Determining IOL power for a cataract patient has always been challenging, but it's gotten a lot tougher lately. Thanks to refractive surgery, more and more individuals with cataracts have altered corneas that make it difficult to ensure an acceptable outcome for cataract surgery. Many surgeons are trying to solve this problem, but so far the result has been a proliferation of formulas and methods with no one solution rising to the top.
Here, several experienced surgeons share their current outlook and insights regarding this situation.
Many Options, No Clear Answer
"Right now, there are more than 20 formulas and methods, and none of them is 'the one,' " says Mark Packer, MD, FACS, clinical associate professor of ophthalmology at
"Some of the current formulas only use axial length; some use corneal topography; some use pre-LASIK values or the contact lens method," he continues. "The problem is that they give you different answers. Plus, most of the supporting data that's been published involves very small numbers of patients. Many papers include no real clinical data at all. Instead, they perform a thought experiment. They say, 'Here are some people who've had LASIK; let's use this method to predict what would happen if we put in a lens of this power.' In reality, you can't know for sure if a method works until you've actually done the surgery."
Among the methods used to calculate IOL power, the historical method, which requires pre-refractive surgery data, is often thought of as the best approach. "I still recommend using the historical and contact lens methods if you can," says Jack T. Holladay, MD, MSEE, FACS, clinical professor of ophthalmology at
Nevertheless, Dr. Holladay notes that even this approach has drawbacks. "This method assumes that the refractive changes that occurred between the pre-refractive-surgery measurements and the current refraction were solely due to the cornea, not the crystalline lens," he says. "Unfortunately, that's usually not true. A cataract growing inside the eye can change the refraction; if you rely on the historical method in this situation, you end up with a refractive surprise."
Dr. Holladay also notes the other, more common problem with the historical method: The information is often not available. "You don't always have the pre-surgery K-readings and know the patient's refractive change," he says. "You may not see the patient until many years after the refractive surgery. Given these caveats, I believe direct measurement is the best way to proceed."
Working with the Formulas
With the proliferation of formulas and methods intended to calculate current corneal power, comparing the results of multiple formula calculations is becoming common practice. Not surprisingly, this can entail a fair amount of work.
Kenneth J. Hoffer, MD, clinical professor of ophthalmology at UCLA in
"Calculating IOL power after refractive surgery is tough for several reasons," says Dr. Hoffer. "First, most keratometers don't measure the central cornea where the effective corneal power is. Second, LASIK and PRK change the index of refraction between the front and back surfaces of the cornea. Third, many modern formulas predict IOL position using the axial length and corneal curvature. That won't work in this situation because these procedures change the corneal curvature—but not the expected position of the IOL."
Dr. Hoffer says there are currently about 24 formulations in use. "They can be divided into two major categories," he explains. "The first group tries to predict the true power of the patient's cornea based on multiple factors. These formulas give you a different K-reading to plug into your IOL power calculation. The remaining methods say, 'OK, your direct K-reading may not be accurate, but go ahead and calculate the IOL power; we'll give you a way to fudge or adjust that power to make up for the error.' Each of these groups can be subdivided into those that require some pre-op historical data and those that don't."
While developing his comprehensive spreadsheet, Dr. Hoffer encountered
How the Spreadsheet Works
Dr. Hoffer explains how to use the IOL Power Tool. "First of all, the program you download from the website is an XLT—an Excel template," he says. "Once it's on your computer, you double-click on it and it opens up a spreadsheet that looks exactly like the template. The spreadsheet you open and use doesn't affect the template, so if you make a mistake, you can just delete it and open a new one." 
Dr. Hoffer says the spreadsheet includes features designed to help the surgeon use it correctly. "As you move your mouse over any box, it tells you what needs to be entered and how to get that data," he says. "The upper area has places for the surgeon's name, patient's name, right or left eye, the date, and so forth. There are boxes for axial length, the K-reading you get from a manual keratometer, the K-reading you get from an IOLMaster and the K-reading you get from a topographer. If you use the contact lens method, there are boxes for the refraction with and without the contact lens, the base curve of the contact lens and the power of the contact lens.
"Once you've entered the correct data for the method you're using, the spreadsheet automatically calculates the K-reading that method would recommend," he continues. "For example, suppose you choose to use the Haigis method. You type in the IOLMaster K-reading on the left-hand side of the spreadsheet and the K-reading recommended by the Haigis method appears on the right-hand side next to the box labeled 'Haigis Method.' If you use the contact lens method, you enter the four pieces of data that are required and the K-reading appears in the appropriate box on the right."
Dr. Hoffer says the spreadsheet is intended to make it easier to coordinate whatever data you're able and willing to collect, but the surgeon still has to decide which number to use to complete the power calculation. "You can run as many formulas as you want," he says. "The spreadsheet shows all of the K-readings that have been calculated and notes which of them is lowest. You may see a trend; maybe one number is an outlier. Hopefully, this will give you an idea of which K-reading to use, but you'll have to make the final choice."
Dr. Hoffer notes that his intention was never to create a program that would calculate the IOL power directly—only the K-reading. "We didn't want to be in direct competition with any of the IOL power calculation programs," he explains, "so you have to use the spreadsheet in conjunction with one of those programs.
"Once you've done the IOL power calculation using the K-reading that you've chosen, you can enter the calculated IOL power back into our spreadsheet in the appropriate box," he continues. "If a fudge factor is required, the spreadsheet will calculate that for you as well. You can save the spreadsheet using the patient's name for easy identification, and you can print it out and place it in the chart if desired."
He adds that the spreadsheet is completely free. "When you charge people, you have to respond to complaints!" he says with a laugh.
Even in the Best Circumstances
Even when all the pre-refractive surgery measurements are available, determining the correct IOL power with certainty can still be difficult or impossible. Dr. Packer experienced this firsthand recently. He performed LASIK on a woman in her early 20s—a moderate myope with no astigmatism to speak of. She ended up 20/15 uncorrected in both eyes. But a few months later, a firecracker explosion in front of one eye triggered a traumatic posterior subcapsular cataract. Because he had all of her preop data, he assumed calculating the IOL power would be straightforward.
"The first method I used to calculate the IOL power was the corneal bypass method," he says. "It was recently published in the Journal of Cataract and Refractive Surgery.1 The idea is pretty elegant; if you know someone's pre-LASIK refraction, topography, keratometry and so forth, you ignore the current state of the cataract and cornea completely. You place the pre-LASIK keratometry values, manifest refraction and current axial length in the
"I did the calculations using the bypass method; the result was 23.5 D," he continues. "I was happy with that, but I always use at least two or three different IOL power calculation methods in post-LASIK patients. So I also used the modified Maloney method and the BESSt formula. The former gave me a result of 25.5 D; the latter said 24 D. Even with all of this information at my disposal, I still got a 2-D spread of suggested powers.
"The way to resolve this dilemma was suggested by Jennifer Smith, assistant professor of ophthalmology at
"As it turned out, the patient came out 20/15 and J2 with a Crystalens AT-50 implant," he concludes. "So she was very happy. But the lesson for me was clear—even if you have all of the preop information, there's no guarantee that one particular formula will give you the IOL power that's going to produce a perfect outcome."
Sources of Error
A major source of error when trying to calculate the IOL power for a post-refractive surgery patient is determining the current refractive power of the cornea. One tool that may be helpful in this situation is the Pentacam Comprehensive Eye Scanner (Oculus,
"There are two sources of error we run into with direct measurement of corneal power," explains Dr. Holladay. "The first is that keratometry and topography don't measure the central part of the cornea. A keratometer fails to measure a scotoma in the center that's about 3.2 mm in diameter for a 44-D cornea. A topographer gets it down to about 2 mm, but that still leaves you with a large area in the center that's not being measured.
"The second source of error is that topographers and keratometers only measure the front surface of the cornea," he continues. "To calculate corneal power, you need to know the ratio of the front surface to the back surface. In a virgin eye, the front surface curvature is sufficient to give you a close approximation of corneal power because the ratio of the back to front surface radii is about 82 percent (6.32/7.70) in the vast majority of normal eyes. (Several studies, including one we're in the process of publishing, have confirmed this.) Refractive surgeries such as LASIK or PRK change only the front surface, altering the ratio. Determining the correct ratio becomes a second source of error.
"If you add these two confounding factors together," he notes, "the error that we'd make by taking direct measurement of the cornea with a keratometer would be 25 percent of the refractive change caused by the refractive procedure. So, if the patient had a -8 D LASIK and we used the paracentral power and the standard ratio, we would end up making a 2-D overestimate of the corneal power, which would cause a 2-D hyperopic refractive surprise."
Working with the Pentacam
Dr. Holladay explains that tomographers like the Orbscan II (Bausch & Lomb) and the Pentacam measure both the central zone of the cornea and the back surface. Recently, he's been working with Oculus to confirm the accuracy of the Pentacam's measurements and determine which optical zone diameter produces the most accurate results.
"We studied 100 consecutive LASIK patients between the ages of 20 and 35," he says. "We chose this age range because we could be nearly certain that the subjects weren't having lenticular changes. This meant we could use the historical method to calculate post-refractive Ks without worrying about error being induced by changes in the crystalline lens.
"We measured the subjects with the Pentacam both before and three months after LASIK," he continues. "We compared the results to the refractive change calculated by the historical method. Then, we used the data to determine which diameter optical zone produced the best agreement with the historical method's estimate. It turned out that the best agreement occurred if we used a 4.5-mm optical zone. The correlation, r2, was about 98 percent—amazingly accurate."
Dr. Holladay says they also did this calculation on former radial keratotomy patients, to see how accurate the Pentacam's measurement could be in this situation. "Warren Hill provided us with pre- and postop numbers for 41 RK eyes," he says. "He used the Holladay IOL Consultant to back-calculate what K-reading would have given him the perfect outcome, based on the patient's postop refraction. Then we took those back-calculated values and measured the RK patients. The 5-mm optical zone gave us the best correlation; it explained about 90 percent of the data. The correlation wasn't as good for RK as for LASIK, but for both types of surgery the results were two or three times more accurate than the readings you get with a standard keratometer."
The EKR Report
With all the data they collected, Dr. Holladay and his team generated a two-page report called the Equivalent Keratometric Reading that can be called up on the Pentacam's screen. "The first page gives you several maps, along with steep, flat and mean K-readings to plug into your IOL power calculation," he says. "The second page has additional details that are especially important for the RK patient.
"In most cases the 4.5-mm value will produce the best outcome, but the report also gives you the readings for other size optical zones, including the front and back surface powers," he continues. "RK patients can have 10 D of variation in corneal power across the cornea; if a patient's pupil never gets wider than 2 or 3 mm, you might get a better result using the 3-mm value. Likewise, if a LASIK patient has a very large pupil, you might use a larger optical zone number."
Dr. Holladay also notes that the EKR report includes a graph of the patient's cornea showing the corneal power as you move from the center to the periphery. This allows you to see how the corneal shape was altered by the surgery. It will also calculate the eye's probable K-reading before the refractive surgery took place, if those numbers are not available.
Measuring Power in the OR
One new approach to determining IOL power is an update on the idea of measuring the patient's refraction on the operating table after the cataract has been removed. The new approach uses a wavefront device manufactured by WaveTec Vision (
"This technology is different from Hartmann-Shack," notes Dr. Holladay. "It's better able to measure highly aberrated eyes. The idea is that after you've removed the cataract, you turn the instrument on and measure the refractive error, either before or after you implant the new lens. Several surgeons are using it to calculate IOL power before implanting the lens, using the Holladay IOL Consultant to get the power for an aphakic eye, but you can also use it to see the refractive result after you implant the lens. The latter will be especially useful for aligning a toric IOL after it's inside the eye. In general, this device has the potential to eliminate refractive surprises, whether the patient has had prior refractive surgery or not."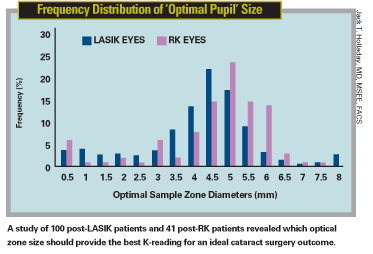
Dr. Holladay notes that so far, the device has been primarily tested with patients who have not had prior refractive surgery. "None of the patients so far have had a refractive surprise that needed correcting as a result of pre-surgery calculations," he says. "Nevertheless, the measurements made by the new device have led to outcomes within a half or quarter D of the desired outcome."
Dr. Holladay points out that surgeons will still have to calculate a power ahead of time. "But if you do find you have a surprise, you'll be able to pick out another lens," he says. "It will supplement what we do today."
Dr. Packer has also been working with the WaveTec instrument. "It does show promise," he says. "We've been measuring the patient's eye on the table in the aphakic state. We take the lens out, inflate the eye to a known pressure, capture the corneal wavefront and back-calculate what the IOL power should be. I presented preliminary data on this at this year's American Society of Cataract and Refractive Surgery meeting, but the number of patients is still small, and most eyes have been virgin eyes. Before this becomes mainstream, we'll need a lot more experience and data."
Dr. Packer says that when using the WaveTec device, he wouldn't choose to wait until the lens was in the eye to do the refraction. "One reason is the added risk of complications and OR time for switching the lens," he says. "The other reason is that in most cases the implant won't be in its final position until long after the patient has left the OR. If you measure before that point, you might take out a lens that would have ended up being perfect."
Dr. Packer notes that previous RK can also cause the refraction to shift after surgery. "With these patients we wait quite a while for the refraction to stabilize," he says. "During surgery, the peripheral cornea swells as a result of the incisions; the cornea flattens, and the patient ends up hyperopic initially. I can't imagine any way to know within an hour of implantation whether you've put in the correct lens."
He admits that the problem of lens or refraction shift is not eliminated by measuring the aphakic eye. "The only real solution is to look at the postoperative data on, say, 1,000 eyes and compare predicted and actual power," he says. "Then you can introduce a fudge factor to compensate for the difference—a personalized A-constant."
Dr. Packer says that in spite of this caveat, the WaveTec device could still be a major step forward. "Because of the problem of final lens position, you can't expect 100-percent accuracy, no matter what you do," he notes. "But this approach should put us into the ballpark of the results we get with standard eyes that haven't had previous surgery. For me right now, that's 95 percent within ±0.5 D of target."
Consolidating the Data
"To a certain extent, finding the right number is like shooting in the dark," notes Dr. Packer. "So the only thing we can do for now is share our results, and warn these patients that there's a high likelihood they'll need a second procedure to correct residual refractive error after cataract surgery."
Dr. Hoffer hopes his spreadsheet will help to resolve this dilemma. "We're inviting surgeons to send their postop results to us," he says. "If they do, we might be able to determine whether one of these formulas really is more effective than the others."
For now, Dr. Packer says he believes the most sensible approach is to look at the results of multiple calculation methods and decide what kind of error you'd rather be correcting at the end. "In most cases, unless the patient has too little corneal tissue for further myopic LASIK, I'd rather be correcting a little myopia than a little hyperopia," he says. "For that reason, I think the best approach is to take the least corneal power the formulas produce and then use the IOL that has the highest power. In fact, if this does result in a little myopia, some patients won't mind a little monovision. But if the eye comes out +1 D, you're stuck. You'll be forced to make a correction."
Dr. Packer adds that one exception to this would be when a patient has too little tissue left to do further corrective ablation. "In that situation, I'd opt for implanting a piggyback IOL, and I'd rather implant a plus-powered piggyback lens than a negative-powered one. The plus-powered lenses are thinner and go in more easily. So in this case I would choose to err on the side of a little hyperopia, to ensure that any piggyback lens correction would be as easy as possible."
1. Walter KA, Gagnon MR, Hoopes PC Jr, Dickinson PJ. Accurate intraocular lens power calculation after myopic laser in situ keratomileusis, bypassing corneal power. J Cataract Refract Surg. 2006;32:3:425-9.