Due to the close anatomic relationship between the orbit and paranasal sinuses, ophthalmologists must be cognizant of certain sinonasal diseases that may present with ocular manifestations. One such disease process, silent sinus syndrome, is a progressive condition in which maxillary sinus pathology causes inferior displacement of the orbital floor, resulting in enophthalmos and hypoglobus. The pathophysiology involves a negative pressure phenomenon within the sinus leading to inward bowing of the sinus walls. As such, SSS is often referred to as “chronic maxillary sinus atelectasis.”1
Since its initial report in the literature in 1994,2 our understanding of SSS has improved substantially. Although the “silent” nomenclature initially referred to the subclinical nature of sinus symptoms in affected patients, we now understand that patients with SSS may present with a range of different sinonasal or ocular symptoms.1
This review will provide an overview of our current understanding of the epidemiology, pathophysiology, diagnosis and management of SSS.
Epidemiology
SSS is a sparsely reported entity, with only about 150 cases reported in the literature.3 However, increased awareness of the condition since its initial description, combined with more widespread use of computed tomography (CT) imaging, has demonstrated that the condition may be more common than previously thought.4
Most reported cases involve adults during the fourth or fifth decade of life, although SSS has also been described in the pediatric population.5 There’s no clear gender predilection. However, for unknown reasons, the patient’s right side is slightly more commonly involved.1,6
Pathophysiology
Multiple theories have been proposed to explain the pathophysiology of SSS. Initially, it was suggested that congenital maxillary hypoplasia contributed to enophthalmos; however, this was disproven by cases of patients with normal sinus anatomy on imaging who subsequently developed SSS.6
The prevailing theory now involves hypoventilation of the maxillary sinus due to obstruction of the ostiomeatal complex (OMC). In normally functioning sinuses, the OMC serves as a channel linking drainage from the frontal, anterior ethmoid and maxillary sinuses to the middle meatus. With obstruction, there is impaired drainage of the sinuses. Over time, this blockage may contribute to the resorption of gases by capillaries within the closed sinus walls, resulting in negative pressure within the sinus. This pressure leads to an inward bowing of the sinus walls and subsequent atelectasis.1
One limitation of this theory is that it doesn’t explain why most patients with OMC obstruction don’t develop hypoventilation and, instead, develop symptoms of acute or chronic sinusitis.
Several additional pathophysiologic mechanisms have therefore been proposed. These include the possibility that a communication between the afflicted sinus and the pterygopalatine fossa may generate a negative pressure gradient during chewing.1 Furthermore, patients with SSS often develop thinning of the orbital floor, which contributes to hypoglobus. At the same time, many patients with chronic maxillary sinusitis may develop orbital floor thickening leading instead to proptosis. This contrast supports a theory that there may be poorly understood cytokine-mediated alterations in resorptive activity, sinus pressure and degree of inflammation that determine whether the disease presents with thickened or thinned orbital walls.1
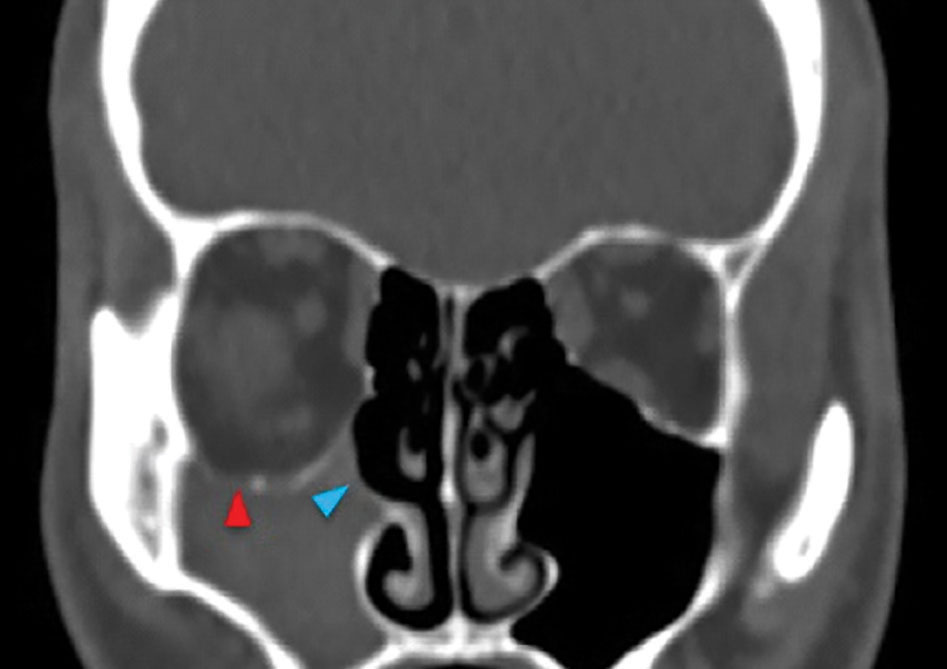 | 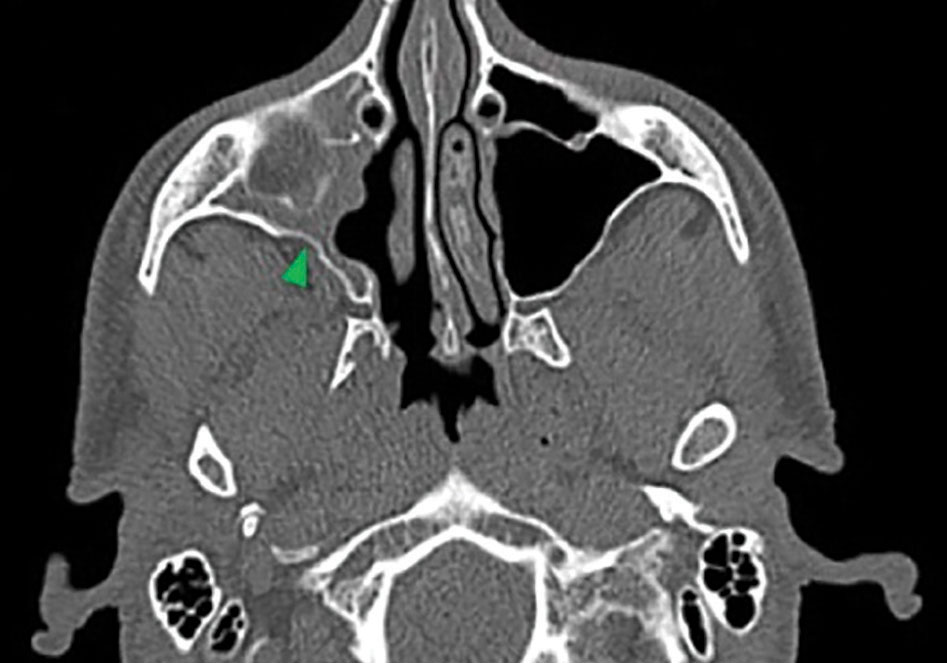 | |
| Figure 1. CT scan of a patient with SSS. A) Coronal plane: Note the inferior displacement of the orbital floor (red arrowhead) and lateral displacement of the uncinate process (blue arrowhead) into an opacified right maxillary sinus. B) Axial plane: Note the inward bowing of the right maxillary sinus walls (green arrowhead). | ||
Diagnosis
Following are the signs, symptoms and imaging results to look for when presented with a potential SSS patient.
• Presentation. The classic presentation of SSS involves painless enophthalmos and hypoglobus with subclinical sinus disease. A patient series from 2004 reported enophthalmos in the range of 2 to 6 mm (mean: 3.4 mm) and hypoglobus in a range of 1 to 6 mm (mean: 3.2 mm).4
Symptom onset is generally considered to be progressive. In a review of 84 published cases, the average duration of symptoms prior to presentation was 6.52 months. Despite this trend, several reports of acute onset of symptoms exist in the literature.4
In cases of severe hypoglobus, there may be constriction of the inferior oblique muscle, leading to restriction of upgaze; however, this occurs in a minority of cases.4 Rates of vertical diplopia due to hypoglobus muscle restriction range widely, but this is considered to be an uncommon presentation.7 Other uncommon symptoms include dental pain and dry eye, which may be attributed to lagophthalmos.1
On physical exam, there may be hypertropia of the afflicted eye and malar depression. Asymmetric eyelid creases can result from dysfunction of the levator palpebrae superioris, secondary to hypoglobus. Patients may appear to have signs and symptoms consistent with ptosis; however this is commonly pseudoptosis due to enophthalmos and hypoglobus. In some cases, patients may also exhibit pseudoretraction and lid lag as the oculomotor nerve fires to balance the inferior mechanical pull, which simultaneously activates the levator muscle.1
Importantly, consideration of sinonasal pathology shouldn’t be neglected in the setting of enophthalmos and hypoglobus. Patients should be questioned about symptoms of nasal obstruction, drainage, facial pressure or pain, as well as a history of sinusitis. Even if patients are asymptomatic, any clinical suspicion should prompt referral to an otolaryngologist for comprehensive evaluation, including nasal examination. Septal deviation is common in patients with SSS, and typically deviates to the ipsilateral side of disease.6 Additional findings on endoscopic examination to support the diagnosis of SSS include a lateralized uncinate process and enlarged middle meatus due to atelectasis of the maxillary sinus. Concomitant sinonasal inflammation may also be present, including edema and/or polypoid disease in the middle meatus.
• Radiology. When considering SSS, radiologic evaluation is critical for establishing a diagnosis, delineating anatomy and planning treatment. A CT scan of the orbit and sinuses is the gold standard for visualizing bony anatomy, although magnetic resonance imaging may also be adequate.
Radiologic hallmarks of SSS include unilateral atelectasis of the maxillary sinus with variable inward bowing of the sinus walls, lateralized uncinate process that may be adhered to lamina papyracea, depression of orbital floor and obstruction of the OMC (Figure 1). The maxillary sinus is most commonly opacified, but there are reports of SSS in patients with aerated sinuses.3
• Differential diagnosis. While the classic symptoms of painless enophthalmos and hypoglobus are suggestive of SSS, they shouldn’t be considered pathognomonic. A broad differential diagnosis may include infectious processes (e.g., chronic sinusitis, osteomyelitis), malignancy, inflammatory or vasculitic diseases (pseudotumor, granulomatosis with polyangiitis or scleroderma), trauma or iatrogenic causes (e.g., surgery, external beam radiation). Many of these listed alternatives may present with other clinical signs and symptoms, which may predate the ocular symptoms and suggest something other than SSS. A thorough history and physical should be obtained to identify the most likely etiology and ensure appropriate treatment.8 (A summary of the clinical and radiologic characteristics of SSS is shown in Table 1, above.)
Treatment
Treatment of SSS involves two primary goals: first, to improve maxillary sinus drainage and relieve obstruction in order to prevent disease progression; and, second, to restore normal orbital anatomy. These two goals may be accomplished simultaneously or separately through a variety of techniques.
Functional endoscopic sinus surgery (FESS) performed by an otolaryngologist is the mainstay of treatment for SSS. At a minimum, it entails a maxillary antrostomy that’s performed by completely removing the lateralized uncinate process and widely opening the natural sinus ostium. This procedure promotes improved sinus drainage and improves aeration. Importantly, because in SSS the uncinate process is lateralized and the orbital floor may be lowered, there’s an increased risk of orbital injury when a maxillary antrostomy is performed. Appropriate preoperative consideration of this anatomy is critical.
In some cases, FESS alone may lead to both improvement of the underlying disease process and resolution of the orbital symptoms.6,9 Studies have demonstrated bony remodeling of the maxillary sinus walls following FESS with resolution of atelectasis, thickening of bone and decreased symptoms.
For patients with persistent enophthalmos or in patients with such severe disease that complete recovery would be unexpected with FESS alone, orbital floor reconstruction can be performed. The timing of this step is controversial. Given that FESS alone may halt progression of ocular symptoms over time, simultaneous reconstruction of the orbital floor may be unnecessary and cause overcorrection of the deficit. Proponents of this strategy tout its reduced risk, noting that it minimizes anesthesia and trips to the operating room. Alternatively, orbital reconstruction can be performed as a second-stage operation two to six months after FESS. This allows for any orbital changes associated with FESS to occur, potentially avoiding overcorrection with orbital floor reconstruction. Furthermore, a two-stage approach prevents placement of an orbital floor implant into a potentially infected sinus cavity.1
 |
Options for surgical floor implants include alloplastic implants (e.g., titanium, hydroxyapetite, silicone, nylon) or autogenous (e.g., septal or costochondral cartilage, calvarial bone grafts). Implants are typically placed via a transconjunctival or subciliary approach and fixated to the orbital rim for stability.1 Alternatively, a nonsurgical option for treatment of enophthalmos after FESS is injection of hyaluronic acid into the intraconal and extraconal space. Studies have shown durable results for six months with this approach.10
In conclusion, SSS is an example of how sinonasal disease can manifest with ocular symptoms. In patients with painless hypoglobus and enophthalmos, a diagnosis of silent sinus syndrome should be suspected. A multidisciplinary approach with referral to an otolaryngologist should be adopted. Management involves functional endoscopic sinus surgery to improve sinus aeration, with or without orbital floor reconstruction. REVIEW
Dr. Rimmer is a fourth year resident in the Department of Otolaryngology-Head and Neck Surgery at Thomas Jefferson University in Philadelphia. Dr. Fastenberg is the Rhinology and Endoscopic Skull Base Surgery fellow in the Department of Otolaryngology-Head and Neck Surgery at Jefferson. Drs. Rosen, Nyquist and Rabinowitz are members of the Division of Rhinology and Skull Base Surgery in the Departments of Otolaryngology-Head and Neck Surgery and Neurological Surgery at Thomas Jefferson University Hospital. They have specialized training in the management of complex diseases of the paranasal sinuses, medial orbit and anterior cranial base.
1. Pula JH, Mehta M. Silent sinus syndrome. Curr Opin Ophthalmol 2014;25:6:480-484.
2. Soparker CN, et al. The silent sinus syndrome. A cause of spontaneous enophthalmos. Ophthalmology 1994;101:4:772-8.
3. Lee DS, Murr AH, et al. Silent sinus syndrome without opacification of ipsilateral maxillary sinus. Laryngoscope 2018;128:2004-2007.
4. Soparker CN, Patrinely JR, Davidson JK. Silent sinus syndrome— New perspectives? Ophthalmology 2004;111:2:414-415.
5. Chang DT, Truong MT. A child with silent sinus syndrome and spontaneous improvement after sinus surgery. Int J Pediatr Otorhinolaryngol 2014;78:1993-1995.
6. Martinez-Capoccioni G, Varela-Martinez E, Martin-Martin C. Silent sinus syndrome an acquired condition and the essential role of otorhinolaryngologist consultation: A retrospective study. Eur Arch Otorhinolaryngol 2016;273:3183-88.
7. Gomez J, Liu D, Palacios E, Nguyen J. Diplopia: An uncommon presentation of silent sinus syndrome. Ear Nose Throat J 2015;94:7:258-60.
8. Numa WA et al. Silent sinus syndrome: A case presentation and comprehensive review of all 84 reported cases. Ann Otol Rhinol Laryngol 2005;114:9:688-94.
9. Kram YA, Pletcher SD. Maxillary sinus posterior wall remodeling following surgery for silent sinus syndrome. Am J Otolaryngol 2014;35:623–625.
10. Mavrikakis I, Detorakis ET, Yiotakis I, Kandiloros D. Nonsurgical management of silent sinus syndrome with hyaluronic acid gel. Ophthal Plast Reconstr Surg 2012;28:e6–e7.



