This month, we discuss the quality of life aspects of ocular allergy and how this seemingly benign condition profoundly affects everyday life. This is also an opportunity to discuss the role of QoL questionnaires in broadening our understanding of disease, and to provide a brief primer on questionnaires used in the allergy space as tools for improving patient management.
While QoL questionnaires aren’t routinely used in clinical practice, they’re becoming an important component of clinical trials. Patient reported outcomes, or PROs, aren’t simply symptom measures graded on different severity scales. In general, QoL questionnaires have domains for impact of the disease on sleep and mood; productivity; perception of physical appearance; interference with daily activities and social interactions; and satisfaction with treatments. The widespread implementation of the Ocular Surface Disease Index as an assessment of dry-eye disease is an example of a success story in which PROs are used not only as an endpoint in clinical trials to assess the efficacy of a therapeutic intervention, but have also crossed over into patient management.1
The Food and Drug Administration has provided extensive guidance on the development and implementation of PROs in clinical trials, recognizing that the patient’s expectations and experience play important roles in the assessment of a therapeutic.2,3 Perhaps just as important, evidence demonstrates a strong predictive value of patient’s perception of his own status using these QoL tools.4 In ocular allergy, we’ve been assessing QoL of patients by adapting questionnaires used in general allergy, rhinitis and asthma. Considering that allergic conjunctivitis is thought to impact 39 percent of the U.S. population,5 and that there is significant comorbidity with other ocular diseases and with other allergic disorders, it behooves the practitioner to listen to his patients and to adopt as holistic approach as possible.
Questionnaires’ Validity
Like all measures of disease and treatment, QoL questionnaires must be developed through rigid statistical analyses of their validity, to confirm that they’re measuring what they say they’re measuring. Psychometric tests must prove to have both convergent and discriminant validity, together defined as construct validity, to be effective tools. Construct validity is simply a determination that the measures of constructs that should be correlated or related to each other actually are (correspondence or convergence), and measures of constructs that theoretically shouldn’t be related are not (discriminant). For example, if four queries on ocular allergy—fatigue, low productivity, itchy eyes and sneezing—are highly correlated, then these four items together show convergent validity (although the interrelationship isn’t known to be due to ocular allergy per se), but not discriminant validity. To establish the latter, you need to show that these four related items are unrelated to other interrelated items, proving that they can discriminate and thus provide a measure of ocular allergy impact and not, for example, a parallel factor such as job satisfaction.
Although correlation testing assures us that the subsets are related to the domain in question and not to each other, how can we be sure that they are actually measuring ocular allergy burden? Other examples of validity testing are concurrent validity (how well this QoL tool correlates with other QoL tools); known-group validity (how it performs in a well-identified group of ocular allergy patients); clinical validity (its ability to discern the severity of allergy among patients); floor and ceiling effects (what percentage of patients rates the highest or lowest scores indicates the level of responsiveness); and scale-to-scale correlations (which determine if individual scales are measuring different attributes).6
Rhinoconjunctivitis
A validated disease-specific questionnaire, the Rhinoconjunctivitis Quality of Life Questionnaire, has been shortened to the miniRQLQ in order to measure the impact of allergies on other aspects of daily activities in adults with rhinoconjunctivitis. Using a seven-point scale, where 6 represents the greatest impairment and 0 represents the least, patients assess the impact of rhinoconjunctivitis in the five domains of activity (daily activities, work/school performance and sleep), practical problems (the need to rub the eyes and blow the nose repeatedly), and nasal, ocular and other symptoms. A clinically significant change in the mini-RQLQ score is
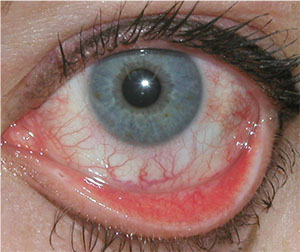 |
| Allergic conjunctivitis may impact 39 percent of the U.S. population.5 |
In one cohort of 447 allergic patients, 58.4 percent were recruited from primary care physicians, and the remainder from specialists. Of these, 44 percent reported moderate to severe ocular and nasal symptoms. Subjects reported that their ocular symptoms were better controlled than their nasal ones: 14.8 percent reported poorly controlled nasal symptoms compared to 5.8 percent with poorly controlled ocular symptoms. Conversely, of the 54 percent who reported having itchy/red eyes, 17.4 percent considered this symptom to be the worst in their spectrum of allergic signs and symptoms. Physicians, on the other hand, overestimate the burden of nasal congestion in their patients and underestimate the burden of sneezing, ocular symptoms, sinus pressure, sore throat, headache, snoring, nocturnal wakening, coughing and wheezing. Although this questionnaire didn’t separate out ocular symptoms for their effect on activities, the presence of allergic symptoms significantly affected sleep in half of patients, with almost 80 percent of chronic allergy sufferers and 50 percent of seasonal sufferers complaining of insufficient sleep. The majority also reported an impact on daily activities and on work/school performance.8
Eye Allergy’s Impact
The validity of one of the few tools developed specifically for ocular allergy, the Eye Allergy Patient Impact Questionnaire, was assessed for reliability as a measure of ocular allergy symptoms and their impact on health-related QoL, work productivity and treatment satisfaction.9 After validity analyses, some of the 49 items were recommended for elimination to provide a more accurate tool with high internal and test-retest reliability. The final tool included 20 items: eight in the daily life impact; four in the psychosocial impact; five in the symptoms category; and three in the treatment satisfaction category. This questionnaire allowed for separation of patients with different eye allergy symptom severities, as rated by patients and clinicians, and provided evidence that the scales used in this tool are responsive to changes in eye allergies.9
A similar report narrowed the focus to ocular symptoms, presenting the results of the miniRQLQ, the Work Productivity and Activity Impairment Allergic Specific Questionnaire, and the Pittsburgh Sleep Quality Index in 1,009 allergic rhinitis patients in four European countries.10 Results showed that the presence of ocular symptoms, found in 69 percent of this cohort, significantly reduced QoL work productivity and increased resource utilization, irrespective of nasal symptom severity. The presence of ocular symptoms had a greater impact on hours of work missed and impairment while working, with an additional half-day off of work in the previous three months due to allergies. Sleep quality was also significantly worse in these patients compared to AR patients without red itchy eyes. Lastly, the severity of ocular symptoms had a significant detrimental impact on all outcomes. These findings are remarkable, considering that allergic conjunctivitis is thought to be the poor cousin of allergic rhinitis in terms of the magnitude of disease burden in the United States and Europe.
A different study assessed sleep and mood disorders in dry eye and allergic conjunctivitis in 715 outpatients in six clinics. The Pittsburgh Sleep Quality Index was again implemented along with another tool, the Hospital Anxiety and Depression Scale. While the main findings focused on the worst effects of dry eye on QoL measured within these domains, more than 35 percent of allergic conjunctivitis subjects reported sleep disorders (cutoff 5/6 on the PSQI), and 42 percent reported mood disorders (cutoff 9/10 HADS).11
Phone and Online Surveys
Telephone surveys are another tool for determining the degree of burden of a disease within a population. In 2012, 2,765 individuals were randomly selected from a pool of those diagnosed with nasal or ocular allergies, as well as 500 health-care providers in seven specialties. Subjects rated symptom distribution seasonally, with peaks in March through May and September. Nasal congestion was most commonly reported as the most bothersome symptom (39 percent), with red itchy eyes not far behind (34 percent). Twenty-nine percent of respondents reported that their daily life was significantly impacted, with workers indicating a 29-percent reduction in productivity when symptomatic. Providers reported that itchy eyes were the most common complaint that prompted patients to seek treatment.5 In a European study of 1,482 allergic rhinitis patients, more than 80 percent reported some impairment of daily life when symptomatic. Notably, physicians once again rated symptoms as less severe than subjects rated their symptoms, affirming the need for these tools in determining disease burden and treatment.12
Ocular symptoms often tip the scales when it comes to detrimental effects on QoL. In a survey of 2,150 adolescents, the prevalence of rhinitis alone was 18.2 percent, and that of rhinitis and conjunctivitis was 20.5 percent. Rhinoconjunctivitis was more frequently associated with female sex, atopic history, household exposure to molds, passive smoke exposure and reported nearby truck traffic, along with higher levels of allergic sensitization. Asthma was found in 1.7 percent of subjects with no rhinitis or conjunctivitis, 5.1 percent in teens with rhinitis, and 10.7 percent in teens with rhinoconjunctivitis—a whopping twofold risk of asthma in adolescents with rhinoconjunctivitis compared to rhinitis alone. QoL was also worse: 10.7-percent impact in rhinoconjunctivitis versus 4.6 percent in rhinitis.13 This means that teens are sicker, more allergic and more adversely affected when ocular allergies are present in addition to rhinitis. Another study that reviewed the use of PROs in rhinoconjunctivitis studies from 2012 to 2014 found that fatigue and malaise are common outcomes in adults and children with rhinoconjunctivitis, recommending a more whole-body approach to assessments of disease burden, rather than the more simplistic focus on the eyes and nose.14 These symptoms are also associated with antihistamines, a reminder to be aware of potential iatrogenic QoL issues.
In a population-based study of 4,019 subjects who had undergone face-to-face interviews, 31.7 percent (1,276 patients) had allergic rhinitis, and 19 percent (763 patients) had ocular allergy. Of the group with ocular allergies, 52 percent also had allergic rhinitis. Again, compared to nasal symptoms, 51.7 percent of subjects rated ocular symptoms more troublesome. The impact of ocular symptoms on daily activities was “very important” or “moderate” in 38.8 percent of subjects, with blurry vision the most commonly reported in 47.8 percent. A total of 16.3 percent of subjects had sleep disturbances; work efficiency was reduced in 25.8 percent; and 12.9 percent of subjects had taken sick leave for up to three days because of ocular symptoms.15
In a telephone survey focusing on allergic conjunctivitis, 205 subjects responded. An overwhelming 83.9 percent reported also having nasal allergies, while fewer people (18 to 31 percent) reported suffering from food or skin allergies or asthma. Year-round allergies were reported in 38 percent of subjects. The second most bothersome symptom after itchy eyes was tearing and not redness.16
Treatment and QoL
Adding QoL endpoints to clinical trials or post-marketing comparative studies allows us to accumulate data on the real and perceived efficacy of treatments. Patients (n=540) rated intranasal treatments with fluticasone furoate, mometasone or fluticasone propionate equally in terms of efficacy and QoL parameters. Furthermore, in allergic patients who experienced both ocular and nasal symptoms (71.6 percent), the mean number of symptom-free days was fewer than in the overall population, indicating a more severe disease presentation in subjects with both nasal and ocular allergies. In this ocular subgroup, fluticasone furoate treatment resulted in a greater number of symptom-free days than mometasone-treated patients.17
Satisfaction with treatment is high on the list of outcomes rated in QoL questionnaires. Surprisingly, in the online U.S. survey, satisfaction was highest with immunotherapy (94 percent), although only 10 percent of subjects are treated this way.18
Cost to the patient is an important consideration, something that we as physicians sometimes forget. Of those subjects who preferred OTC medications, 72 percent noted that it was important that their medication be within their budget. For those using prescription meds, 69 percent and 65 percent, respectively, reported “covered by my prescription plan” and ‘“available at my lowest copay” as important criteria.18 Cost may be one reason the 2013 survey found that as many as 71 percent of seasonal and 53 percent of perennial allergy sufferers didn’t seek treatment from an eye specialist, and 40 percent didn’t even purchase over-the-counter medications for allergy management. Nevertheless, 80 percent of those who did use drops rated them as effective all or most of the time.16
Staying Mindful
QoL tools help us to help our patients. We should remind ourselves to discuss with our patients more holistic health issues that may seem to have nothing to do with ocular signs and symptoms. Itchy and tearing eyes are making our patients miserable, and the collateral effects of these seemingly benign complaints have more far-reaching consequences for an individual’s well-being and contribution to society. We as health practitioners owe it to our patients to be mindful of the psychological, emotional and societal effects of even non-vision-threatening ocular diseases such as ocular allergy. REVIEW
Dr. Abelson is a clinical professor of ophthalmology at Harvard Medical School. Mr. Gomes is vice president of allergy at the ophthalmic research and consulting firm Ora Inc.; Ms. Smith is a medical writer at Ora. Dr. Hollander is chief medical officer at Ora, and assistant clinical professor of ophthalmology at the Jules Stein Eye Institute at the University of California, Los Angeles.
1. Schiffman RM, Christianson MD, Jacobsen G, et al. Reliability and validity of the ocular surface disease index. Arch Ophthalmol 2000;118:5:615-621.
2. Guidance for Industry. Patient-reported outcome measures: Use in medical product development to support labeling claims. U.S. Department Health and Human Services. FDA; CDER, CBER, CDHR. December 2009.
3. McLeod LD, Coon CD, Martin SA, Fehnel SE, Hays RD. Interpreting patient-reported outcome results: U.S. FDA guidance and emerging methods. Expert Rev Pharmacoeconom Outcomes Res 2011;11:2:163-169.
4. Asadi-Lari M, Tamburini M, Gray D. Patients’ needs, satisfaction, and health related quality of life: Towards a comprehensive model. Health and Quality of Life Outcomes 2004;2:32.
5. Bielory L, Skoner DP, Blais MS, et al. Ocular and nasal allergy symptom burden in America: The AIRS surveys. Allergy and Asthma Proc 2014;35:211-218.
6. Leidy NK, Revicki DA, Geneste B. Recommendations for evaluating the validity of quality of life claims for labeling and promotion. Value in Health 1999;2:2:113-117.
7. Bousquet J, Khaltaev N, Cruz AA, et al. Allergic Rhinitis and its Impact on Asthma 2008 update. Allergy 2008;63(S86):8-160.
8. Schatz M. A survey of the burden of allergic rhinitis in the USA. Allergy 2007;62:85:9-16.
9. Alexander M, Berger W, Buchholz P, et al. The reliability, validity, and preliminary responsiveness of the Eye Allergy Patient Impact Questionnaire. Health and Quality of Life Outcomes 2005;3:67.
10. Virchow JC, Kay S, Demoly J, Mullol J, Canonica W, Higgins V. Impact of ocular symptoms on quality of life (QoL), work productivity and resource utilization in allergic rhinitis patients – an observational, cross sectional study in four countries in Europe. J Med Econom 2011;14:3:305-314.
11. Ayaki M, Kawashima M, Negichi K, et al. Sleep and mood disorders in dry eye and allied irritating ocular disorders. www.nature.com/Scientific Reports 2016 6:22480/doi:10.1038/srep22480.
12. Canonica GW, Bousquet J, Mullol J, et al. A survey of the burden of allergic rhinitis in Europe. Allergy 2007;62(Suppl 85):17-25.
13. Cibella F, Ferrante G, Cuttitta G, et al. The burden of rhinitis and rhinoconjunctivitis in adolescents. Allergy Asthma Immunol Res 2015;7:1:44-50.
14. Larenas-Linnemann D, Pfaar O. Patient-reported outcomes and quality-of-life questionnaires in the assessment of rhinoconjunctivitis in childhood. Curr Opin Allergy Clin Immunol 2014;14:192.
15. Klossek JM, Annesi-Maesano I, Pribil C, Didier A. The burden associated with ocular symptoms in allergic rhinitis. Int Arch Allergy Immunol 2012;158:411-417.
16. Schoemmell E, Gomes P, Welch D. Ocular allergy 2013: A survey of current trends. Invest Ophthalmol Vis Sci 2013;54:2552.
17. Small M, Piercy J, Demoly P, Marsden H. Burden of illness and quality of life in patients being treated for seasonal allergic rhinitis: A cohort survey. Clin Translational Allergy 2013;3:33.
18. Meltzer EO, Rosen Farrar J, Sennett C. Findings from an online survey assessing the burden and management of seasonal allergic rhinoconjunctivitis in US patients. J Allergy Clin Immunol Pract 2016;In Press.



