Spasms of the lid and face can significantly impact a patient’s quality of life, but getting to the root cause of the spasm—it can be myokymia, benign essential blepharospasm or hemifacial spasm—can be a challenge for the clinician. In this article, we’ll provide advice that can help you diagnose and manage these sometimes challenging conditions.
An Overview
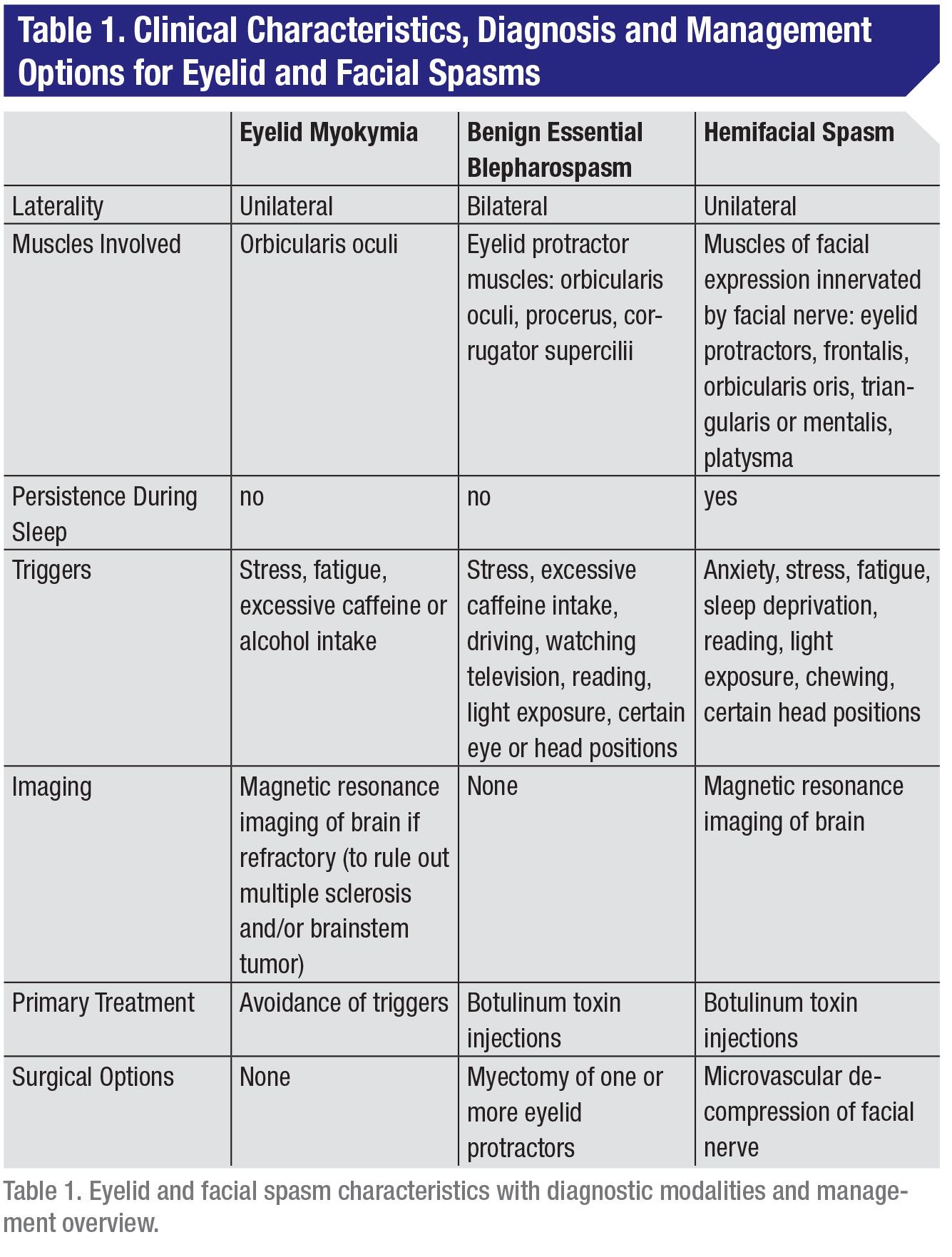
Within neurologic and ophthalmic practice, ranked from least to most severe, the spectrum of eyelid and facial spasms includes eyelid myokymia (involuntary, small contractions of the lid), benign essential blepharospasm (involuntary spasms that cause the eye to close partially or completely) and hemifacial spasm (spasms in the muscles controlling facial expressions) (Table 1). These disorders all consist of involuntary facial movements that can be difficult for patients to deal with in daily life.
Eyelid Myokymia
The most common involuntary facial movement disorder is eyelid myokymia. Eyelid myokymia consists of involuntary, fine, continuous, undulating, non-synchronous contractions of the striated muscle fibers of the eyelid protractors. In electrophysiology, it’s characterized by spontaneous asynchronous discharge of adjacent motor units in doublets or triplets at a rate of 30 to 70 Hertz with intervals of 100 to 200 ms separating individual discharges.2,3 Eyelid myokymia is considered a benign, self-limiting process that’s unilateral and intermittent, with the lower eyelid affected more than the upper lid. Episodes are transient, lasting anywhere from a few days to a few weeks or months, with spasms occurring intermittently throughout the day for up to several hours at a time.4
Inciting factors include stress, exhaustion, excessive caffeine intake or alcohol use, and physical exertion.5 Elimination of these triggers is recommended as part of management. Eyelid myokymia is most commonly isolated to the orbicularis oculi muscle, but may spread to additional muscles of one or both sides of the face, in which case it is referred to as facial myokymia.2
Facial myokymia is caused by damage to the facial nerve nucleus in the pons from demyelinating diseases such as multiple sclerosis or compression from brainstem tumors. Rarely, persistent eyelid myokymia has been reported as a presenting sign of multiple sclerosis or a brainstem tumor.3 If the condition is chronic, refractory to elimination of inciting factors and affects the patient’s quality of life, botulinum toxin injections can be successful in treatment. The mechanism and efficacy of botulinum toxin injections is discussed extensively in the following section. Persistent eyelid myokymia refractory to the above treatments should raise suspicion for a brainstem lesion as described above and warrants analysis with magnetic resonance imaging.
Benign Essential Blepharospasm
BEB was first described in 1857 as a disorder of involuntary spasms of the eyelid protractor muscles that results in partial or complete eyelid closure. BEB has an incidence of 1.4 to 13.3 cases per 100,000, affects females three times more often than men, and most often occurs in the fifth to seventh decades of life.7,8 BEB is most commonly a sporadic disorder, but 27 percent of patients have been reported to have a family member with dystonia, suggesting a possible genetic component.1
The pathophysiology of BEB isn’t well understood. Positron emission tomography and functional magnetic resonance imaging studies have shown activation of brain regions involved in the control and regulation of the muscles responsible for blinking, including the thalamus, striatum, anterior visual cortex, primary motor cortex and superior cerebellum.7,9Animal studies have shown that dopamine depletion in the substantia nigra decreases inhibition of the trigeminal blink reflex leading to oversensitivity of this reflex in response to light and dryness.10 Inciting factors for BEB include stress, excessive caffeine intake, driving, watching television, reading, bright lights, polluted air, wind, noise, and certain eye and head movements.11,12
The clinical presentation of BEB consists of bilateral involuntary spasms of one or more of the eyelid protractor muscles including the orbicularis oculi, procerus and corrugator muscles, with symptoms ranging from mildly increased blink rate to forceful eyelid closure that can result in functional blindness.1 BEB symptoms don’t persist during sleep and it’s a progressive condition in which additional muscles become involved over time.7 A subset of patients may experience mid or lower-facial spasms, a condition termed Meige syndrome.1 An additional subset of patients may develop apraxia of eyelid opening in which there is a loss of co-inhibition between eyelid protractors and retractors resulting in a nonparalytic inability to open the eyelids in the absence of muscle spasm.1
Patients also often complain of the sensory symptom of photophobia. Proposed mechanisms for photophobia include a sympathetically maintained pain disorder, for which superior cervical ganglion blocks have provided relief in prior reports.11 Additional management options to control photophobia include photochromatic modulation with FL-41 tinted lenses or scleral contact lenses with fluorescein in the fluid reservoir, both of which increase the intensity of light tolerated by BEB patients.13
The treatment of choice for BEB is botulinum toxin injections into the affected eyelid protractor muscles. Botulinum toxin inhibits the release of acetylcholine from the presynaptic terminal of the neuromuscular junction, blocking an injected muscle’s ability to contract. There are seven serotypes of botulinum toxin (A through G). Types A and B are U.S. Food and Drug Administration-approved for clinical use in the United States. OnabotulinumtoxinA (Botox) was approved by the FDA for BEB treatment in 1989 and is the most commonly used formulation, though incobotulinumtoxinA, abobotulinumtoxinA, and rimabotulinumtoxinB are also licensed for use.14 Injections are required approximately every three to four months.11 The FDA recommends an initial dose of 1.25 to 2.5 units injected into each affected site with a maximum dose of 15 units and three injection sites per side.14 However, surveys have showed that oculoplastic surgeons tend to treat BEB with an average initial dose of 22.5 ±9.5 units.14
Typically, five to eight sites are injected around each eye, with no more than 0.1 ml of botulinum toxin injected at each site to prevent diffusion of the drug into adjacent muscles.12 Clinicians must take care to inject medially and laterally when treating the pretarsal portion of the orbicularis oculi muscle in the upper eyelids, in order to avoid injection into the levator palpebrae superioris muscle, which can result in ptosis. Similarly, in the lower eyelid, injections are carried out centrally and laterally in order to avoid injection into the inferior oblique, which would result in diplopia. The incidence of adverse effects of botulinum toxin injections for BEB patients has been reported to be around 20 percent, including ecchymosis at the injection site, dry eye, tearing, foreign body sensation, lagophthalmos, diplopia and ptosis.7
Other medications that have been used in the treatment of BEB include antipsychotics, antiepileptics, anxiolytics, antidepressants, antihistamines, sedatives and stimulants, though none have been shown to have long-term efficacy.15 In particular, dopamine agonists and dopamine uptake inhibitors have been shown to be effective in reducing eyelid spasms given the fact that dopamine deficiency has been implicated in the pathogenesis of BEB.11 Methylphenidate, which blocks presynaptic reuptake of dopamine and norepinephrine, has been shown to decrease eyelid spasms and disability scoring.11 BEB patients have shown partial response to gamma-aminobutyric acid agonists such as benzodiazepines, but their use is limited by the side effect of drowsiness.11
When BEB is refractory to medical management or a patient can’t tolerate botulinum toxin injections, surgical intervention with myectomy may be considered. The primary goals of this procedure are to reduce spasm severity and to increase the time interval needed between botulinum injections.11 In surgical myectomy, one or more of the protractor muscles are resected. A limited myectomy involves resection of only part of the orbicularis oculi muscle in the upper eyelid, versus an extended myectomy in which the corrugator supercilii and protractor are additionally removed.15
Adverse effects of surgery include poor cosmesis and need for additional surgeries. Recurrence of symptoms after surgical myectomy has been reported to range from 30 to 50 percent of cases after six months.12 An alternative procedure involves sectioning of the facial nerve, which effectively denervates the eyelid protractors but secondarily results in facial nerve palsy and so is rarely used.15
 |
 |
Hemifacial Spasm
Lastly, HFS is a unilateral involuntary facial movement disorder involving spasms of the muscles of facial expression innervated by the facial nerve. Estimated incidence is approximately 10 cases per 100,000, based on studies in Minnesota and Norway.16,17 HFS tends to occur in the fourth or fifth decade of life, with females being affected two to three times more often than men, and Asian populations more commonly affected than Caucasians.18 Up to one-third of patients report aggravating factors for HFS, including anxiety, stress, fatigue, sleep deprivation, reading, light exposure, chewing or particular head positions.18 HFS is often misdiagnosed as functional or psychogenic (in 38 percent of cases), tics (in 29 percent) and facial nerve palsy (in 9 percent).19 Clinical presentation involves initial spasms of the orbicularis oculi which gradually progress or spread over time to involve other muscles on one half of the face, such as the frontalis, orbicularis oris, triangularis or mentalis, and even the platysma muscle.7,18 Symptoms of HFS may persist during sleep, whereas those of BEB do not.
The pathophysiology of HFS involves compression of the facial nerve at its root exit zone (REZ) from the brainstem, which was first described in 1947.20 Compression may be caused by aberrant vessels, including the anterior inferior cerebellar artery, posterior inferior cerebellar artery, basilar artery, vertebral artery; arteriovenous malformations; and very rarely by tumors such as acoustic schwannomas, meningiomas, parotid gland tumors, and pilocytic astrocytomas.7 While diagnosis of HFS is primarily clinical, MRI should always be obtained to rule out facial nerve compression as described above. Electromyography testing may also be carried out to demonstrate a pathognomonic lateral spreading response of impulses between neighboring fibers of the facial nerve (“ephaptic transmission”).20
The mainstay of treatment for HFS are botulinum toxin injections as described above for BEB. For HFS, five to 10 sites are injected on the affected side of the face with total doses of onabotulinumtoxinA ranging from 10 to 34 units per treatment.20 The definitive treatment for HFS is neurosurgical microvascular decompression (MVD) to relieve facial nerve compression at its REZ. In prior studies, approximately 95 percent of patients who underwent MVD for HFS achieved good or even excellent results, with adverse effects including facial nerve palsy in 19 percent, hearing deficits in 7 percent, and lower cranial nerve palsies in 2.8 percent.21 Given the invasiveness and risks associated with undergoing neurosurgical intervention, it’s of the utmost importance to have a frank discussion with patients regarding the risks and benefits of MVD versus botulinum toxin injections, or a combination of the two, for management of HFS.
| The definitive treatment for hemifacial spasm is neurosurgical microvascular decompression (MVD) to relieve facial nerve compression at its root exit zone. |
In conclusion, eyelid and facial spasms represent a spectrum of involuntary facial movement disorders that can have a severe impact on patients’ quality of life and psychological wellbeing. While avoidance of inciting factors is recommended in management, botulinum toxin injections have emerged as first-line treatment, particularly for BEB and HFS. Surgical therapy for BEB and HFS, however, may be pursued in refractory cases or in patients intolerant of botulinum toxin injections. Given the frequent misdiagnosis of these disorders, it’s important for physicians to be familiar with the most common presenting signs and symptoms in order to connect these patients with neurologists or ophthalmologists for further evaluation and management. REVIEW
Dr. Gervasio is an ophthalmology resident at Wills Eye Hospital. Dr. Moster is an attending physician on the Neuro-Ophthalmology Service at Wills and professor of neurology and ophthalmology at Thomas Jefferson University in Philadelphia. He is chair of neuro-ophthalmology at Einstein Medical Center in Philadelphia.
1. Hwang CJ, Eftekhari K. Benign essential blepharospasm: What we know and what we don’t. Int Ophthalmol Clin 2018;58:1:11-24.
2. Miller NR. Eyelid myokymia. Surv Ophthalmol 2011;56:3:277-8.
3. Barmettler A, Dinkin MJ, Lelli GJ. Eyelid myokymia: Not always benign. Orbit 2011;30:6:289-90.
4. Jordan DR, Anderson RL, Thiese SM. Intractable orbicularis myokymia: Treatment alternatives. Ophthalmic Surg 1989;20:4:280-3.
5. Horowitz SH. Hemifacial spasm and facial myokymia: Electrophysiological findings. Muscle Nerve 1987;10:422-427.
6. Mackenzie W. Case of intense and long-continued photophobia and blepharospasm, relieved by the inhalation of chloroform. Med Chir Trans 1857;40:175-178.
7. Ozzello DJ, Giacometti JN. Botulinum toxins for treating essential blepharospasm and hemifacial spasm. Int Ophthalmol Clin 2018;58:1:49-61.
8. Bradley EA, Hodge DO, Bartley GB. Benign essential blepharospasm among residents of Olmsted County, Minnesota, 1976-1995: An epidemiologic study. Ophthal Plast Reconstr Surg 2003;19:177-181.
9. Baker RS, Andersen AH, Morecraft RJ, et al. A functional magnetic resonance imaging study in patients with benign essential blepharospasm. J Neuroophthalmol 2003;23:11-15.
10. Schicatano EJ, Basso MA, Evinger C. Animal model explains the origins of the cranial dystonia benign essential blepharospasm. J Neurophysiol 1997;77:2842-2846.
11. Yen MT. Developments in the treatment of benign essential blepharospasm. Curr Opin Ophthalmol 2018;29:440-44.
12. Coscarelli JM. Essential blepharospasm. Semin Ophthalmol 2010;25:3:104-8.
13. Herz NL, Yen MT. Modulation of sensory photophobia in essential blepharospasm with chromatic lenses. Ophthalmology 2005;112:2208-2211.
14. Broadbent TJ, Wesley RE, Mawn LA. A survey of current blepharospasm treatment patterns among oculoplastic surgeons. Ophthalmic Plast Reconstr Surg 2016;32:1:24-7.
15. Ben Simon GJ, McCann JD. Benign essential blepharospasm. Int Ophthalmol Clin 2005;45:49-75.
16. Auger RG, Whisnant JP. Hemifacial spasm in Rochester and Olmsted County, Minnesota, 1960 to 1984. Arch Neurol 1990;47:1233-34.
17. Nilsen B, Le KD, Dietrichs E. Prevalence of hemifacial spasm in Oslo, Norway. Neurology 2004;63:1532-33.
18. Lefaucheur JP, Daamer NB, Sangla S, Guerinel CL. Diagnosis of primary hemifacial spasm. Neurochiurgie 2018;64:2:82-86.
19. Martinez AR, Nunes MD, Immich ND, et al. Misdiagnosis of hemifacial spasm is a frequent event in the primary care setting. Arq Neuropsiquiatr 2014;72:119-22.
20. Campbell E, Keedy C. Hemifacial spasm: A note on the etiology in two cases. J Neurosurg 1947;4:342-47.
21. Huh R, Han IB, Moon JY, et al. Microvascular decompression for hemifacial spasm: Analyses of operative complications in 1582 consecutive patients. Surg Neurol 2008;69:153-57.



