Anatomy
The sixth cranial nerve nuclei are located in the dorsal pons. Each nucleus contains the primary motor neurons for the ipsilateral lateral rectus muscle as well as the interneurons that travel through the medial longitudinal fasciculus to reach the contralateral third cranial nerve nucleus. Lesions or injury in this region will result in a complete ipsilateral horizontal gaze palsy, due to a deficit of abduction from the lateral rectus as well as an adduction deficit from the contralateral medial rectus. These lesions are usually also associated with ipsilateral facial nerve palsy (CN VII) due to the close proximity of these two nerves in the pons, as well as other brainstem signs such as hemiparesis and hemisensory loss.3
Each sixth cranial nerve nucleus issues sixth nerve fascicles that travel ventrally and laterally within the pontine tegmentum to exit at the pontomedullary junction. The nerve then enters the subarachnoid space and climbs vertically along the surface of the clivus. It subsequently travels over the petrous apex of the temporal bone to enter through the Dorello canal into the posterior cavernous sinus. Due to its long path along the subarachnoid space, the sixth nerve is particularly susceptible to damage due to traction forces of trauma and elevated intracranial pressure along this space.
Upon entering the posterior cavernous sinus, the sixth cranial nerve is joined by cranial nerves III, IV and V. It then enters into the substance of the cavernous sinus, where it runs lateral to the internal carotid artery and medial to the ophthalmic division of the trigeminal nerve. Due to the proximity of these structures, lesions occurring in the cavernous sinus often present with multiple cranial nerve palsies.
After exiting the anterior cavernous sinus, the sixth nerve then transverses the superior orbital fissure and enters the orbit through the annulus of Zinn to innervate the lateral rectus muscle.3, 4
Etiology
Lesions causing dysfunction of the sixth cranial nerve can be congenital or acquired, and can occur anywhere along its path, from the nucleus in the dorsal pons to the lateral rectus muscle in the orbit. They are composed of:
• Congenital dysfunctions. When a child presents with a congenital esodeviation and abduction deficit, the most common cause is Duane syndrome, followed by isolated congenital sixth nerve palsy. Duane syndrome is characterized by anomalous co-contraction of the medial and lateral rectus muscles on adduction of the involved eye, which causes the globe to retract. Studies have shown the cause of this motility disorder might be a hypoplastic or absent sixth nerve nucleus, with an aberrant branch of the third cranial nerve innervating the lateral rectus muscle.5 These two presentations may be difficult to differentiate, as the globe retraction feature of Duane syndrome may not be evident or may be difficult to elicit in infants. A distinguishing characteristic is that the deviation in primary position is usually much larger in sixth nerve palsy than it is in esotropic Duane syndrome.6
An isolated congenital sixth nerve palsy is rare, and, in most cases benign. It’s thought to occur secondary to increased intracranial pressure associated with the birth process and usually resolves spontaneously.6 It has also been associated with neurological conditions such as hydrocephalus and cerebral palsy.
• Acquired dysfunctions. The most common cause of an acquired sixth cranial nerve dysfunction in children is a neoplasm. This is followed by trauma, elevated intracranial pressure, inflammatory causes and post-viral etiologies.1,2,7-9
- Intracranial neoplasm. Studies report that tumors and tumor removal surgery account for 20 to 45 percent of sixth nerve palsies in children.1,2, 7-9 Tumors, either benign or malignant, can cause compression of the sixth nerve anywhere along its path and produce a unilateral or bilateral palsy. The most common tumors found in the pediatric population are posterior fossa tumors, such as brainstem glioma, medulloblastoma, ependymoma and cystic cerebellar astrocytoma. These patients usually present with other neurological symptoms such as ataxia and disturbance of gait along with sixth nerve palsies.10
- Trauma. Traumatic sixth nerve palsies can occur secondary to head trauma and skull base fractures. Approximately 12 to 42 percent of acquired sixth nerve palsies are of traumatic etiology.1,8
Trauma causes indirect pressure on the nerve, which is very susceptible to shearing forces as it passes over the apex of the petrous temporal bone and enters the cavernous sinus through the Dorello canal. These palsies may or may not improve over time, with maximum improvement typically occurring during the first six months after onset.11
- Elevated intracranial pressure. Elevated intracranial pressure can produce downward displacement of the brainstem, causing damage to the sixth nerves by pressure or traction of the nerves where they are tethered in the Dorello canal. Patients may also present with other signs of increased intracranial pressure such as headache, nausea, vomiting or papilledema. In children, this can occur secondary to a variety of underlying causes, including hydrocephalus, shunt failure, pseudotumor cerebri, posterior fossa tumors, neurosurgical trauma, venous sinus thrombosis, meningitis and Lyme disease.
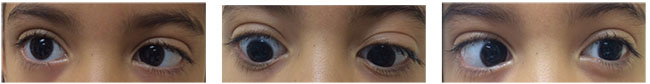 |
| Moderate angle esotropia in primary gaze with limited abduction on both right and left gaze, compatible with a diagnosis of bilateral sixth nerve palsy. |
- Inflammatory and infectious disorders. Pathologies such as meningitis, Lyme disease, varicella zoster and cytomegalovirus, among others, can cause inflammatory damage of the nerve along its course through the subarachnoid space. Chronic inflammation of the petrous bone in children with infections of the middle ear may cause an ipsilateral sixth cranial nerve palsy and facial pain, in a condition known as Gradenigo syndrome.4
Clinical Manifestations
Patients with sixth nerve palsy will present with dysfunction of the ipsilateral lateral rectus muscle, characterized by limited or no abduction of the affected eye on versions and ductions. When the nucleus is affected, patients will present with a complete horizontal gaze palsy. Other sixth nerve dysfunctions will present as an esodeviation on primary gaze, which increases with gaze towards the affected muscle. Characteristic features that differentiate an esotropia secondary to sixth nerve dysfunction from other types are the slowing of saccadic velocities of the affected lateral rectus muscle and weakness of the muscle on active force generation.
Older children with acquired sixth nerve palsies with good visual acuity may report binocular horizontal diplopia that worsens with gaze towards the paretic lateral rectus muscle. They will often have a compensatory head turn towards the side of the affected muscle to alleviate the double vision.6 However, younger children may not complain of diplopia due to suppression abilities. If the child presents soon after onset, suppression of the non-dominant eye has usually not been present long enough to produce suppression amblyopia, but a difference in visual acuity may be present later on.
Evaluation
The first aim of evaluation of these patients should be to identify the underlying cause of the sixth nerve palsy. Be sure to take a careful history, focusing on the presence of clinical features indicating a mass lesion and/or elevated intracranial pressure, such as focal neurologic complaints, headache, nausea and vomiting. The history should also include recent head trauma, infections, immunizations and other possible inciting factors. A complete neurologic examination should follow, evaluating for papilledema as well as any other neurologic abnormalities.
Urgent neuroimaging of infants and children with sixth nerve palsy depends upon whether the palsy is unilateral or bilateral and whether the palsy is isolated or accompanied by other neurologic abnormalities. Magnetic resonance imaging with and without gadolinium is the preferred modality of imaging to rule out an intracranial process, given the superior imaging capability of posterior fossa structures. Pay special attention to features such as a mass lesion, hydrocephalus, malformations and venous thrombosis. In adolescents, demyelination may be the cause, in which case MRI with fluid-attenuated inversion recovery imaging typically reveals T2 hyperintensities consistent with multiple sclerosis.4
If you find that the neuroimaging is normal, you can perform a lumbar puncture in order to measure the opening pressure in cases of suspected pseudotumor cerebri, or to measure the cerebrospinal fluid cell count, glucose and protein in cases of suspected meningitis. In addition, you can also perform serologic analysis, such as Lyme titers.8
Imaging Recommendations
Unilateral sixth nerve palsies in the absence of other neurological abnormalities are generally thought to be due to traumatic, postviral or idiopathic causes. However, recent studies looking exclusively at isolated palsies have found that tumors are actually the most frequent underlying cause in these patients, accounting for up to 30 percent of cases, followed by viral (19 percent) and traumatic (6 percent) etiologies.12 Due to the variability of these findings, recommendations regarding immediate neuroimaging differ. If follow-up can be assured, some authors recommend following an isolated unilateral sixth nerve palsy without imaging until resolution, unless neurologic symptoms and signs develop, the angle of strabismus deteriorates or the palsy fails to improve after three months of observation.12,13
On the other hand, in light of the high prevalence of associated intracranial lesions, the American Academy of Ophthalmology recommends obtaining an MRI in all children presenting with acquired sixth nerve palsy, even in the absence of other focal neurologic findings.6 Of note, the development of a sixth cranial nerve palsy following minor head trauma is unusual, and the child should also undergo neuroimaging for the high suspicion of an accompanying compressive lesion.
In patients with non-isolated unilateral or bilateral sixth nerve palsies, the recommendations are more conclusive. Neuroimaging is indicated emergently in patients with the presence of bilateral palsies or patients with either unilateral or bilateral palsies presenting in association with other neurological abnormalities and/or papilledema.
Management
The treatment of sixth nerve palsies depends on the underlying cause. The goal of ophthalmological management in pediatric cases of sixth nerve palsy is to first and foremost identify the etiology and refer the patient for specialized treatment of the underlying pathology as needed.
Secondly, other key goals are to maintain binocular vision and promote visual development.
Careful and close follow-up with the patient with a nerve palsy, preferably by a trained pediatric ophthalmologist, is required to assess any changes in the patient’s visual acuity levels and for proper sensorimotor evaluation of the patient. Spontaneous recovery may occur in patients with unilateral, isolated sixth nerve palsies. In younger patients, patching may be necessary to prevent or treat amblyopia. If a compensatory head posture allowing binocular fusion is present, patching may be optional unless there is a change in visual acuity or angle of deviation. Press-on prisms may be recommended to promote binocular vision and, in older patients, to alleviate diplopia in primary gaze. Some authors advocate the use of botulinum toxin injection of the ipsilateral medial muscle in order to cause temporary paralysis of the antagonist muscle.14,15 This may decrease the angle of esotropia while preventing secondary contracture of the medial rectus muscle due to unopposed contraction.
In cases of nerve palsies that don’t resolve after six months of management or observation, strabismus surgery may be indicated. Options for this surgical intervention include horizontal rectus muscle surgery—if the abduction function of the lateral rectus is partially preserved—or vertical rectus muscle transposition to the lateral rectus muscle if abduction is absent.
Counseling
Counseling patients and parents with cranial nerve palsies is often challenging, especially in cases of tumors. Many times, the psychological burden and stress, as well as follow-up with other specialties to treat the underlying pathology, will relegate ophthalmological evaluations to a lesser plane.
In any case, when faced with cases such as these, it’s important to provide the patient’s parents with the adequate information and reinforce the importance of continuous follow-up for visual development, as survival rates in children can be high depending on the pathology, and maximizing visual function is key for their future development. REVIEW
Dr. Osigian is an instructor at the Bascom Palmer Eye Institute, University of Miami Miller School of Medicine. Drs. Chang and Cavuto are assistant professors of clinical ophthalmology at Bascom Palmer.
1. Kodsi SR, Younge BR. Acquired oculomotor, trochlear, and abducent cranial nerve palsies in pediatric patients. Amer J of Ophthalmol 1992;114:5:568-574.
2. Harley RD. Paralytic strabismus in children. Etiologic incidence and management of the third, fourth, and sixth nerve palsies. Ophthalmology 1980;87:1:24-43.
3. Lee A, Brazis, PW. Sixth cranial nerve (abducens nerve) palsy in children. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on August 30, 2016.).
4. Foroozan R, Bhatti, MT, Falardeau J, et al. Neuro-Ophthalmic Anatomy: Ocular Motor Cranial Nerves. In: Cantor, LB, Rapuano, CJ, Cioffi, GA, (Eds.) Basic and clinical science course (BCSC) section 5: Neuro-Ophthalmology. American Academy of Ophthalmology (AAO), San Francisco; 2015.
5. Lueder GT, Archer, SM, Hered, RW, et al. Special Forms of Strabismus: Duane retraction syndrome In: Cantor, LB, Rapuano, CJ, Cioffi, GA, (Eds.) Basic and clinical science course (BCSC) section 6: Pediatric Ophthalmology and Strabismus. American Academy of Ophthalmology (AAO), San Francisco; 2015.
6. Lueder GT, Archer, SM, Hered, RW, et al. Esodeviations: Sixth Nerve Palsy. In: Cantor, LB, Rapuano, CJ, Cioffi, GA, (Eds.) Basic and clinical science course (BCSC) section 6: Pediatric Ophthalmology and Strabismus. American Academy of Ophthalmology (AAO), San Francisco; 2015.
7. Robertson DM, Hines JD, Rucker CW. Acquired sixth nerve paresis in children. Arch Ophthalmol 1970;83:5:574-579.
8. Lee MS, Galetta SL, Volpe NJ, Liu GT. Sixth nerve palsies in children. Pediatric Neurology 1999;20:1:49-52.
9. Merino P, Gomez de Liano P, Villalobo JM, Franco G, Gomez de Liano R. Etiology and treatment of pediatric sixth nerve palsy. J AAPOS 2010;14:6:502-505.
10. Brodsky M. Ocular motor nerve palsies in children. . In: Pediatric Neuro-ophthalmology. 2nd edition. New York: Springer; 2010: 253–308.
11. King AJ, Stacey E, Stephenson G, Trimble RB. Spontaneous recovery rates for unilateral sixth nerve palsies. Eye (London, England) 1995, 9 (Pt 4):476-478.
12. Dotan G, Rosenfeld E, Stolovitch C, Kesler A: The role of neuroimaging in the evaluation process of children with isolated sixth nerve palsy: Child’s nervous system. Childs Nerve Syst 2013;29:1:89-92.
13. Aroichane M, Repka MX. Outcome of sixth nerve palsy or paresis in young children. J AAPOS 1995;32:3:152-156.
14. Chuenkongkaew W, Dulayajinda D, Deetae R. Botulinum toxin treatment of the sixth nerve palsy: An experience of 5-year duration in Thailand. J Med Assoc Thai 2001;84:2:171-176.
15. Leiba H, Wirth GM, Amstutz C, Landau K. Long-term results of vertical rectus muscle transposition and botulinum toxin for sixth nerve palsy. J AAPOS 2010;14:6:498-501.



