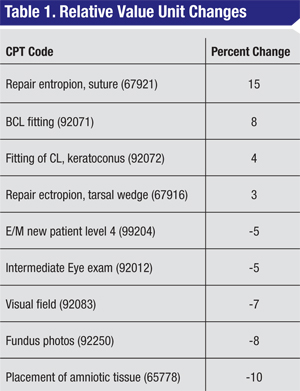
A . The Medicare Physician Fee Schedule, published in November 2013, contains several relative value unit (RVU) changes. For information on the percentage of change from 2013, please see Table 1, below.
 |
Q. Are there any changes to ICD-9 diagnosis codes that require attention?
A . No new ICD-9 codes were published, in anticipation of ICD-10 implementation on October 1, 2014. The Centers for Medicare & Medicaid Services expects to move forward with ICD-10 despite requests for another postponement by the American Medical Association and various specialty societies.
Q. Are there new drug codes pertinent to ophthalmology in the 2014 Healthcare Common Procedural Coding manual?
A . Yes, the 2014 HCPCS manual contains a new code for what is more commonly known as Jetrea (J7316, Ocriplasmin 0.125 mg).
The J7316 code will require four units on the claim form for appropriate reimbursement of the drug vial.
Q. Are there any Category III code changes for 2014?
A . There are a number of changes in the hard copy of the 2014 Current Procedural Technology Manual. However, these Category III codes, released semiannually by the American Medical Association, were implemented on July 1, 2013:
• 0329T Monitoring of intraocular pressure for 24 hours or longer, unilateral or bilateral, with interpretation and report;
• 0330T Tear film imaging, unilateral or bilateral, with interpretation and report;
• 0333T Visual evoked potential, screening of visual acuity, automated.
The following Category III codes are deleted in the 2014 handbook:
• 0192T Insertion of anterior segment aqueous drainage device, without extraocular reservoir, external approach (replaced with new Category I code);
• 0124T Conjunctival incision with posterior extrascleral placement of pharmacological agent.
Coverage and payment for Category III codes remains at carrier discretion.
Q. What changes were published with Category I codes in CPT 2014?
A . Several changes exist in the CPT 2014 manual. There is a new code to replace Category III code 0192T, which is deleted from the 2014 CPT handbook:
• 66183 Insertion of anterior segment aqueous drainage device, without extraocular reservoir, external approach.
With this change, the 2014 Medicare Physician Fee Schedule will establish RVUs and a payment rate for the surgeon; previously, the reimbursement was determined by the Medicare Administrative Contractor.
Revisions were made to the two codes for amniotic membrane placement, and an additional adjustment to the language of a third:
• 65778 Placement of amniotic membrane on the ocular surface, without sutures;
• 65779 Placement of amniotic membrane on the ocular surface, single layer sutured.
The revised text below 65780, Ocular surface reconstruction; amniotic membrane transplantation, multiple layers states:
“For placement of amniotic membrane without reconstruction using no sutures or single layer suture technique, see 65778, 65779.”
Code 13150, Repair, complex, eyelids, nose, ears and/or lips; 1.0 cm or less has been deleted, and the following codes revised to reflect this change:
• 13151 Repair, complex, eyelids, nose, ears and/or lips; 1.1 cm to 2.5 cm;
• 13152 2.6 cm to 7.5 cm;
• +13153 each additional 5 cm or less (list separately in addition to code for primary procedure).
In the integument section, add-on code +15777, Implantation of biologic implant (e.g., acellular dermal matrix) for soft tissue reinforcement (i..e, breast, trunk), has supplemental parenthetical instructions that might apply in oculoplastic surgery with xenografts. The new text reads:
“For implantation of biologic implants for soft tissue reinforcement in tissues other than breast and trunk, use 17999.”
Q. What types of regulatory issues were identified in the Office of Inspector General’s annual work plan as areas of concern for ophthalmology in 2014?
A . Unfortunately, we don’t know at this time. The annual publication of the OIG work plan, usually occurring in the fall, was delayed until January 2014. According to the OIG website, “This change from the usual October release will better align with priorities OIG has set for the coming year, a time of continuing fiscal challenges.”
Q. Is the Recovery Audit Contractor program continuing to report successful recoupment of overpaid dollars?
A . Yes. The RAC program continues to thrive. Several states have Medicaid RAC programs in place. Medicaid RACs operate at the direction of the states with the discretion to determine what areas of their Medicaid programs to target.
Total corrections since the Medicare Fee-for-Service Recovery Audit Program began in October 2009 stand at $5.7 billion. Between FY 2011 and 2012, corrected payments doubled from $939 million to $2.4 billion. As of June 2013, corrected payments for FY 2013 were already at $2.3 billion.
In June 2013, all four RACs added blepharoplasty for review of physician services, hospital services and ASC services. The review is listed as a “complex” review, which means that medical records are requested prior to any overpayment demand letter.
Q. Do any Medicare Part B changes affect beneficiaries in 2014 from a cost perspective?
A . The Medicare Part B premiums remain $104.90 for most beneficiaries. The Part B deductible also remains unchanged at $147. These beneficiary costs are the same as in 2013.
Q. Are there any bonuses for participating in the Physician Quality Reporting System or the Electronic Prescribing Incentive Program in 2014?
A . While 2014 is the final year to secure a bonus for successful participation in the PQRS, the final year for the eRx bonus program was 2013. It is expected that all providers except those that are exempt are using electronic prescribing systems. If not, penalties exist. For the PQRS, the bonus remains at 0.5 percent of total Medicare allowed dollars for professional services to those who successfully participate in the program. This (2014) is the final year to secure a bonus for successful participation.
Q. Will the Electronic Health Record Incentive Program continue to pay bonuses to eligible providers?
A . Yes. As of September 2013, the EHR Incentive Bonus Program had paid out $3.9 billion to eligible providers: $115 million paid to ophthalmologists and $157 million to optometrists. Providers who have successfully attested to Stage 1 for two years, some three years, will be required to meet Stage 2 requirements for 2014. For those not yet utilizing electronic health records, they must start by July 1, 2014 and complete their meaningful use attestation for Stage 1 by October 1, 2014 to avoid penalties in 2015. The published penalty is 1 percent for 2015, 2 percent for 2016 and 3 percent for 2017. Beyond 2017, penalties are up to 5 percent.
Q. Will the Multiple Procedure Payment Reduction continue in 2014?
A . Yes. The MPPR policy reduces the technical component of the second and subsequent diagnostic tests by 20 percent when more than one diagnostic test is performed at one patient encounter on the same day by the same physician or group. The list of tests includes ultrasounds, imaging and visual fields. Tests that do not have a technical component (i.e., gonioscopy) are not subject to this policy. There are no changes to the list of affected ophthalmic codes from 2013 to 2014.
Q. What’s happening with Ambulatory Surgery Centers’ facility fees in 2014?
A . ASC facilities realize an increase in reimbursement. For 2014, the Consumer Price Index and Multifactor Productivity Adjustment update the ASC facility rate conversion factor by 1.2 percent. This increase results in a very small but positive change to facility reimbursement.
Q. Did hospital outpatient department rates increase similarly to ASC facility rates?
A . Yes. Hospital outpatient department rates increased approximately 1.7 percent for 2014.
Ms. McCune is vice president of the Corcoran Consulting Group. Contact her at DMcCune@corcoranccg.com.



