Presbyopia by the Numbers
Presbyopia’s incidence and impact are rising in conjunction with the aging of the population, both in the United States and around the world. Now, as the baby boomers transition into their 60s, our aging population continues to expand: By 2030, about one in five Americans will be older than 65. Globally, data collected from 228 countries in 2005 estimated that presbyopia affected more than 1 billion people. By 2050, this number is projected to increase to almost 1.8 billion.1
Although the incidence rates of presbyopia are very high, public awareness of this condition, its origins and what can be done to correct it are surprisingly low. A 1996 study reported that more than half of patients queried did not know the meaning of the word,2 while a more recent survey found that more than eight in 10 American consumers couldn’t provide a definition for presbyopia; of those that could, more than six in 10 mistakenly believed that it could lead to blindness.3
Typically, a patient learns of presbyopia at an age when more frequent ocular health examinations are recommended because of the increased risk of age-related ocular diseases such as glaucoma, cataracts and macular degeneration.4 This timing can have the effect of downplaying the significance and impact of the condition. Once diagnosed, many patients resist seeking treatment due to the stigma associated with aging. Most grudgingly accept the inevitability of reading glasses, but a growing contingent want more information and are interested in exploring treatment options for presbyopia that include contact lenses, LASIK and other approaches.5 A number of surgical options are also available, but results can be variable, and some procedures are contraindicated in certain patient populations. The limitations of current treatment options, together with the growing numbers of affected individuals, have driven increased scientific and clinical interest in presbyopia in recent years.
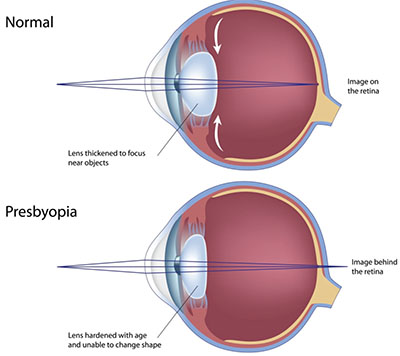 |
| A look at our current understanding of presbyopia’s physiology. |
The Physiology of Presbyopia
The process of accommodation, or focusing on near objects, occurs by a concerted action of the ciliary muscle on the zonule fibers which hold the lens in place. The ciliary muscle is a ring of smooth muscle that, upon contraction, relaxes the tension on the zonular fibers and allows the lens to become more spherical. This increase in axial thickness results in an increase in the dioptric power, facilitating accommodation for improved near vision. With ciliary muscle relaxation, the tension on the zonules increases, resulting in lens flattening and a reduction in dioptric power. All of these structures are modified by the aging process, but it is the reduction in lens flexibility that’s most associated with loss of accommodation.6-8 Decreased lens flexibility limits the lens rounding and thickening needed for near focus. Models of the process suggest that as the lens becomes less flexible with age, ciliary muscles apply greater tension to zonules, causing ligament fatigue. Muscular atrophy is also a contributing factor.9 These paired changes in the physical properties of the focal apparatus collectively result in the presbyopic condition. The end effect is loss of near focus, accompanied by blurred vision, eye strain and headaches in many individuals. For some, these secondary effects are exacerbated after reading or computer use.6
Much of what we know about accommodation comes from research on non-human primates. Studies in the rhesus monkey have shown that ciliary body displacement plays a key role in the accommodation process. The force of contraction moves the muscle in an anterior direction, and this displacement is attenuated in aging animals. One study confirmed the roles of both muscle atrophy and decline in ligament elasticity in the development of presbyopia,9 and another recently showed that similar movement occurs in the human eye.10 Hopefully, these findings can provide clues to pharmacological strategies to reverse or reduce accommodative loss.
The use of contact lenses to address visual changes associated with presbyopia has been an area of great promise but only modest success. The limitation to this approach may be that the great diversity of visual tasking and visual activity among patients has led to limited success for individual approaches including bifocal or progressive lenses, monovision lenses (one eye for distance, one eye for near vision), and reduced-aperture (pinhole) lenses. Development of predictive algorithms may be the key to making progress with this treatment modality.11
Inlays and Surgeries
In recent years, surgery has been the new frontier for presbyopia therapy, by either direct intraocular replacement of the lens or by modification of extraocular structures such as the cornea or sclera.12 Modification involves the use of corneal inlays, devices that are surgically placed within the corneal stroma of the non-dominant eye to change the optical properties of the cornea.12 Inlays serve as an attractive option for correction of presbyopia, since they are an additive, removable technology. Unlike laser refractive surgery, which ablates corneal tissue, the inlay can easily be removed if the patient is unable to adapt to this type of vision correction. The development of corneal inlays has become possible through major advancements made in biomaterials as well as surgical devices such as the femtosecond laser.5
Femtosecond laser technology has numerous advantages over traditional surgical techniques and can be applied to a variety of different ophthalmic procedures. Still, this is a relatively new technology and is limited by training and cost issues.13 Examples of corneal inlays employing femtosecond laser technology include the KAMRA inlay (AcuFocus), which is an opaque polymer ring that employs the pinhole concept to expand the depth of focus and allow for improvement in near visual acuity.5 In one study of 223 presbyopes who first underwent LASIK to correct their pre-existing refractive error, subsequent KAMRA implantation increased their average uncorrected near vision from J8 to J2.14 Another recently approved inlay, the Raindrop Near Vision Inlay (ReVision Optics), is a clear hydrogel implant that increases the anterior corneal curvature to add optical power, with a refractive index approximating that of the cornea.12 Other devices are in development, such as the Presbia Flexivue Microlens (PresbiBio).15
Although the safety of corneal inlays is well-established, the use of some, such as the KAMRA inlay, has been associated with the development of corneal changes.5 The improvement of uncorrected near vision may be accompanied by decreased distance vision, increased glare or halos, reduced contrast sensitivity, and the development of dry-eye symptoms secondary to the creation of the corneal flap/channel for inlay insertion.12 Despite these issues the frequency of explants has been reasonably low.
Another approach to presbyopia is excimer laser surgery, which remodels the corneal curvature to improve uncorrected vision and reduce dependence on glasses or contacts. Approved in 2001, one of the most popular techniques, LASIK, has seen tremendous improvements in technology, which now includes correction of presbyopia, or “presbyLASIK.” While conventional monovision LASIK corrects the dominant eye for distance and the non-dominant eye for near, presbyLASIK involves a number of different approaches.12 Nevertheless, there is no “one size fits all” for presbyopia; each surgical strategy presents its own unique benefits and limitations that involve some degree of compromise between distant and near visual acuities.16 For example, crystalline lens removal and replacement with an IOL may not be preferable in a young patient with presbyopia without refractive error.16 Similarly, treatment of the crystalline lens may not be a suitable choice for a patient with early cataract. Thus, there are a number of considerations that have limited the widespread acceptance of surgical correction, keeping the correction of presbyopia a significant challenge for refractive surgeons.
Pharmacotherapy
Pharmacotherapy for presbyopia is on the rise, as many patients seek freedom from spectacle use without the risk and cost of surgical approaches. For some time, there has been interest in a topical drop that can provide a safe, albeit transient, improvement in accommodation, even if it reduces distance vision. In a recent study, use of miotic parasympathomimetic drugs such as carbachol, alone or in combination with alpha-adrenergic agonists such as brimonidine, was shown to provide acceptable reading vision for presbyopes.17 Many of these drugs have the benefit of a well-established safety profile due to a long history of usage in diagnostics.
Another study, evaluating a cocktail of topical agents that included pilocarpine and phenylephrine, showed an improvement in near vision without a significant effect on distance vision.18
Other research efforts have reached the clinic for study: The California-based startup Presbyopia Therapies is evaluating the efficacy and safety of their proprietary drop PRX-100 in presbyopes.19 PRX-100 has been formulated to induce miosis without stimulation of accommodation, and has been shown to be safe and effective in increasing near vision for up to eight hours. Presbyopia Therapies hopes that PRX-100 will supplement current presbyopia treatments by providing short-term, self-administered correction for daytime near vision.
An interesting alternative to pharmacological modulation of the ciliary apparatus is being pursued by another emerging biotech firm, Encore Vision. Their approach is aimed at directly increasing flexibility of the aging lens. A key aspect of lens rigidity involves formation of disulfide crosslinks between lens protein fibers, and studies have shown a strong positive correlation between lens age and the number of disulfide bonds in lenses.20 In preclinical studies, researchers have shown that treatment of isolated lenses with a lipoic-acid reducing agent can reduce disulfide bonds and increase elasticity.21 This approach holds real promise, especially if the prevailing wisdom that lens inflexibility is the most significant contributing factor in the development of presbyopia proves to be true.
The aging population represents a impending explosion of unmet medical need. Though sight-threatening conditions such as AMD often dominate the conversation of age-related diseases, it’s important to remember that conditions such as presbyopia impact the quality of life of many more individuals. While surgical and device-based treatments for this condition hold promise, it’s also exciting to see the innovations provided by pharmaceutical approaches. REVIEW
Dr. Abelson is a clinical professor of ophthalmology at Harvard Medical School. Dr. Hollander is chief medical officer at the ophthalmic consulting firm Ora, and assistant clinical professor of ophthalmology at the Jules Stein Eye Institute at the University of California, Los Angeles. Dr. Slocum is a medical writer at Ora. Dr. Abelson may be reached at MarkAbelsonMD@gmail.com.
1. Holden BA, Fricke TR, Ho SM, et al. Global vision impairment due to uncorrected presbyopia. Archives of Ophthalmology 2008;126:12:1731-1739.
2. Walline JJ, Zadnik K, Mutti DO. Validity of surveys reporting myopia, astigmatism, and presbyopia. Optom Vis Sci 1996; 73:376-81.
3. Wakefield survey on behalf of Transitions Optical, among a representative sample of more than 2,200 Americans ages 18+. April 2011. Available at: http://pro.transitions.com/NewsEvents/Press%20Releases/nr_Multicultural%20Research.pdf
4. Mancil GL, Bailey IL, Brookman KE, et al. Optometric clinical practice guideline care of the patient with presbyopia. American Optometric Association 2011. Available at: http://www.aoa.org/documents/optometrists/CPG-17.pdf
5. Naroo SA and Bilkhu PS. Clinical utility of the KAMRA corneal inlay. Clinical Ophthalmology 2016;10:913-919.
6. Truscott RJ. Presbyopia. Emerging from a blur towards an understanding of the molecular basis for this most common eye condition. Experimental Eye Research 2009;88:241-247.
7. Scarcelli G, Kim P, Yun SH. In vivo measurement of age-related stiffening in the crystalline lens by brillouin optical microscopy. Biophysical Journal 2011;101:1539-1545.
8. Ostrin LA, Glasser A. Edinger-Westphal and pharmacologically stimulated accommodative refractive changes and lens and ciliary process movements in rhesus monkeys. Exp Eye Res 2007;84:302-313.
9. Kaufman PL. Enhancing trabecular outflow by disrupting the actin cytoskeleton, increasing uveoscleral outflow with prostaglandins, and understanding the pathophysiology of presbyopia. Exp Eye Res 2008;86:1:3-17.
10. Croft MA, McDonald JP, Katz A, et al. Extralenticular and lenticular aspects of accommodation and presbyopia in human versus monkey eyes. Invest Ophthalmol Vis Sci 2013;54:5035.
11. Sivardeen A, Laughton D, Wolffsohn JS. Investigating the utility of clinical assessments to predict success with presbyopic contact lens correction. Cont Lens Anterior Eye 2016;39:5:322.
12. Paley GL, Chuck RS, Tsai LM. Corneal-based surgical presbyopic therapies and their application in pseudophakic patients. Journal of Ophthalmology 2016;doi:http://dx.doi.org/10.1155/2016/5263870
13. Callou TP, Garcia R, Mukai A, et al. Advances in femtosecond laser technology. Clinical Ophthalmology 2016;10:697-703.
14. Tomita M, Kanamori T, Waring GO, et al. Small-aperture corneal inlay implantation to treat presbyopia after laser in situ keratomileusis. J Cataract Refract Surg 2013;39:898-905.
15. https://clinicaltrials.gov/ct2/show/NCT02110472?term=Presbia&rank=1. Accessed 12 Sept 2016
16. Gil-Cazorla R, Shah S, Naroo SA. A review of the surgical options for the correction of presbyopia. Br J Ophthalmol. 2016; 100: 62-70.
17. Abdelkader A. Improved presbyopic vision with miotics. Eye & Contact Lens 2015;41:323-7.
18. Renna A, Vejarano LF, De la Cruz E, Alió JL Pharmacological treatment of presbyopia by novel binocularly instilled eye drops: A pilot study. Ophthalmol Ther 2016;5:1:63–73.
19. https://clinicaltrials.gov/ct2/show/NCT02554396?term=PRX-100&rank=1. Accessed 13 Sept 2016.
20. Lou MF, Dickerson JE. Protein-thiol mixed disulfides in human lens. Exp Eye Res 1992;55:889-896.
21. Garner WH, Garner MH. Protein disulfide levels and lens elasticity modulation: Applications for presbyopia. Invest Ophthalmol Vis Sci 2016;57:2851–2863.



