A method that combines saturating an abnormal cornea with riboflavin and then activating its tissue-strengthening properties with ultraviolet-A light is showing promise in early clinical trials for the treatment of keratoconus and post-LASIK ectasia.
The procedure is known as collagen cross-linking, and our group in
An Answer for Conventional Approaches
Collagen cross-linking may be an answer for some of the problems with the conventional treatments for treating corneal ectasia: typically, first glasses for astigmatism, then contact lenses to manage the optical effects of the distortion, then corneal transplantation if the contact lenses don't adequately address the disorder. In the last several years, other surgical options have been added between contact lenses and corneal transplantation:
• implantation of Intacs segments to improve corneal rigidity;
• conductive keratoplasty to apply radiofrequency pulses to shrink the bulging area of the cornea; and
• a combined Intacs/CK procedure.
However, these approaches have their shortcomings. They likely don't address the potentially progressive nature of ectasia. So if they are effective but the condition progresses anyway, the patient is symptomatic again. These procedures also have regression in some patients, as the effect, especially of CK, is typically greatest initially.
An Answer for Ectasia?
Early laboratory work found that applying the vitamin riboflavin followed by UV light to abnormal corneal tissue strengthened the tissue. This remains the basic premise for the procedure, although variations have been adapted to the technique as investigators try to define the best parameters for corneal cross-linking.
Ophthalmologists in Germany started working with corneal cross-linking in animal eyes in the late 1990s.1 A few years later, investigators from the same group published their results of collagen cross-linking in 23 human eyes.2 They found that the progression of keratoconus was halted in all eyes, and that maximal keratometry readings had been reduced in 16 eyes. A more recent investigation out of
In 2007, a group in
The Procedural Steps
In corneal cross-linking, the surgeon debrides the epithelium as in PRK. Next, the surgeon saturates the surface with riboflavin, and then applies UV light to activate the cross-linking process. Postoperatively, the regimen replicates PRK: a bandage lens with antibiotic and anti-inflammatory drops.
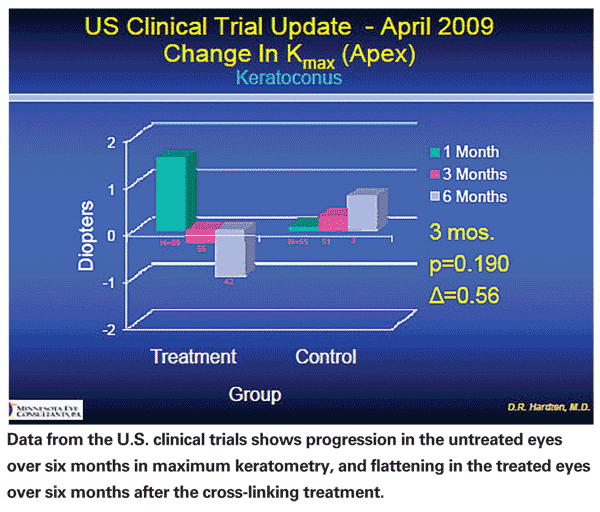
The obvious issues with an investigative procedure such as corneal cross-linking are twofold: Does it work? And, is it safe? From my perspective, the answer to both questions is yes. It does appear to reduce the progression of keratoconus. Some corneas have exhibited a small degree of flattening in areas that had shown bulging.
Investigators have used corneal cross-linking in two other settings involving difficult-to-treat corneas:
• Patients who had radial keratotomy as long as 20 years ago.These corneas exhibit weakening over time. The thinking is that cross-linking might reduce progression of incisional weakening and strengthen the corneal structure. However, long-term analysis on the efficacy of corneal cross-linking in this setting is lacking.
• Infectious keratitis. The thinking is that UV light sterilizes the cornea and the absorptive properties of the riboflavin prevent excess UV light from damaging intraocular structures.
The riboflavin used in the initial trials we participated in, however, is not yet approved by the Food and Drug Administration. Enrollment for one of the recent clinical trials is closed and now follow-up is proceeding. Corneal cross-linking for keratoconus, post-LASIK ectasia and other corneal disorders is more widely available in Europe and
Dr. Hardten practices with Minnesota Eye Consultants and is on the adjunct faculty in the Department of Ophthalmology at the University of Minnesota Medical School.
1. Spörl E, Huhle M, Kasper M, Seiler T. Increased rigidity of the cornea caused by intrastromal cross-linking. Ophthalmologe 1997;94:902-906.
2. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol 2003;135:620-627.
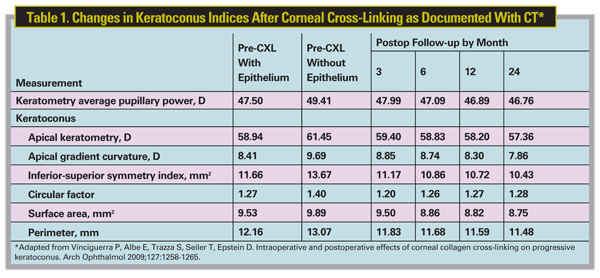
3. Vinciguerra P, Albe E, Trazza S, Seiler T, Epstein D. Intraoperative and postoperative effects of corneal collagen cross-linking on progressive keratoconus. Arch Ophthalmol 2009;127:1258-1265.
4. Hafezi F, Kanellopoulos J, Wiltfang R, Seiler T. Corneal collagen crosslinking with riboflavin and ultraviolet A to treat induced keratectasia after laser in situ keratomileusis. J Cataract Ref Surg 2007;33:2035-2040.