it impossible to bring patients into the office for training in how to use the iCare device. “We created a virtual training visit,” she says. “We’d send the patient the device; they’d open it and then have a virtual training visit with the technician. They’d keep the device for a week, checking their pressure six times a day, and then mail it back to us.
“We did charge patients for that, to cover the cost of the device, the cost of shipping the device back and forth, and the cost of training,” she continues. “After we got the data back, we’d do a virtual visit with the patient to discuss the data and how their management would or wouldn’t change. The patients really liked this system. In fact, we had a wait-list of patients wanting to use the device, and we purchased an additional 10 devices to help meet the demand.”
Dr. Al-Aswad notes one issue they had with the iCare tonometer: They couldn’t plug the data directly into their EHR. “One of the NYU medical students in my lab, Jaideep Prasad, built an analog-to-digital connector for the iCare device,” she says. “Without that, we could only access the tonometry data on the iCare website; then we’d have to input the data manually or scan it into our EHR. With a digital connection, the data goes directly into our research electronic data capture, REDCap.”
Another key piece of information Dr. Al-Aswad wanted to collect was visual field data. “Prior to COVID we acquired virtual-reality visual fields to try using it for screening as part of our teleophthalmology mobile unit,” she says. (Virtual visual fields involve the use of goggles and oculokinetic perimetry, in which the patient’s eye moves to focus on changing stimuli, rather than remaining focused on a central point. To learn more, see “Checking Visual Fields Using Virtual Reality” in the March 2021 issue of Review.) “We thought, why not just send them to patients’ homes and let them do the testing?
“It took a while to get approval for this, and then do training and so forth,” she acknowledges. “Once we had that, we ran a small pilot study as a summer project for two of our NYU medical students, Galen Hu and Jaideep Prasad, to investigate the feasibility and acceptability of virtual-reality visual fields and the iCare Home when used by patients at home, and to validate their accuracy. We trained the patients in the office, then did a virtual training with them after they received the devices, and after using the devices they sent them back. The data produced by the virtual reality devices was clinically significant. Based on the data we got, I saw some patients sooner than we’d planned. Meanwhile, the response from patients was amazing; those who took the devices home loved them. Some said they liked doing the virtual visual field more than the Humphrey visual field. Similarly, 42 percent of these patients liked using the iCare Home device more than undergoing Goldmann in the office.
“At this point,” she notes, “it’s feasible to have patients monitor their own visual acuity, IOP and visual field at home.”
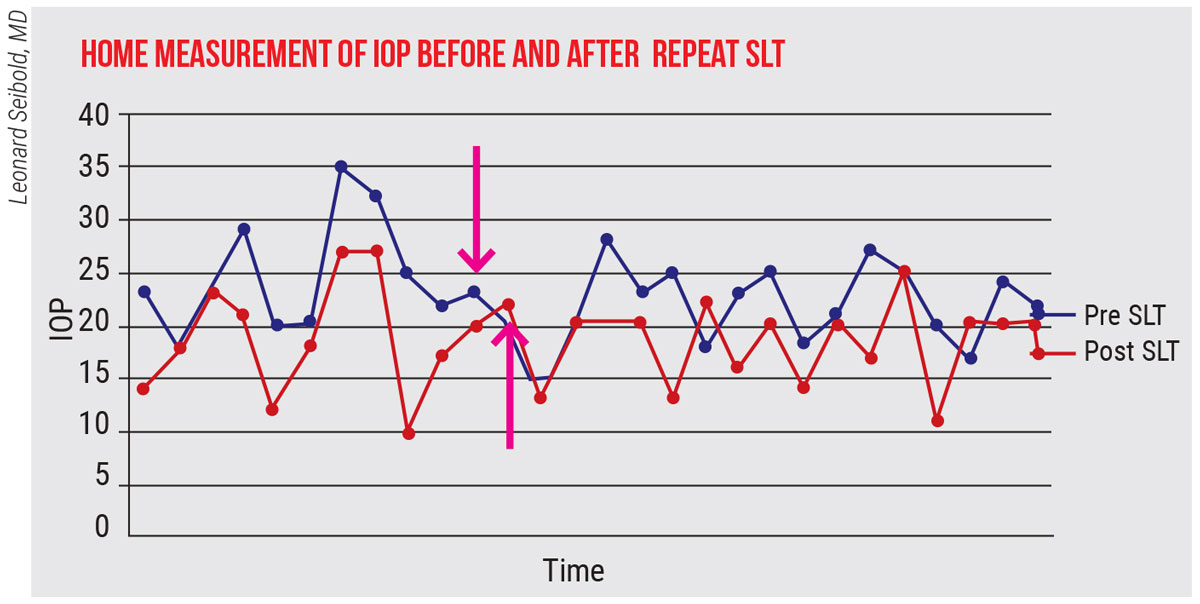 |
| These IOP measurements made by a patient before and after a repeat selective laser trabeculoplasty treatment illustrate a key advantage of multiple daily measurements. If in-office measurements had been taken at the time point shown by the arrows, a clinician might have concluded that the treatment didn’t lower the pressure significantly. However, with multiple home measurements, the mean pressure was shown to have dropped from 22.8 mmHg pre-treatment to 18.5 mmHg post-treatment, a 19-percent reduction. |
Retinal Imaging and OCT
Two more data points that would clearly be helpful are optic disc photos and optical coherence tomography scans. “Right now, patients taking their own retinal photos is theoretically possible, but it’s not easy to do,” Dr. Al-Aswad says. “With the current technology you need to have an experienced user and you have to dilate the pupil to get a good image, although some systems claim that dilation isn’t necessary. The bottom line is that it’s still early for this technology—but it will become more practical soon. I’ve also been looking at patient-performed OCT. It’s still in development and the cost is prohibitive, so it’s not going to be available to everybody for home use.”
Dr. Al-Aswad points out that technological advances, including artificial intelligence, are already suggesting new ways to obtain the kind of information OCT devices can reveal. “Felipe Medeiros published a study showing that you can train an AI algorithm to evaluate the optic nerve and predict nerve fiber layer thickness from a disc photo using machine-to-machine learning,” she says. “Maybe instead of home OCT, the future will include using AI to derive data like retinal or nerve fiber layer thickness from patient-captured images of the retina and optic nerve.”
One other practical issue also needs to be resolved: getting reimbursed when working with patients who are doing home monitoring. “Most remote-monitoring reimbursement codes are somewhat problematic because they’re not specific for ophthalmology,” she points out. “These codes were not created with high-cost devices—such as the ones used in ophthalmology—in mind. For example, the current codes require that the patient use a device more than 20 days a month. If you give a patient one of these devices for 20 days, you won’t recoup the cost of the device for a few years. Sometimes we use the codes in our practice, but we always get prior authorization from the insurance company.
“The other economic approach we found that made this work during COVID is to charge patients a flat fee to take the device home for a week,” she says. “With this system, you might recoup the cost of the device quicker just because you’re not asking the patient to keep it for 20 days. Theoretically, you can send the device to a different patient every week.”
Empowering Patients
“Part of the reason for my interest in home monitoring is that I believe patients need to be empowered to take care of themselves,” Dr. Al-Aswad says. “Although I’ve been a glaucoma specialist for 18 years, I hadn’t really thought about this until one of my patients said to me, ‘Doctor, I feel powerless between visits. I wait three to six months to see you, and I don’t know if I’m stable or getting worse. I need to have some power over my glaucoma.’ That opened my eyes. If patients have blood pressure problems or diabetes, they can measure their blood pressure or blood sugar between visits. But with glaucoma you can’t do anything between visits.
“Patients monitoring their own eyes is definitely going to happen; it’s only a matter of time,” she continues. “The technology we have today for accomplishing this is OK, but new technologies on the horizon will be even better. Right now, for example, companies are creating implantable devices that can measure IOP. One of them is already approved in Europe, and it’s only a matter of time before they’re available in the United States. Imagine being able to continuously monitor your IOP through your smartwatch, to see if there are changes that need to be addressed.”
Dr. Al-Aswad points out that patients are likely to be the biggest beneficiaries of the shift to home monitoring. “We don’t take into consideration how much time and effort regular office visits cost the patient,” she notes. “Yes, many of our patients are elderly and retired, but not all of them are, and even those who are retired may find these office visits challenging to manage. We need to respect the patient’s time and reduce the need for visits to our offices. And, we need to remember that our patients often feel powerless between visits.
“Home monitoring will address all of this,” she concludes. “We’ll be better able to understand and control the disease because we’ll have better data. Patients won’t have to give up so much time coming to our offices. And patients will finally feel that they have some control over their disease.
“This is the future,” she adds, “and it’s not far off.”
Dr. Al-Aswad reports no financial ties relevant to the article.