Myopia is an increasingly prevalent refractive disorder which can ultimately lead to vision loss. An estimated 28.3 percent of the world’s population is myopic and 4 percent of the world population has high myopia (greater than -5 D). This percentage is even higher among the East Asian population.1 High myopia can lead to visual impairment and vision loss from glaucoma, cataracts, retinal detachment and choroidal neovascularization.2 This public health burden is likely to increase over time, with one study projecting that the prevalence of myopia will increase to 49.8 percent by 2050 and high myopia to increase to 9.8 percent.1
Over the past decade, much research has been done on slowing the progression of myopia, and there are now several effective treatment options. This review will highlight the latest treatment options available.
Myopia Progression
Myopia often progresses as a result of axial lengthening. Proposed pathophysiology involves some level of defocus which sets off a chemical cascade of neurotransmitters that signal the eye to elongate. Specifically, peripheral light rays focus behind the retina, creating relative hyperopic peripheral refractive error (hyperopic defocus).3-6 As a result, some optical myopia treatments are designed to stimulate myopic defocus. Other treatments discussed—outdoor sunlight and atropine—are thought to work directly on the neurochemical cascade.7,8
Spectacle Correction
Glasses, utilizing various designs, have been studied for inhibiting myopia progression. In the Control of Myopia Evaluation Trials, COMET and COMET 2, protocols were based on the theory that myopic progression could be controlled by relaxing accommodative effort. However, these studies showed that progressive addition lenses were not clinically effective at slowing progression.9,10
A recent study from Hong Kong showed some success with a different spectacle design. A lens called the Defocus Incorporated Multiple Segments (DIMS) spectacle lens is designed to induce myopic defocus. This lens has a central 9-mm zone for distance viewing, with multiple segments of +3.5-D defocus surrounding the central zone. The design is based on the theory of creating peripheral defocus (i.e., myopic defocus) to inhibit myopic progression instead of relaxing the accommodative system. Although this study was done solely on Chinese children, the results from a two-year, randomized trial were promising: Axial myopia progression was reduced by 0.55 ±0.09 D.11
This study’s generalizability is limited due to its homogenous patient sample, and further studies are needed to determine the lens’ true ability to limit myopia progression across ethnic populations. Since parents tend to understand the benefits of glasses and myopic children already wear them, it’s possible that a myopia-control spectacle lens may be met with less resistance than other forms of treatment.
Outdoor Sunlight
Time spent outdoors has been shown to have a modest effect on reducing myopia progression. One study in a Chinese population showed that an increase of 40 minutes/day of outdoor time during school was enough to have an effect: The cumulative incidence of myopia was 39.5 percent in the control group vs. only 30.4 percent in the intervention group after three years. In children who already had myopia, however, the change in refractive spherical equivalent was only 0.17 D between groups, with no difference in axial length.12 A separate one-year study done in Taiwan showed a modest effect (a reduction of 0.12 D) with 11 hours per week of increased outdoor time. This study also demonstrated myopia control, even in a lower-light-intensity environment.13
 |
Although incorporating outdoor sunlight is cost-effective and easy for parents to understand, the implementation has been challenging. In the studies, even though parents were instructed to increase outdoor time at home, the compliance was poor. In addition, the effect of myopia control was modest in the three-year study. These studies also have limited generalizability due to their patient population. More studies are required to determine the long-term reduction in myopia progression across different ethnic populations. It’s for these reasons that increasing outdoor time for children would be most effective as an adjunct, rather than a primary, treatment option.
Low-dose Atropine
Treatment with low-dose atropine has become a more popular treatment option following the results of the Atropine for the Treatment of Myopia studies, ATOM 1 and ATOM 2. In these studies, investigators used doses of atropine that were low enough to avoid photophobia and near vision blur, yet could also control myopic progression over time. Specifically, ATOM 2 demonstrated that the 0.01% concentration was most effective for myopia control after five years, when taking into consideration the effect following treatment cessation and side-effect profile (SE progression of -1.38 ±0.98 D; axial length progression of 0.75 ±0.48 mm).8
However, within the past year, the Low-Concentration Atropine for Myopia Progression (LAMP) study demonstrated that a higher concentration of atropine could have a more profound effect than lower concentrations. After two years, children treated with 0.05% atropine were found to have less myopic progression and less axial lengthening compared to 0.01% (spherical equivalent refraction progression of -0.55 ±0.86 D vs. -1.12 ±0.85 D and axial length progression of 0.39 ±0.35 mm vs. 0.59 ±0.38 mm).14 However, the LAMP study is still ongoing and the effects of treatment cessation remain unknown.
A major benefit of atropine is that it’s effective and easy to implement for parents of young children. Also, studies have shown that these relatively low doses have minimal side effects. However, long-term data in regards to toxicity to ocular structures is not yet available. Additionally, the price of the drug may be a barrier for some patients: A month’s supply of a low dose of atropine has to be compounded and can cost up to $50 for a bottle if not covered by insurance (totaling $600 per year). Increased availability in non-compounded forms, which would be possible with some changes from the pharmaceutical industry, or improved insurance coverage for these medications could decrease the therapy’s cost-barrier for families.
 |
Contact Lenses
Researchers and clinicians have been exploring ways that contact lenses can help suppress an eye’s drift toward nearsightedness.
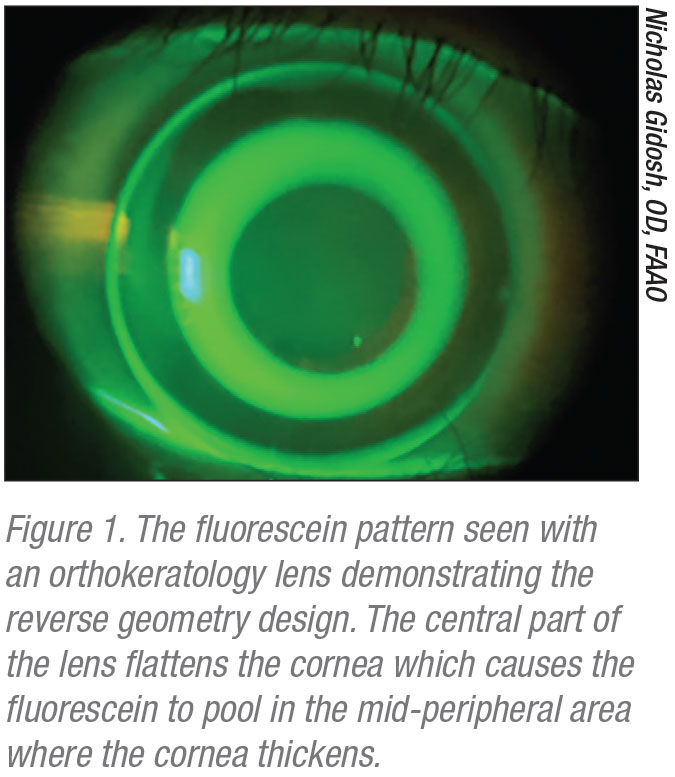
• Orthokeratology. Until very recently, orthokeratology contact lenses were the only lenses regularly used for myopia control. These rigid lenses are worn overnight and removed in the morning. While the patient is sleeping, the lens flattens the central cornea by epithelial cell compression (See Figure 1). This corneal shape change temporarily reduces myopic refractive error, allowing the patient to see clearly during the day without the lenses. This can be an advantage for patients in sports, especially watersports. As the central cornea flattens, the mid-peripheral cornea simultaneously thickens, which creates peripheral myopic defocus. This myopic defocus is thought to inhibit axial lengthening, thereby inhibiting myopic progression overall.
A recent literature review found that orthokeratology lenses reduced axial elongation by 50 percent over a two-year period (a change of
0.30 mm in the treatment group vs. 0.60 mm in a control group).15 In these studies, there wasn’t sufficient data on a washout period. Therefore, the authors of the study couldn’t determine if there was a rebound effect (as happened with some doses of atropine) after treatment cessation.
There are several concerns to address with orthokeratology treatments. One drawback is that these lenses aren’t covered by insurance. The initial orthokeratology fitting process and contact lenses generally range from $1,000 to $2,000, but can be as high as $4,000. Additionally, this is a niche treatment, so the patient must search for an eye-care provider who is specially trained to fit these lenses.
There are other restrictions that can prove to be prohibitive for this form of treatment: The lenses must be worn for eight hours for full correction, and they will only correct up to 6 D of myopia, due to the limits of epithelial cell compression. Also, while adverse events are rare due to the high oxygen permeability of the lenses, microbial keratitis can occur with poor hygiene and poor compliance.15 This may be a considerable barrier for children, in whom ulcerative keratitis could lead to permanent vision loss.
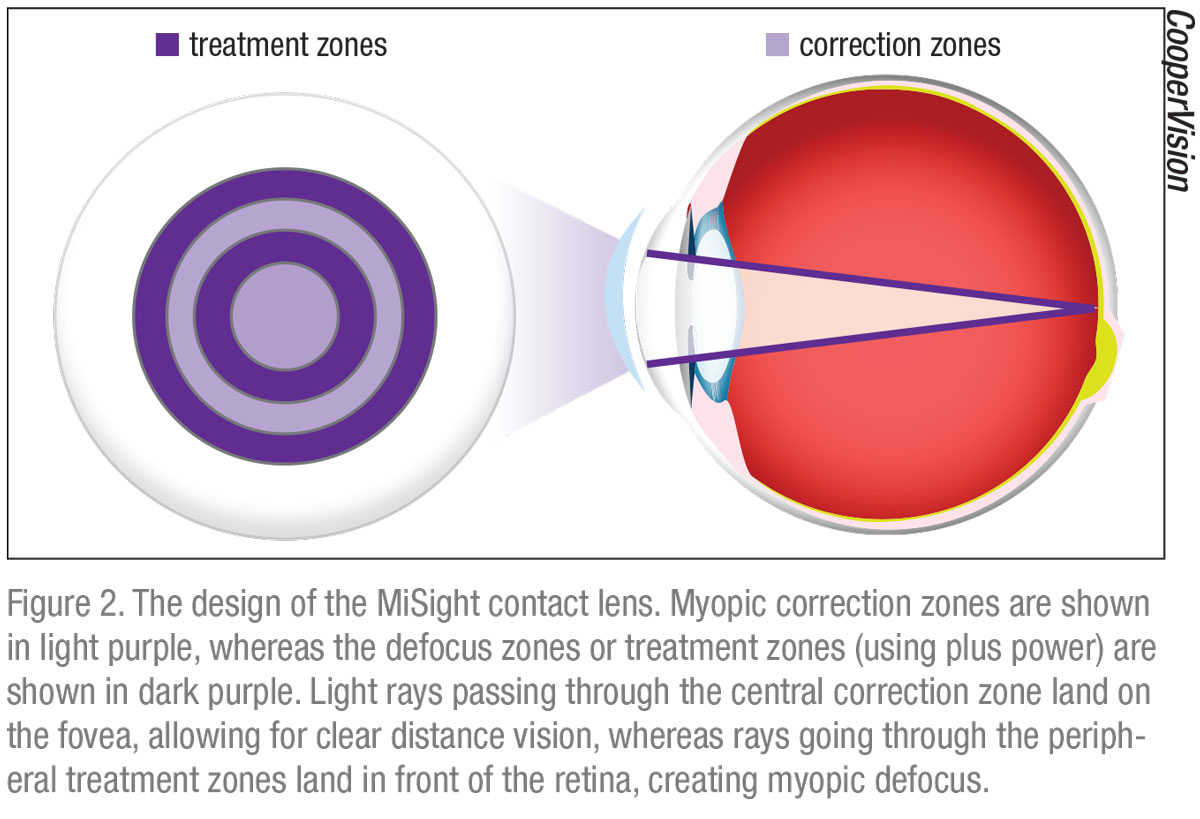
• MiSight Lens. CooperVision is in the process of launching the first FDA-approved daily wear contact lens for myopia control, called MiSight. The dual-focus design of this contact lens creates myopic defocus to inhibit myopia progression (See Figure 2 for design details).
The company’s three-year clinical trial showed a reduction in myopic progression of 0.73 D and axial length reduction of 0.32 mm at three years compared to the control group. In stark contrast to the orthokeratology lenses, which are hard and worn while sleeping, this lens is a daily disposable made from a material similar to the Proclear 1-Day contact lens. There were no serious adverse events (e.g., microbial keratitis) during the three-year study, and wear-time averaged 13 hours per day for the lens.16 While this is the first randomized clinical trial done with this lens, there have been similar results for other dual-focus contact lens designs.17
The MiSight clinical trial is ongoing and will continue for seven years. At the end of the study, the authors hope to be able to assess the long-term myopia control and safety profile of the lens, as well as any rebound effect after treatment cessation. This study also included a more heterogeneous demographic to better understand myopia control across different ethnicities.
There are barriers to this treatment similar to orthokeratology. In order to fit the MiSight lens, the practitioner must be certified through a training program (launched in the spring of 2020). Lenses are only available to practitioners who have completed the certification process. The lens-fitting process is similar to orthokeratology: There’s a charge for the fit, the lenses and any follow-up care required. Unfortunately, the lenses are not covered by insurance and the cost is similar to orthokeratology lenses.
In conclusion, multiple treatment options are currently available to slow the progression of myopia in pediatric patients. Optical defocus spectacles and contact lenses, orthokeratology and low-dose atropine all show similar results in the reduction of myopic progression and a reduction of axial lengthening. The practitioner must decide on the most appropriate option(s) for each patient.
There was less myopic reduction with outdoor sunlight alone. Therefore, it may be appropriate to encourage additional outdoor sunlight exposure in patients who are still emmetropic, or as an adjunct therapy to one of the other options.
Low-dose atropine may be a preferable treatment in young patients, as parents may be hesitant to start contact lenses at an early age. However, adverse effects are rare with contact lenses in children, and the new daily disposable lens option may make it easier for children and parents to maintain proper hygiene. In the future, a spectacle lens option may be available, and it may be more widely embraced than contact lenses, if the dual-focus design is introduced in the United States.
All of these treatment options for myopia progression currently come with some financial burden for the family, so education and discussion with parents is crucial when selecting the appropriate treatment plan for the patient. REVIEW
Dr. Wasserman is a clinical instructor of pediatric ophthalmology and strabismus at Wills Eye Hospital and a clinical associate professor at Thomas Jefferson University Hospital in Philadelphia.
Drs. Franz and Rana practice in the Pediatric Ophthalmology department at Wills Eye Hospital.
None of the authors has a financial interest in any of the products mentioned in the article.
Please direct correspondence to:
Dr. Tara Franz
Pediatric Ophthalmology
Wills Eye Hospital
Sidney Kimmel Medical College of Thomas Jefferson
University
840 Walnut Street
Suite 1210
Philadelphia, Pennsylvania, 19107
Phone: (267) 733-9693
Fax: (215) 928-3983
Email: tfranz@willseye.org
1. Holden BA, Fricke TR, Wilson DA, Jong M, Naidoo KS, Sankaridurg P, Wong TY,Resnikoff S. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology 2016;123:5:1036-1042.
2. Saw SM, Gazzard G, Shih-Yen EC, Chua WH. Myopia and associated pathological complications. Ophthalmic Physiol Opt 2005;25:5:381-91.
3. Mutti DO, Hayes J, Mitchell GL, et al. Refractive error, axial length, and relative peripheral refractive error before and after the onset of myopia. Invest Ophthalmol Vis Sci 2007;48:6:2510-2519.
4. Hung LF, Crawford ML, Smith EL. Spectacle lenses alter eye growth and refractive status of young monkeys. Nat Med 1995;1:761-5
5. Smith EL III, et al. Peripheral vision can influence eye growth and refractive development in infant monkeys. Invest Ophthalmol Vis Sci 2005;46:11:3965-72.
6. Mutti DO, Sholtz RI, Riedman NE, Zadnik K. Peripheral refraction and ocular shape in children. Invest Ophthalmol Vis Sci 2000;41:5:1022-30.
7. Cohen Y, Peleg E, Belkin M, et al. Ambient illuminance, retinal dopamine release and refractive development in chicks. Exp Eye Res 2012;103:33-40.
8. Chia A, Lu Q, Tan D. Five-year clinical trial on atropine for the treatment of myopia 2. Ophthalmology 2015;123:2:391-399.
9. Gwiazda J, Hyman L, Hussein M, et al. A randomized clinical trial of progressive addition lenses versus single vision lenses on the progression of myopia in children. Invest Ophthalmol Vis Sci 2003;44:4:1492-1500.
10. Correction of Myopia Evaluation Trial Study 2 Group (part of the Pediatric Eye Disease Investigator Group). Progressive-addition lenses versus single-vision lenses for slowing progression of myopia in children with high accommodative lag and near esophoria. Invest Ophthalmol Vis Sci 2011;52:5:2749-2756.
11. Lam CSY, Tang WC, T DYY, et al. Defocus incorporated multiple segments (DIMS) spectacle lenses slow myopia progression: A 2-year randomised clinical trial. British Journal Ophthalmology 2020;104:3:363-368.
12. He M, Xiang F, Zeng Y, et al. Effect of time spent outdoors at school on the development of myopia among children in China. JAMA 2015;314:11:1142-1148.
13. Wu PC, Chen CT, Lin KK, et al. Myopia prevention and outdoor light intensity in a school-based cluster randomized trial. Ophthalmology 2018:125:8:1239-1250.
14. Yam JC, Li FF, Zhang X, et al. Two-year clinical trial of the Low-Concentration Atropine for Myopia Progression (LAMP) study. Ophthalmology 2020;126:1:113-124.
15. VanderVeen DK, Kraker RT, Pineles SL, Hutchinson AK, Wilson LB, Galvin JA, Lambert S.R. Use of orthokeratology for the prevention of myopic progression in children: A report by the American Academy of Ophthalmology. Ophthalmology 2019;126:4:623-636.
16. Chamberlain P, Peixoto-de-Matos S, Logan N, et al. A 3-year randomized clinical trial of MiSight lenses for myopia control. Optom Vis Sci 2019;96:8:556-567.
17. Anistice N, Phllipa J. Effect of dual-focus soft contact lens wear on axial myopia progression in children. Ophthalmology 2011;118:6:1152-1161.



