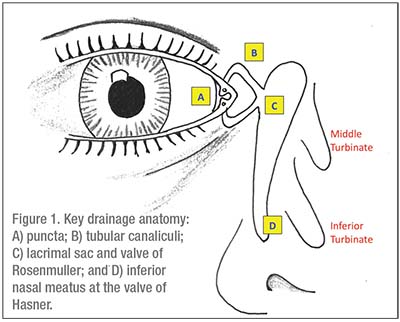
Review of Anatomy
Epiphora results from obstruction at any level of the lacrimal drainage apparatus. After tears accumulate in the tear lake, they’re
 |
Acquired nasolacrimal duct obstruction is most commonly due to inflammation and obstructive fibrosis, but may be secondary to more dangerous processes. Thus, careful history and clinical evaluation is essential to rule out etiology such as trauma, prior surgery, infection, systemic inflammatory conditions or neoplasms. When a cause can’t be identified, the term primary acquired nasolacrimal duct obstruction (PANDO) is used. In one study, Nancy Tucker, MD, and her colleagues at Montreal’s McGill University performed lacrimal sac biopsies in 150 patients undergoing DCR for clinically suspected PANDO. Histopathologic findings for 147 patients were consistent with inflammation or fibrosis, and only three had identifiable underlying etiology (sarcoidosis, oncocytoma and lymphoma).
Locating the Blockage
Assuming a normal blink and anterior segment exam, patency of the lacrimal drainage system must be assessed. You can localize the obstruction by systematic testing. Palpation of the lacrimal sac can reveal distention or expression of fluid from the puncta, thus diagnosing obstruction, patent canaliculi and blockage proximal to the lacrimal sac or nasolacrimal duct. Further probing should be avoided to prevent iatrogenic scarring. Significant inflammation or infection of the sac can signify dacryocystitis due to stasis in the setting of nasolacrimal duct obstruction. Severe cases can be associated with preseptal cellulitis, orbital cellulitis or systemic infection, and these patients should be admitted for IV antibiotics.
Incision and drainage may be necessary to control acute infection, and surgical correction of underlying nasolacrimal duct obstruction should be considered after treatment of the acute infection. (The presence of dacryocystitis is diagnostic and additional probing isn’t necessary, particularly during acute infection.)
If the lacrimal sac is normal, patency can be further assessed using the fluorescein dye disappearance test (FDDT), Jones I test, Jones II test and/or lacrimal probing and irrigation. Radiographic techniques such as lacrimal scintillography and dacryocystography are options but are rarely employed clinically. There is no standard testing algorithm, and a poll of members of the American Society of Ophthalmic Plastic and Reconstructive Surgery showed a little less than half of the surgeons relied primarily on lacrimal irrigation, 21 percent used Jones Testing, 19 percent used the FDDT and 13 percent used Schirmer’s testing.
To perform the FDDT, instill one drop of fluorescein in the inferior cul-de-sac, wait five minutes, then reassess the patient. Minimal fluorescein in the tear lake indicates a patent drainage system, and retained fluorescein indicates impaired drainage. This is particularly useful in unilateral cases, where the asymptomatic eye can be used as a control. While useful conceptually, few clinicians perform Jones testing, and most rely on the FDDT and lacrimal irrigation alone. Lacrimal irrigation starts
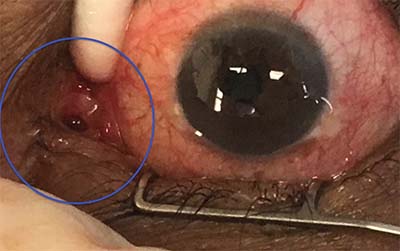 |
| Figure 2. The Jones tube is designed for easy removal, since it’s only held in place by friction and a small collar. |
Canalicular Obstruction
Once the obstruction is located, management consists of bypassing or recanalizing the site of obstruction. Distal obstructions involving the canaliculi can be particularly challenging, and often require completely circumventing the lacrimal drainage apparatus. Conjunctivodacryocystorhinostomy (CDCR) with placement of a glass Jones tube is the gold standard in management. This procedure connects the tear lake and the nasal cavity through a surgical ostomy, and relies on a glass tube to maintain a patent conduit over time. Success rates range as high as 90 percent, but patients may have difficulty tolerating and maintaining the Jones tube. By design, the Jones tube is easily removed to accommodate the frequent cleaning necessary to prevent blockage and biofilm development. It’s held in place simply by friction and a small collar to prevent inward migration (See Figure 2), and consequently tube dislocation and extrusion can occur in up to half of patients. Patients may also experience recurrent plugging, malposition, difficulty achieving the desired rate of tear outflow, and retrograde airflow when sneezing or using a CPAP.
The high rate of complications should be considered before CDCR, and alternate methods have been attempted in an effort to spare patients the burden of a Jones tube. In cases of distal obstruction with at least 8 mm of patent upper or lower canaliculus continuous with the punctum, canaliculo-DCR may be attempted. For this procedure, external dacryocystorhinostomy is performed as described in the next section, followed by probing of the canaliculi to the point of blockage. The obstruction is resected, and the patent canaliculus is anastomosed directly into the lacrimal sac. Results are best with smaller obstructions and those located near or within the common canaliculus. Long-term success of this technically challenging procedure is rare and these cases often require subsequent CDCR with Jones tube.
As an alternative to bypassing the obstruction, a blocked canaliculus can be recanalized using trephination. For this procedure, a hollow tube with a cutting edge inside a protective stylus is advanced into the canalicular system. When an obstruction is encountered, the stylus is withdrawn and trephination is performed with gentle rotation of the instrument. This is continued until the instrument reaches the hard lacrimal fossa, taking extreme caution to avoid creation of a false passage along the way. Success rates may vary depending on the location of obstruction and surgeon experience. One recent study reported functional success of 78 percent and anatomical success of 84 percent at six months when combined with endoscopic DCR and silicone tube stenting. Anatomic success increased to 95 percent for surgeons with greater than five years of experience. This method has been performed as an initial surgical intervention or after a previously failed DCR.
Nasolacrimal Duct Obstruction
Dacryocystorhinostomy is the mainstay of treatment for nasolacrimal duct obstruction without canalicular involvement. DCR creates a channel between the lacrimal sac and nasal cavity, and can be performed externally or endonasally.
The external DCR procedure begins with an initial incision at the level of the medial canthal tendon, which is extended 15 to 20 mm inferolaterally along the side of the nose. Blunt dissection is used to expose the periosteum, which is reflected to expose the intact lacrimal sac. After lifting the sac out of the lacrimal fossa, an ostomy is created in the lacrimal fossa to access the nasal mucosa, which is anastomosed to the lacrimal sac mucosa. A successful DCR connects a patent canalicular system to the nasal canal at the level of the lacrimal sac, bypassing any additional nasolacrimal duct obstruction proximally.
Endonasal DCR accomplishes the same task, but accesses the lacrimal sac through the nasal cavity. The endonasal approach offers certain advantages such as the absence of an external scar, lower complication rate, shorter operative time and complete preservation of the pump function by avoiding any manipulation of the orbicularis oculi. However, endonasal DCR is often
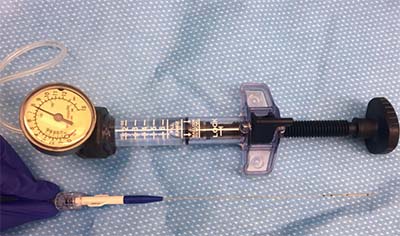 |
| Figure 3. Balloon catheter dilation can be effective in some patients suffering from nasolacrimal duct obstruction. |
To Stent or Not to Stent
Monocanalicular or bicanalicular silicone tube stents may be placed during external or endonasal DCR surgery. Several methods have been attempted to prevent ostial closure, with limited efficacy. Success rates of DCR without stenting are excellent, and studies have failed to show any clear benefit of stenting. Furthermore, the practice is not entirely benign, and stent-related complications include prolapse, corneal abrasion, canaliculitis, adhesions, false passage creation during intubation and cheese wiring of the puncta.
Alternative Modalities
While DCR is the gold standard, other procedures aimed at recanalizing the nasolacrimal duct have been studied. Some success has been reported using balloon catheter dilation to treat acquired partial NLDO in patients (See Figure 3). In a small, prospective study, 60 percent of patients reported subjective improvement, and 73 percent were objectively patent upon irrigation at six months. NLD probing with adjuvant mitomycin C has also been attempted, with one study reporting 84-percent patency at nine months. A group of Chinese surgeons reports using high-frequency electric cautery to recanalize the NLD, with a reported 87 percent success rate. While the evidence is far from conclusive, these minimally invasive techniques could offer patients additional options, pending further study.
Adult patients with tearing are challenging to evaluate, but a correct diagnosis is critical for guiding treatment. Surgical management is based on the level of obstruction, and thus a thorough understanding of lacrimal system anatomy and physiology, combined with a good systematic evaluation, are indispensible. Keep in mind that a thorough history may reveal serious underlying pathology requiring additional workup and treatment beyond just the correction of tearing.
Dr. Luther is a current second-year ophthalmology resident at Boston University. Dr. Armstrong is the director of oculoplastics, orbital and reconstructive surgery at Boston University, where she is also an assistant professor of ophthalmology.
Suggested Reading:
1. Cannon PS, Chan W, Selva D. Incidence of canalicular closure with endonasal dacryocystorhinostomy without intubation in primary nasolacrimal duct obstruction. Ophthalmology 2013;120:8:1688-1692.
2. Chen D, Ge J, Wang L, et al. A simple and evolutional approach proven to recanalise the nasolacrimal duct obstruction. Br J Ophthalmol 2009;93:11:1438-1443.
3. Conway ST. Evaluation and management of “functional” nasolacrimal blockage: Results of a survey of the American Society of Ophthalmic Plastic and Reconstructive Surgery. OPRS 1994;10:185-188.
4. Chong KKL, Lai FHP, Ho M, Luk A, Wong BW, Young A. Randomized trial on silicone intubation in endoscopic mechanical dacryocystorhinostomy (SEND) for primary nasolacrimal duct obstruction. Ophthalmology 2013;120:10:2139-2145.
5. Daubert J, Nik N, Chandeyssoun PA, el-Choufi L. Tear flow analysis through the upper and lower systems. Ophthal Plast Reconstr Surg 1990;6:3:193-196.
6. Doane MG. Blinking and the mechanics of the lacrimal drainage system. Ophthalmology 1981;88:8:844-851.
7. Dolmetsch AM. Nonlaser endoscopic endonasal dacryocystorhinostomy with adjunctive mitomycin C in nasolacrimal duct obstruction in adults. Ophthalmology 2010;117:5:1037-1040.
8. Feng Y, Cai J, Zhang J, Han X. A meta-analysis of primary dacryocystorhinostomy with and without silicone intubation. Can J Ophthalmol 2011;46:6:521-527.
9. Foster JA, Carter KD, Durairaj VD, Kavanagh MC, Korn BS, Nelson CC, Hartstein ME. Basic and Clinical Science Course, Section 07: Orbits, Eyelids, and Lacrimal System. San Francisco: Amer Academy of Ophthalmology 2016-2017:251-256, 267-276, 284-285.
10. Jawaheer L, MacEwen CJ, Anijeet D. Endonasal versus external dacryocystorhinostomy for nasolacrimal duct obstruction. Cochrane Database Syst Rev 2017 February 24;2:CD007097.
11. Kalin-Hajdu E, Cadet N, Boulos PR. Controversies of the lacrimal system. Surv Ophthalmol 2016;61:3:309-313.
12. Kashkouli MB, Beigi B, Murthy R, Astbury N. Acquired external punctal stenosis: Etiology and associated findings. Am J Ophthalmol 2003;136:6:1079-1084.
13. Kong YJ, Choi HS, Jang JW, Kim SJ, Jang SY. Surgical outcomes of canalicular trephination combined with endoscopic dacryocystorhinostomy in patients with distal or common canalicular obstruction. Korean J Ophthalmol 2015;29:6:368-374.
14. Liarakos V, Boboridis K, Mavrikakis E, Mavrikakis I. Management of canalicular obstructions. Curr Opin Ophthalmol 2009;20:5:395-400.
15. Linberg JV, McCormick SA. Primary acquired nasolacrimal duct obstruction. A clinicopathologic report and biopsy technique. Ophthalmology 1986;93:8:1055-1063.
16. Nathoo NA, Rath S, Wan D, Buffam F. Trephination for canalicular obstruction: Experience in 45 Eyes. Orbit 2013;32:5:281-284.
17. Patel BC. Management of acquired nasolacrimal duct obstruction: External and endonasal dacryocystorhinostomy. Is there a third way? Br J Ophthalmol 2009;93:11:1416-1419.
18. Patrinely JR, Anderson RL. A review of lacrimal drainage surgery. Ophthal Plast Reconstr Surg 1986;2:2:97-102.
19. Perry JD, Maus M, Nowinski TS, Penne RB. Balloon catheter dilation for treatment of adults with partial nasolacrimal duct obstruction: A preliminary report. Am J Ophthalmol 1998;126:6:811-816.
20. Rose GE, Welham RA. Jones’ lacrimal canalicular bypass tubes: Twenty-five years’ experience. Eye (Lond) 1991;5(Pt 1):13-19.
21. Steele EA. Conjunctivodacryocystorhinostomy with Jones tube: A history and update. Curr Opin Ophthalmol 2016;27:5:439-442.
22. Tsai CC, Kau HC, Kao SC, Hsu WM, Liu JH. Efficacy of probing the nasolacrimal duct with adjunctive Mitomycin-C for epiphora in adults. Ophthalmology 2002;109:1:172-174.
23. Tucker N, Chow D, Stockl F, Codere F, Burnier M. Clinically suspected primary acquired nasolacrimal duct obstruction: Clinicopathologic review of 150 patients. Ophthalmology 1997;104:11:1882-1886.
24. Wearne MJ, Pitts J, Frank J, Rose GE. Comparison of dacryocystography and lacrimal scintigraphy in the diagnosis of functional nasolacrimal duct obstruction. Br J Ophthalmol 1999;83:9:1032-1035.
25. Yeatts P. Acquired nasolacrimal duct obstruction. Ophthalmol Clin North Am 2000;13:4:719.



