The Literature: Visual Outcomes
Here are some key studies that appeared in the past two years addressing the issue of whether FLACS produces better outcomes and/or fewer complications than manual cataract surgery. (Two meta-analysis studies are summarized at the end.)
First, the outcomes debate:
• A multicenter, case-control study conducted at multiple clinics in 18 European countries and Australia compared visual, refractive and adverse outcomes in 2,814 FLACS cases and 4,987 manual cases. Eyes were matched for preoperative corrected distance visual acuity, age and preoperative risk factors. For FLACS and manual, respectively:
— Seventy-two percent vs. 74.3 percent of eyes were within ±0.5 D of target.
— Postoperative logMAR CDVA in the two groups was 0.05 (6/6-3) for FLACS vs. 0.03 (6/6-2) for manual (zero=20/20); 96.3 percent vs. 97.1 percent had a CDVA of 0.3 (6/12) or better.
— One percent vs. 0.4 percent had a worse postoperative CDVA (five letters or more) at follow-up.
The authors also note that posterior capsule complications were observed in 0.7 percent of eyes (FLACS) vs. 0.4 percent (manual), and postoperative complications were seen in 3.4 percent vs. 2.3 percent, respectively.1
• A prospective, multicenter, comparative case series involving 1,876 eyes of 1,238 patients who underwent cataract surgery between January 2012 and June 2014 was conducted to evaluate visual outcomes after FLACS (988 eyes) vs. manual surgery (888 eyes). Among the findings:
— Postoperative BCVA was better after FLACS (20/24.5 vs. 20/26.4; p=0.0003).
— A greater proportion of FLACS cases achieved BCVA better than 20/30 (FLACS: 89.7 percent; manual: 84.2 percent; p=0.0006), and better than 20/40 (FLACS: 96.6 percent; manual: 93.9 percent; p=0.0077). However, manual cases gained more letters than FLACS cases (13.5 vs. 12.5 letters; p=0.0088), reflecting baseline BCVA differences.
— Mean absolute error was higher in FLACS than in manual surgery (0.41 D vs. 0.35 D; p<0.0011).
— The percentage of eyes within 0.5 D of target refraction was higher in the manual group (FLACS: 72.2 percent; manual: 82.6 percent; p<0.0001).
The authors concluded that FLACS did not demonstrate clinically meaningful improvements in visual outcomes over conventional phaco.2
• A 2016 single-center, single-intervention, prospective, comparative evaluation of 66 manual surgeries and 67 femto-assisted surgeries found:
— Postoperative UDVA was 20/20 or better in 62.7 percent of FLACS eyes and 61.5 percent of manual eyes (p=0.075). Postoperative UDVA was 20/25 or better in 81.5 percent of FLACS eyes and 78.5 percent of manual eyes (p=0.042).
— in the FLACS group, 80.6 percent of eyes were within ±0.5 D of targeted refractive equivalent; in the manual group, 75.2 percent were (p=0.8732). A slight undercorrection trend was noted in the manual group.
The researchers also looked at a subgroup of patients receiving toric lenses. Average residual manifest cylinder in the femto surgery toric subgroup was -0.45 D; in the manual toric subgroup it was -0.50 D.3
• A non-randomized, single-surgeon, prospective, comparative cohort case series compared the outcomes of patients receiving toric implants between January 2012 and July 2014; 95 eyes received manual surgery and 323 eyes received FLACS.
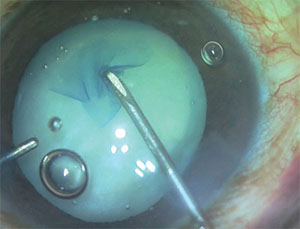 |
| Femto lasers are able to create very precise capsulorhexes, but they’re occasionally incomplete, requiring manual intervention. |
— In terms of postoperative BCVA, 97.5 percent of the laser group achieved 20/40 or better, while only 85.3 percent of the manual group did. However, this appeared to reflect preoperative differences; there was no significant difference in the amount of change. (The laser group gained 11 EDTRS letters; the manual group gained 10.3 letters [p=0.64].)
— Mean postoperative absolute refractive error was not significantly different (0.65 D for laser, 0.56 D for manual, p=0.18).4
• A prospective, randomized intra-individual cohort study of 100 patients compared visual recovery and refractive changes following FLACS in one eye and standard cataract surgery in the other. Six months postoperatively, 196 eyes were analyzed. The data showed that 90 eyes (92 percent) in the FLACS group and 70 eyes (71 percent) in the conventional group were within ±0.5 D of the target refractive outcome. (All eyes in both groups were within ±1 D of the target refraction.) In addition, FLACS yielded faster visual recovery and earlier stabilization of refraction.5
Complications & Safety
Here are some of the studies that focused on whether FLACS results in fewer complications:
• A prospective, consecutive, comparative cohort case series study conducted in Tasmania, Australia, compared the intraoperative complications and safety of FLACS (1,852 eyes) and conventional phaco surgery (2,228 eyes). It found:
— Anterior capsule tears occurred in 1.84 percent of FLACS eyes and 0.22 percent of manual eyes (p<0.0001).
— Anterior capsulotomy tags occurred in 1.62 percent of FLACS eyes.
— There was no significant difference in posterior capsule tears between the two groups (0.43 percent vs. 0.18 percent).
— The incidence of significant intraoperative corneal haze and miosis was higher in the FLACS group.
— Effective phacoemulsification time was significantly lower in the FLACS group (p<0.001).6
• A 2016 retrospective review compared rates of posterior capsule opacification in the first three months after surgery in 29 FLACS patients and 50 manual surgery patients. Seven of the 29 FLACS cases developed PCO requiring capsulotomy at three months; none of the manual cases required a capsulotomy during the same time period (p<0.05).7
• A 2016 paper reported the results of a retrospective case study done at two surgery centers in California, designed to compare YAG capsulotomy rates after manual vs. femto-assisted surgeries performed between August 2011 and August 2014. The data indicated that YAG capsulotomy rates were significantly lower in the femto-assisted group than in the manual group (p=0.04). The IOL material appeared to make a difference: Hydrophobic acrylic IOLs were associated with a lower capsulotomy rate than hydrophilic IOLs.8
• A 2016 prospective study conducted at a tertiary care ophthalmic institution compared manual to laser-assisted surgery in cases of white cataract. There were 40 eyes of 40 patients in each group. The data showed:
— The size of the capsulotomy/capsulorhexis was 4.9 ±0.1 mm in the laser group and 5.3 ±0.4 mm in the manual group (p<0.001).
— Mean circularity index was 0.996 ±0.003 in the laser group and 0.909 ±0.047 in the manual group (p<0.001).
— In the laser group, 52.5 percent of eyes had free-floating circular capsulotomies; 37.5 percent had microadhesions; 10 percent had an incomplete capsulotomy spanning one to two clock hours.
— Cases with release of white milky fluid had a higher incidence of residual adhesions (p=0.003).
— In the manual group, a multistep capsulorhexis was performed in 70 percent of the eyes.
— The data showed no difference in terms of visual outcomes or intraoperative complications.9
• A retrospective case series analyzed the vitreous loss seen in 3,784 manual surgeries performed by four surgeons between 2010 and 2012, and 3,371 femto-assisted surgeries performed in the following two years (2013 and 2014). The data showed:
— The rate of vitreous loss with exclusions was 1.17 percent in the manual group and 0.65 percent in the femto-assisted group; without exclusions, rates were 1.4 vs. 0.77 percent.
— Odds ratio analysis indicated that FLACS procedures performed in 2013 and 2014 were 1.6 times less likely (with exclusions) or 1.8 times less likely (without exclusions) to have vitreous loss than the manual surgeries performed earlier.
The authors conclude that converting from manual to FLACS resulted in a statistically significant decrease in vitreous loss.10
• In 2013, an interventional case series conducted in China studied 153 eyes that underwent FLACS and 161 eyes that underwent manual surgery to evaluate their comparative safety and effectiveness. The data showed:
— Effective phaco time was significantly lower in the FLACS group (14.05 vs. 23.65 seconds, p<0.05).
— The cumulative delivered energy was significantly lower in the FLACS group (4.78 vs. 8.82 percent, p<0.05).
— Corneal endothelial cell loss was significantly lower in the laser group than in the manual group at one month postoperatively (p<0.05).
— Postoperative anterior chamber flare was significantly greater in the manual group at day one and at one month (p<0.05).
— There were no severe surgical complications in either group.11
• A prospective, consecutive, nonrandomized, comparative cohort study of 70 eyes undergoing FLACS and 54 eyes undergoing manual surgery during a six-month period compared the efficacy and safety of the two procedures. The data showed:
— Endothelial cell density in both groups decreased significantly postoperatively but remained stable during follow-up; ECD was lowest at one month. FLACS showed a greater, but not statistically significant, endothelial cell loss than manual surgery.
— CCT in both groups increased, reaching maximum thickness on day one and tending to decrease thereafter. No significant differences were found between the groups.
— Flare values following manual surgery were greater than following FLACS, reaching statistical significance at six months (p=0.001).12
• A nonrandomized, single-surgeon, prospective, comparative cohort case series conducted in Australia sought to evaluate the incidence of postoperative clinical cystoid macular edema associated with femto-assisted surgery using the Catalys Precision Laser System (Abbott Medical Optics) in 833 eyes, vs. manual surgery in 458 eyes.
There were seven cases of postoperative CME (0.8 percent) in the FLACS group, vs. one case (0.2 percent) in the manual surgery group. This correlated with a change in laser treatment speed due to a software upgrade, suggesting that retinal safety thresholds need further careful analysis.13
• A retrospective chart review at a private surgical center in Hawaii compared the complication rate of FLACS (273 consecutive eyes) and traditional phaco (553 eyes) during the first 18 months of FLACS use at the center. The overall complication rate for FLACS was 1.8 percent, vs. 5.8 percent for the traditional procedure. A majority of the surgeons (80 percent) had a lower complication rate while using FLACS.14
Meta-analyses
• A 2016 paper reported the results of a meta-analysis of 14,567 eyes from 15 randomized controlled trials and 22 observational cohort studies that compared the efficacy and/or safety of manual and femto-assisted surgery:
— There was no significant difference between FLACS and manual surgery in UDVA (p=0.19); CDVA (p=0.26); or mean absolute error (p=0.57).
— There was no significant difference in total surgery time (p=0.07) or corneal endothelial cell count (p=0.07) between the techniques.
— There was a significantly higher concentration of prostaglandins after FLACS (p<0.001).
— There were no significant differences in the overall incidence of complications between FLACS and manual surgery (p=0.16), but posterior capsular tears were significantly more common in FLACS (p=0.005).15
• A meta-analysis of nine randomized, controlled trials and fifteen cohort studies compared outcomes in 2,861 eyes undergoing FLACS and 2,072 undergoing manual surgery. No significant differences were observed in the final CDVA or in surgically induced astigmatism. However, there were significant postoperative differences between the two groups favoring FLACS in endothelial cell loss percentage (at one week, one month and three months); in CCT (at one day, one month and at the final follow-up); in CDVA (at one week postoperatively); and in UDVA at the final follow-up. They also found significant differences in mean absolute error; effective phaco time; phacoemulsification power; and the circularity of the capsulorhexis.
The authors conclude that FLACS is a safer and more effective method for reducing endothelial cell loss and postoperative central corneal thickening than manual surgery, and achieves better and faster visual rehabilitation and refractive outcomes (although there was no difference in final CDVA or surgically induced astigmatism).16
Surgeons: The Pros & Cons
Amar Agarwal, MD, chairman of Dr. Agarwal’s Group of Eye Hospitals in Chennai, India (composed of more than 70 hospitals) has pioneered numerous surgical procedures, including bimanual phaco. He performs both FLACS and manual surgery, using either the Lensar or Victus laser system for FLACS. “There are pluses and minuses to both femto and manual phaco techniques, and it’s important to note the strengths of each,” he says.
Among the advantages
 |
| Different FLACS systems use different approaches to doc king with the cornea. Loss of suction may occasionally occur. |
• The laser centers and sizes the capsulorhexis very accurately. “It’s an absolutely circular opening, and you can create the exact size you want and locate it exactly where you want it to be,” says Dr. Agarwal. “That’s especially useful when working with a mature cataract; creating a good ‘rhexis can be difficult using the manual technique in that situation.”
Sheri Rowen, MD, who practices at NVision Eye Centers in Newport Beach, Calif., and is a clinical assistant professor of ophthalmology at the University of Maryland, agrees. “Also, if the lens is subluxed or decentered, the capsulorhexis can be made more accurately for proper placement,” she says. “In addition, when dealing with a white cataract, the capsulorhexis can be made more safely with the laser.”
• The femto laser is very reliable for making arcuate incisions to correct astigmatism. “When you’re doing an astigmatic correction, you’re making a partial-thickness radial incision in a particular axis,” says Dr. Agarwal. “I think the laser does a much better job of that than a surgeon can do manually.”
“Many surgeons don’t correct astigmatism at all because they don’t feel comfortable making arcuate incisions,” notes Dr. Rowen. “With femto-assisted surgery, as long as you can mark the eye you can easily correct a diopter or less of astigmatism, which will leave you with a much happier patient. I’ve also had patients who needed a diopter more astigmatism correction than I could give them with a toric intraocular lens. The laser let me add that extra diopter of correction.”
• It softens and fragments dense lenses, requiring less use of phaco energy inside the eye. “ That’s particularly beneficial in Fuchs’ endothelial dystrophy patients,” Dr. Rowen notes.
Concerns About FLACS
On the other hand, like manual surgery, using the laser has potential downsides:
• A laser capsulorhexis may not always be complete. “It’s important to pay attention and complete the capsulorhexis manually if this occurs, because if you continue with the surgery, you’ll cause a capsular tear,” says Dr. Agarwal.
• A capsulorhexis made by the laser may be inherently less strong than a manual capsulorhexis. “The laser is making a string of many small dots that join together to create the capsulorhexis,” Dr. Agarwal points out. “That doesn’t mean a femto capsulorhexis is bad, but I believe this makes it inherently weaker than one made manually. Fortunately, at a practical level that’s not a big deal; it doesn’t matter much in most surgeries.”
• A femto-generated bubble may break the posterior capsule. “A bubble is sometimes created when you’re doing the emulsification of the cataract,” says Dr. Agarwal. “When you remove the cataract, the bubble can break the posterior capsule.”
• FLACS can trigger intraoperative miosis. “This can happen because the laser energy causes prostaglandins to be released,” Dr. Agarwal explains. “For that reason, when you go in to remove the nucleus, you might end up with a small pupil. Fortunately, this doesn’t matter too much if you’re a good surgeon; you can redilate the iris using iris hooks or other devices.”
Dr. Rowen says she routinely uses Omidria (Omeros) during these surgeries. “It does an excellent job of getting the pupil back to where it was and keeps it from coming down during the case,” says Dr. Rowen. “I’m using the Malyugan ring less often to keep the pupil open since adopting Omidria.”
• Some femto machines have a problem making a clear corneal incision. “A clear corneal incision has to be full-thickness, and it has to be at an exact location,” notes Dr. Agarwal. “Sometimes, when you try to make the incision with a femto laser, it doesn’t go all the way through and you have to complete it manually. We recently had a case in which the femto system made the incision a little more towards the center of the cornea than we intended, creating a massive scar and leaving the eye with extra astigmatism. I believe you’re better off making a clear corneal incision manually.”
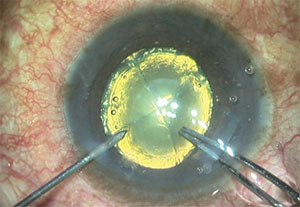 |
| Some FLACS systems have difficulty making a clear corneal incision because of their docking technology. In this case, the incision ended up too far anterior, causing excessive postoperative astigmatism. |
• Femto-assisted surgery can take significantly longer than manual cataract procedures. “It doesn’t take more time to do the manual portion of the surgery,” Dr. Rowen notes. “It’s a question of throughput time, because the patient has to go to two different rooms.”
She notes, however, that a solution to this problem may be on the horizon. “I recently tried the Ziemer system, and it allows the patient to stay in one room,” she says. “The Ziemer is portable and can be brought right into the OR, and because of its design it can be used in a sterile manner. I was able to do a sterile laser procedure and then slide my chair to the side of the bed and start performing the rest of the surgery immediately. The time savings were pretty remarkable.”
• A femto system is a big financial investment. “Clearly the cost of the laser is an issue,” says Dr. Rowen. “You need to be making a profit every month to pay off the cost, and the technology could become obsolete after five or six years.
“The question is, do you want to have a state-of-the-art practice?” she continues. “Patients are coming in to our clinic asking for laser surgery. Part of this may be the result of other surgeons telling them that the surgery can be done with ‘the laser or the blade,’ but whatever the reason, they’ve heard about it and they want it.”
• The patient has to pay more for femto surgery. “There’s no question that the femto laser creates a good capsulorhexis, but that shouldn’t be the only thing justifying the cost increase for the patient,” says Dr. Agarwal. “The question is, are we giving the patient enough benefit to justify the cost? It’s a very debatable point.”
Some Key Questions
Surgeons debating the value of offering FLACS to their patients are asking a number of questions:
• Does femto-assisted cataract surgery change the outcome? Dr. Agarwal says that in his experience, using the femto laser does not improve outcomes. “Both manual and femto techniques give fantastic results,” he says. “If you don’t have any complications, I don’t think there’s any difference in the end result at all, and patients are happy either way.”
• Are there fewer complications using the laser? Jeffrey B. Morris, MD, MPH, medical director of Morris Eye Group in Encinitas, Calif., says FLACS outcomes can be affected by the surgeon’s learning curve, but the surgeons in his group haven’t had to deal with too many complications. (Dr. Morris recently gave up performing cataract surgery, but still supervises multiple cataract surgeons in his group.) “We’ve had some incomplete capsulotomies, but that has improved over time with experience,” he says. “On rare occasions we get little tags that prevent a free-floating capsulotomy, but those are easily dealt with at the time of surgery. To the best of my knowledge, we haven’t had any ruptures of the posterior capsule, and all the fragmentations we’ve done were excellent. I don’t believe we’ve had any radial tears since using the femto laser, which is a departure from our manual capsulotomy experience, where you can unintentionally dissect out to the equator. Although we haven’t analyzed our data statistically, our surgeons believe they’re having fewer complications with femto-assisted surgeries than they did with manual.”
• Is femto better when implanting a premium lens? Dr. Agarwal says he doesn’t find that he gets better outcomes using the laser when the implant is a premium lens. “The reason some say it’s better is that the capsulorhexis made by the laser is very good,” he notes. “But you can do a perfectly good capsulorhexis using a manual technique. In my experience it doesn’t make any difference.”
Dr. Morris disagrees. “Our laser gives us a choice between pupil centration and visual axis centration to make sure we get accurate placement of the IOL, which is important with a premium implant,” he notes. “Since we switched to FLACS, we’ve had no problems with our premium IOLs. When we were doing strictly manual surgeries we got many more complaints about the premium implants.”
“In our experience, a laser-created capsulorhexis does make a difference when implanting premium IOLs,” says Dr. Rowen. “However, there aren’t any studies that demonstrate significantly better premium IOL outcomes as a result of using FLACS.”
• Is femto better if it’s a complicated case? “Femto-assisted surgery is really great when you’re dealing with a white cataract, pseudoexfoliation or floppy iris syndrome,” says Dr. Morris. “You don’t have to worry about complications. We’re able to make a perfect three-planar incision for the entry wound, and we haven’t had any instances of iris prolapse in those cases.”
• Is femto a good idea if the doctor isn’t a master surgeon? “I don’t buy that theory,” says Dr. Agarwal, “because in the end every surgeon has to do a bit of manual surgery—even those using a femto system. It’s true that the femto laser makes chopping easy, and the amount of energy used is much less. But you’re still performing manual surgery.”
Dr. Rowen disagrees. “If you’re an average surgeon, and you don’t always create a good capsulorrhexis, and you don’t always chop the nucleus well, the laser makes everything easier—with the exception of cortical removal. If you’re not good at chopping and you consistently create a bowl instead of nice segments, then the laser procedure would definitely make your surgeries go more smoothly.”
“Proving” Superiority
In the debate about whether FLACS is actually better than manual, two important factors are often overlooked. The first is that different laser systems have different pros and cons, which means your comparison will be affected by the strengths and limitations of the femtosecond laser system you’re using for the comparison.
“One issue that impacts the outcomes you achieve with the laser is which type of technology the instrument uses to dock with the cornea: immersion or non-immersion,” notes Dr. Morris, who has used multiple systems. “Immersion technology allows very good imaging of the posterior structures, the lens and the capsulotomy, so, in theory, you get a slightly more accurate fragmentation and capsulotomy. However, the layer of water undermines the ability of the system to image the cornea well enough to make very precise incisions, astigmatic keratotomies or pockets for corneal inserts like the Raindrop. In contrast, non-immersion technology allows more accurate corneal imaging, but it’s less accurate for imaging posterior structures, the capsulotomy and the nucleus for doing fragmentation.”
Dr. Morris’s practice uses the Bausch + Lomb Victus system, which allows the use of both technologies—immersion and non-immersion. “During the first part of the procedure we use immersion, which provides us with real-time imaging so we can watch the fragmentation taking place,” he explains. “Once fragmentation and capsulotomy are complete, the water bath is eliminated and we have flat, surface-to-surface contact with the cornea. This allows excellent corneal imaging and very accurate incisions and astigmatic keratotomies.”
Dr. Rowen agrees that different lasers have different strong points. “For example,” she says, “the energy level and spot separation used when chopping the nucleus can make the segments easier or more difficult to remove. I’ve worked with several different lasers, and the difference in this respect can be very noticeable.”
Dr. Rowen also notes that some docking systems are easier to manage than others. “The Lensar docks very easily,” she says. “You can stand over the patient and put the first piece on; then you plug the laser into it. The Ziemer system is even easier. Some other systems are more challenging.”
A second issue when comparing manual surgery to FLACS is that “proof” of superiority may be difficult or impossible to find. “Which data are you going to compare?” asks Dr. Morris. “When you look at manual surgery, which surgeons are you going to use as the basis for comparing complication rates? How do you equate the skill of the manual surgeon to the skill of the femto surgeon? How do you draw your sample to ensure that you’re comparing apples to apples? You’re always going to select your data according to some bias.”
Dr. Morris adds that accepting something as “proof” is subject to personal bias anyway. “You can always take studies that some consider to be ‘proof’ and argue with them,” he says. “In the case of femto-assisted surgery, having made such a large financial investment could also influence a surgeon’s perspective. So I think that deciding whether FLACS is better than manual is ultimately going to come down to your experience. I don’t think there will ever be conclusive proof.”
The Last Word
“I think the bottom line is, both techniques have benefits and limitations,” says Dr. Agarwal. “It all depends on the surgeon. Some surgeons are very comfortable with manual surgery and worry about the extra time and cost associated with performing femto cataract; others are very comfortable performing femto-assisted surgery.
“I always tell surgeons, do whichever type of cataract surgery you’re good at and comfortable with,” he concludes. “However, if you’re going to use the laser, don’t do it half-heartedly. Pay attention and be careful.”
Dr. Morris says that in his practice, the laser has been worth the cost. “It’s at least as safe as manual surgery,” he says. “It’s good for a practice to be involved in cutting-edge technology, and femto-assisted surgery is probably the future for ophthalmology. It can have a positive financial impact on a practice and pay for itself because you get higher rates of reimbursement. Ultimately, however, if you don’t believe in it, you probably won’t end up using it.”
“If you’re comparing standard, run-of-the-mill cataract surgeries in healthy eyes, I don’t think you’ll find any evidence that FLACS is better than manual surgery,” says Dr. Rowen. “But there are plenty of studies showing that using the laser has advantages, and I think it may allow a surgeon reaching his or her 60s or 70s to continue performing surgery instead of taking early retirement, because it assists with some of the more difficult and critical steps in the surgery.”
“I haven’t met a surgeon yet who wouldn’t want the femto laser used if they had cataract surgery themselves, whether or not they use the laser in their own practices,” adds Dr. Morris. “I think that’s very telling.” REVIEW
Drs. Agarwal, Rowen and Morris have no financial ties to any product mentioned.
1. Manning S, Barry P, et al. Femtosecond laser-assisted cataract surgery vs. standard phacoemulsification cataract surgery: Study from the European Registry of Quality Outcomes for Cataract and Refractive Surgery. J Cataract Refract Surg 2016;42:12:1779-1790.
2. Ewe SY, Abell RG, et al. A comparative cohort study of visual outcomes in femtosecond laser-assisted vs. phacoemulsification cataract surgery. Ophthalmology 2016;123:1:178-82.
3. Kanellopoulos AJ, Asimellis G. Standard manual capsulorhexis/ultrasound phacoemulsification compared to femtosecond laser-assisted capsulorhexis and lens fragmentation in clear cornea small incision cataract surgery. Eye Vis (Lond) 2016;3:20.
4. Oakley CL, Ewe SY, et al. Visual outcomes with femtosecond laser-assisted cataract surgery vs. conventional cataract surgery in toric IOL insertion. Clin Exp Ophthalmol 2016;44:7:570-573.
5. Conrad-Hengerer I, Al Sheikh M, et al. Comparison of visual recovery and refractive stability between femtosecond laser-assisted cataract surgery and standard phacoemulsification: Six-month follow-up. J Cataract Refract Surg 2015;41:7:1356-64.
6. Abell RG, Darian-Smith E, et al. Femtosecond laser-assisted cataract surgery vs. standard phacoemulsification cataract surgery: Outcomes and safety in more than 4000 cases at a single center. J Cataract Refract Surg 2015;41:1:47-52.
7. Rostami B, Tian J, et al. High rate of early posterior capsule opacification following femtosecond laser-assisted cataract surgery. Case Rep Ophthalmol 2016;7:3:213-217.
8. Tran DB, Vargas V, Potvin R. Neodymium:YAG capsulotomy rates associated with femtosecond laser-assisted vs. manual cataract surgery. J Cataract Refract Surg 2016;42:10:1470-1476.
9. Titiyal JS, Kaur M, et al. Comparative evaluation of femtosecond laser-assisted cataract surgery and conventional phacoemul-sification in white cataract. Clin Ophthalmol 2016;10:1357-64.
10. Scott WJ, Tauber S, et al. Comparison of vitreous loss rates between manual phacoemulsification and femtosecond laser-assisted cataract surgery. J Cataract Refract Surg 2016;42:7:1003-8.
11. Zhang GB, Ye XY, et al. Clinic analysis of femtosecond laser-assisted cataract surgery. Zhonghua Yan Ke Za Zhi 2016;52:2:93-8.
12. Yu Y, Chen X, et al. Comparative outcomes of femtosecond laser-assisted cataract surgery and manual phacoemulsification: A six-month follow-up. Clin Exp Ophthalmol 2016;44:6:472-80.
13. Ewe SY, Oakley CL, et al. Cystoid macular edema after femtosecond laser-assisted vs. phacoemulsification cataract surgery. J Cataract Refract Surg 2015;41:11:2373-8.
14. Chen M, Swinney C, Chen M. Comparing the intraoperative complication rate of femtosecond laser-assisted cataract surgery to traditional phacoemulsification. Int J Ophthalmol 2015;8:1:201-3.
15. Popovic M, Campos-Möller X, et al. Efficacy and safety of femtosecond laser-assisted cataract surgery compared with manual cataract surgery: A meta-analysis of 14,567 eyes. Ophthalmology 2016;123:10:2113-26.
16. Chen X, Chen K, et al. Comparing the curative effects between femtosecond laser-assisted cataract surgery and conventional phacoemulsification surgery: A meta-analysis. PLoS One 2016;11:3:e0152088.



