Sun Pharmaceutical Industries announced U.S. FDA approval for the New Drug Application of Xelpros (latanoprost ophthalmic emulsion) 0.005% for the reduction of elevated intraocular pressure in individuals with open-angle glaucoma or ocular hypertension. (Sun Pharmaceutical in-licensed Xelpros from SPARC in June 2015.) Xelpros—the first and only form of latanoprost that isn’t formulated with benzalkonium chloride—was developed using SPARC’s proprietary swollen micelle microemulsion technology. In randomized, controlled clinical trials of individuals with OAG or OH with a mean baseline IOP of 23 to 26 mmHg, Xelpros lowered IOP by a mean of 6 to 8 mmHg. For more information, visit MyXelpros.com.
New Heidelberg OCTA Module
The Spectralis expandable diagnostic imaging platform can now be upgraded with the OCT Angiography Module to perform noninvasive, layer-by-layer exams of flow in the vascular networks of the retina and choroid. The module can be added to new and existing Spectralis devices with the OCT2 Module. The multimodal imaging platform enables clinicians to compare OCT angiographies to other modalities such as structural OCT and dye-based angiographies as well as infrared, MultiColor and BluePeak images. To learn more, visit heidelbergengineering.com.
Alcon UV-absorbing-only IOLs
Alcon recently expanded its AcrySof intraocular lens portfolio with the introduction of multifocal and multifocal toric ultraviolet-absorbing IOLs, and the introduction of the AcrySof UV-absorbing monofocal IOL with the UltraSert Pre-loaded Delivery System. These lenses come without blue light filtering, which some surgeons dislike.
The new multifocal and multifocal toric options are the AcrySof ReSTOR +2.5 Multifocal UV-Absorbing IOL with
For more information on the new UV-absorbing-only intraocular lenses, visit myalcon.com.
New Laser Accessory
Following FDA 510(k) clearance, Iridex introduced its updated TruFocus LIO Premiere laser accessory to the U.S. market. The light combination and reflection viewing system, used with Iridex retina laser systems, is worn on the physician’s head and combines a laser treatment beam from an Iridex laser source with the illumination beam of a binocular indirect ophthalmoscope into a mixed optical beam. The physician uses a handheld ophthalmic exam lens to view and treat the retina through the pupil. For more information, visit iridex.com.
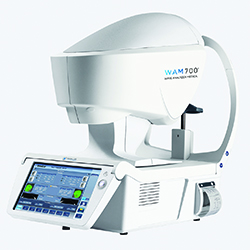 |
Essilor Launches Wavefront Aberrometer
Essilor Instruments launched the WAM700+ wavefront aberrometer for anterior-chamber analysis and visual-needs assessment. Based on Shack-Hartmann wavefront technology, Essilor says the device is designed to be a fast, effective and space-saving fully automatic wavefront aberrometer. Built for ease of navigation with a large touch screen, WAM700+ provides seven detailed measurements in 90 seconds for both eyes. The company says the device helps simplify screening for conditions such as cataract, glaucoma
 |
The Falcon Takes Off
Ocutech recently released the VES Falcon Autofocus Bioptic Telescope for low-vision patients.
Ocutech says the
The device is described as using crisp, bright, high-contrast, wide-field 4X Keplerian optics to provide a 12.5-degree field of view, with a focusing distance as close as 13 inches. The Falcon runs for eight hours on a single charge, and weighs 3.2 oz., making it comfortable to wear, Ocutech says.
The suggested patient price for the Falcon is $3,900, excluding customization charges and professional fees. For more information, visit Ocutech.com. REVIEW



