Off-label Defined
An understanding of the issues of off-label drug use requires a review of the FDA label process. The FDA is a public-health agency whose mission is to oversee the use and marketing of regulated medical products. The FDA reviews clinical research presented in an Investigational New Drug (IND) application and, when appropriate, approves a drug for use and marketing. When a drug is approved, the FDA issues a label that describes and defines the drug's specific medical indication, dose, dosage form, side effects and chemical structure. The format of the FDA label has received widespread criticism for its confusing and almost impenetrable structure. Recently, FDA announced a more "user-friendly" format designed to facilitate use by both patients and physicians.2
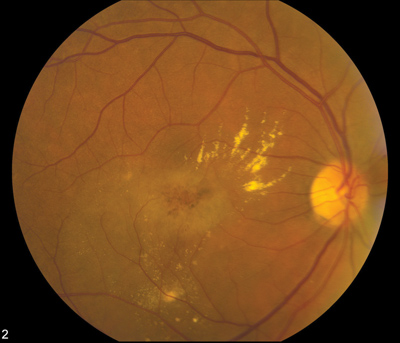 |
| The approval of photodynamic therapy for a limited range of choroidal neovascularization was a big step in bringing the concept of "off-label" treatment to ophthalmologists. |
Any use of a drug not listed on the label is considered off-label. Off-label uses include giving an approved drug for a disease or indication other than the disease for which it is approved; at a different dose, frequency or route of administration than specified in the label; or to treat a child when the product is approved to treat adults. Although the FDA label has important marketing implications, the use of an approved product by physicians is not restricted by the FDA to the limitations of the label. Physicians are allowed to use FDA-approved drugs in the treatment of a specific patient as medical practice. The FDA recognizes that off-label use of drugs by physicians is often appropriate and may represent the standard of care. The most obvious example of off-label drug use constituting the standard of care in ophthalmology is the use of intravitreal antibiotics for endophthalmitis despite the fact that no antibiotics are approved for intravitreal injection.
The FDA has issued specific guidance on off-label drug use as follows:
"Good medical practice and the best interests of the patient require that physicians use legally available drugs, biologics and devices according to their best knowledge and medical judgment. If physicians use a product for an indication not in the approved labeling, they have the responsibility to be well-informed about the product, to base its use on firm scientific rationale and on sound medical evidence and to maintain records on the product's use and effects. Use of a marketed product in this manner when the intent is in the practice of medicine does not require the submission of an Investigational New Drug Application … or review by an Institutional Review Board (IRB). However, the institution at which the product will be used may, under its own authority, require IRB review or other institutional oversight."2
If the FDA recognizes off-label drug use as appropriate and if the agency does not regulate medical practice in the treatment of an individual patient, what role does the FDA play in off-label drug use? One important role is that the FDA precludes drug sponsors from marketing drugs to physicians or patients for off-label use. "Pharmaceutical manufacturers cannot proactively discuss off-label uses, nor may they distribute written materials (promotional pieces, reprints of articles, etc.) that mention off-label use."2 However, the FDA does not restrict other parties such as physicians or specialty societies from discussing off-label uses or distributing written materials concerning them.
Investigational Use
The FDA also becomes involved in off-label drug use when the drug is in "investigational use." The term investigational use suggests the use of an approved drug in the context of a clinical study protocol. When the primary intent of the investigational use is to develop information about a drug's safety or efficacy, or if the off-label use involves a route of administration or dosage level or use in a subject population or other factor that significantly increases the risks associated with the use of the drug, submission of an IND is required.3 Physicians can obtain the IND application (form 1571) from the Internet.2 The application process is self-explanatory, though rather tedious.
The legal implications of off-label drug use primarily involve risk management issues. The keys to addressing these risk management issues are patient selection and informed consent. As discussed above, FDA approval status does not necessarily define appropriate medical practice nor does the FDA regulate medical practice. Medical practice is the therapeutic relationship between a physician and an individual patient. The physician must decide what is appropriate medical care for each patient and this decision must fall within the standard of care. The question then becomes "when does off label drug use become the standard of care?" The answer depends on who is defining the standard of care. Payers may use specific definitions of the standard of care to establish coverage determinations, which will be discussed later. From a risk-management perspective involving malpractice, a jury, judge or arbitrator determines whether treatment is within the standard of care. This determination is based upon such factors as supporting authoritative literature, expert consensus, scientific rationale and local or national medical practice patterns.
Informed Consent
An important factor in establishing the standard of care is informed consent. This is particularly critical with off-label drug use. OMIC, the Ophthalmic Mutual Insurance Co., recommends that physicians inform patients about the off-label status of a proposed treatment.4 This discussion should include the known and potential unknown risks as well as the rationale for the off-label drug use. The physician should also discuss why any available FDA approved or CMS covered therapies are not appropriate. This discussion should be well-documented in the chart. OMIC also recommends that a specific off-label informed consent be used. OMIC provides a specific consent for bevacizumab (Avastin) with useful suggestions concerning patient selection, preparation of the medication, and informed consent and documentation.4
Perhaps the most difficult issue concerning off-label drug use concerns coverage for the drug and associated services. In ophthalmology, this often means coverage by Medicare. Coverage by Medicare can be established either by the local Medicare carrier via a local carrier decision (LCD) or by CMS as a National Coverage Determination (NCD). The Medicare Benefit Policy Manual provides that individual carriers may establish on a case-by-case basic coverage for off-label drug use based upon their assessment of the medical indication as reasonable and necessary.
"FDA approved drugs for indications other than what is indicated on the official label may be covered under Medicare if the carrier determines the use to be medically accepted, taking into consideration the major drug compendia, authoritative medical literature and accepted standards of medical practice …," according to the agency.5
Carrier Criteria
Some carriers have outlined criteria they consider when determining coverage of off-label drug use on a case-by-case basis. AdminiStar, a Medicare carrier, considers the following factors:
• whether alternative treatments were tried before considering off-label use;
• whether there are published recommendations from specialty societies or in other authoritative guidelines;
• whether authoritative medical literature supports the off-label use; (Such literature must include peer-reviewed publications that demonstrate efficacy. Such studies may not include case reports, opinions, book chapters or abstracts.)
• whether there is other evidence of broad scientific support; and
• whether the use is an accepted standard of medical practice.6
Less Clear-Cut Cases
For some off-label drug use such as the use of intravitreal antibiotics for endophthalmitis, the above criteria are easily satisfied. For others, such as intravitreal triamcinolone for retinal disease, fulfillment of the criteria is perhaps not as clear, and for still others such as intravitreal bevacizumab (Avastin, Genentech), the above criteria are simply not met.
CMS can establish a national coverage mandate to all Medicare carriers through a National Coverage Determination (NCD). CMS uses an NCD to determine whether an item or service is "reasonable and necessary." While there is no statutory or regulatory definition of "reasonable and necessary," CMS has generally interpreted the term to mean that the item or service should improve health outcomes for Medicare beneficiaries.7 CMS may consider an NCD when:
• There are questions about the safety, effectiveness or appropriateness of a therapy including off-label use of drugs.
• Local coverage policies are inconsistent.
• There is wide variation in billing practices not related to variation in clinical need.
• The health technology represents a substantial clinical advance and is likely to result in significant health benefits if it diffuses more rapidly to all patients for whom it is indicated.
To address the previous issues, CMS relies on generally accepted principles of evidenced-based medicine.7 For drug coverage, an NCD typically requires strong evidence from clinical trials on safety and efficacy for the indication.
Interestingly, FDA approval of the indication is not required for CMS coverage. This is the case for the coverage of photodynamic therapy with verteporfin for the treatment of occult with no classic and minimally classic subfoveal choroidal neovascularization and evidence of recent disease progression.1 This indication is not listed in the verteporfin FDA label. This NCD, however, is based on multiple clinical trials, independent review by CMS of the clinical trial data and the recommendation of a Medicare Carrier Advisory Committee. Such a process typically takes a year or longer. At present, CMS believes there is insufficient data to warrant an NCD for either off-label triamcinolone or bevacizumab. (Personal communication CMS coverage team.) CMS therefore has deferred these coverage decisions to the local carriers.
New Mechanism
A relatively new mechanism by which CMS may cover off-label drug use is through a clinical trial. To be eligible for coverage, the clinical trial must meet a variety of requirements including:
• The principal purpose of the trial should be to test whether the intervention improves the participant's health outcome.
• The trial should either be well-supported by available scientific and medical information, or be intended to clarify or establish the health outcomes of interventions already in common clinical use.
• The trial must not duplicate existing studies.
• The trial must be appropriately designed and sponsored by a credible organization.
One obvious way to conduct such a trial is through the National Eye Institute, and such trials on off-label drug use are either in progress or preparation.
At the time of this writing, most off-label drug use in ophthalmology is at the discretion of the local Medicare carriers. To date, no carrier has committed in an LCD to cover any off-label drug use in ophthalmology. Usually, coverage will hinge on whether the carrier considers the off-label drug use reasonable and necessary. The Medicare Carrier Manual states that if a medication is determined not to be reasonable and necessary, the carrier excludes the entire charge, including any charges for services that were primarily for the purpose of administering a non-covered medication. This means that for intravitreal injection of a therapeutic agent (CPT code 67028), if the drug is not covered, then neither is the injection.
Considering the above aspects of off-label drug coverage by Medicare, how should ophthalmologists proceed when they believe off-label drug is in a patient's best interest? At present, ophthalmologists should seek specific guidance from their local Medicare carrier. If the carrier declines coverage, the ophthalmologist may bill the patient for non-covered services associated with the off-label drug use by using an Advance Beneficiary Notice (ABN). The necessary ABN form is available on the CMS website, cms.medicare.gov. The use of an ABN requires that the patient be informed concerning why the proposed treatment is not covered by Medicare and that the ABN form be signed prior to the services. The ABN form is then submitted to the local Medicare carrier with the bill on form 1500 using either the GA or GY modifier. Physicians should contact their local carrier to determine which modifier is preferred and to confirm how the carrier handles the ABN process. With an appropriate ABN, physicians can bill the patient for the non-covered services.
As new and effective drugs are introduced for retinal disease, off-label drug use will escalate as ophthalmologists attempt to optimize treatment outcomes. With this increase in off-label drug use comes the responsibility to establish safety and efficacy in order to allow off-label drugs to become the standard of care. Our patients deserve nothing less.
Dr. Williams is chairman of the Department of Ophthalmology and director of the Beaumont Eye Institute, William Beaumont Hospital. He is a clinical professor of biomedical sciences at the Eye Research Institute, Oakland University, Rochester, Mich. Contact him at (248) 551-2176, (248) 551-4362, or gwilliams@beaumont.edu.
1. CMS Manual System. Pub 100-03. Medicare National Coverage Determinations. April 1, 2004. Change request 3191.
2. www.fda.gov.
3. Feinsod M, Chambers WA. A primer on successfully navigating the waters of the Food and Drug Administration. Ophthalmology 2004;111:1801-1806
4. www.omic.com.
5. U.S. Food and Drug Administration. Off-label and investigational use of marketed drugs. Biologics and medical devices. www.fda.gov/oc/ohrt/irbs/off-label.html.
6. Off-label use of non-oncology drugs and biologicals LCD No. L18709 AdminiStar Federal, Inc (February 10, 2005).
7. CMS Draft Guidance for NCDS's, cms.gov March 9, 2005.



