At this year's meeting of the American Society Cataract and Refractive Surgery in May, investigators presented some of the first results of the Phase III U.S. Food and Drug Administration clinical trial of Alcon's AcrySof ReStor intraocular lens, which is designed to give presbyopes a measure of pseudoaccommodation. Here's a look at how the lens performed in the trial, and how it's performing for international surgeons who have begun using it in their practices.
How It Works
The ReStor is designed on the principle of diffraction, or the ability to bend light by passing it through sharp edges or slits.
 |
| The ReStor has 16 inner rings that are designed to focus light at near. |
"It's not like the Array lens where one zone is for distance and one is for near," says Dr. Carones. "Since the steps in the lens follow diffractive principles, each ring puts two things in focus, one at near and one at distance, and the brain chooses between them."
Since the lens transfers light into two main points of focus, distance and near, Dr. Carones says the intermediate is "sort of penalized." He's quick to add, however, that the patients in whom he's implanted the ReStor don't complain about their intermediate vision.
How It Performs
In the FDA Phase III study, 566 patients received the ReStor and 194 received the AcrySof monofocal IOL as a control. The mean age of the patients was 69 years.
Ninety-nine percent of the ReStor patients and 98 percent of the monofocal IOL patients see 20/40 or better uncorrected. Eighty-eight percent of the ReStor patients see 20/25 or better uncorrected, compared to 92 percent of the controls.
For near vision tasks, 74 percent of the bilateral ReStor patients read J1 uncorrected, compared to just 14 percent of the monofocal patients. For intermediate vision (defined as 60 cm) 85 percent of the ReStor patients see 20/40 or better, compared to 67 percent of the monofocal patients. Eighty percent of the ReStor patients say they never have to use glasses for near or distance vision, versus 8 percent of controls.
As for contrast sensitivity, James Davison, MD, of Marshalltown, Iowa, a ReStor investigator, says, "In mesopic and photopic conditions, there was no clinically significant reduction in contrast sensitivity with the ReStor lens compared to the monofocal."
Richard Packard, MD, FRCS, a consultant surgeon at King Edward VII Eye Hospital in Windsor, England, has analyzed 22 ReStor patients.
The average age of the patients in his study was 58.5 years, with a range of errors from -6.5 to +6.25 D, with up to 2.5 D of cylinder.
"I was able to operate on that range because nine of the patients got limbal relaxing incisions for their astigmatism, and four had IOLs piggybacked to extend the ReStor's range."
Fifteen of the patients had their lenses removed for cataract and seven were clear-lens extraction/IOL implantation patients.
In six patients (six eyes) that had uniocular ReStor implantation, 95 percent see 20/30 uncorrected at distance, and 92 percent see J2 or better uncorrected at near. In the 16 bilaterally implanted patients, 100 percent are 20/25 or better uncorrected at distance, and 82.4 percent see 20/20 or better. At near, 89 percent are J1 or better.
"All the patients save for one are happy," says Dr. Packard. "The unhappy person can see 20/20 at distance uncorrected, but J3 at near. She has a small amount of age-related macular degeneration that I didn't see until I had removed her cataract."
Dr. Carones has studied his ReStor results in 54 eyes. The patients are within 0.87 D of the intended correction, see 20/25 or better uncorrected at distance and J2 or better at near.
"I've also implanted it in patients who already had a monofocal lens in the other eye," he says. "Though they're still relatively satisfied with the result, they can't avoid using glasses for extended near work, but they can read a mobile phone display quickly without having to take out their reading glasses. Implanting it bilaterally gives better performance."
Drs. Carones, Packard and Davison say patient complaints of visual disturbances have been minor. In the FDA study, 15 percent of the patients have moderate halos around lights, though 99 percent of them said they would have the surgery again.
Though the lens's near and distance vision results impress the surgeons, it seems that more visual compromises must be made at that 60-cm intermediate vision distance with the ReStor. While distance and near come in at around 20/20 to 20/25 for the most part on defocus curves, Dr. Packard says intermediate averages around 20/40. Three of his patients have to wear -1 D spectacles for intermediate work to negate the ReStor's near add.
"As for intermediate vision, all my patients except one are at least J4," says Dr. Carones. "One wasn't able to get satisfactory intermediate vision and uses glasses for the computer.
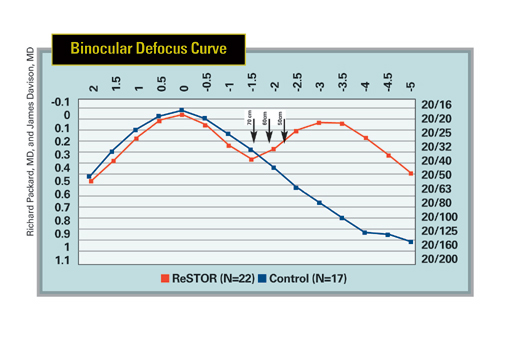 |
| The ReStor has two points of sharp focus, far and near, while the monofocal lens is designed for distance acuity, as shown by the peaks of the graphs. Intermediate distances are represented by the arrows. |
"One of the challenges will be choosing the patients who will be satisfied with the lens, given what we know about what it can do. Patients who are demanding of their intermediate vision aren't good candidates."
Dr. Davison says that preop biometry is very important with the lens, because leaving a patient -0.5 D won't give the person as full a range of vision. "Intermediate vision is variable, and you have to have the right lens power in the eye," he says. "If the result is slightly hyperopic or plano, it's perfect. If it's a little myopic, it's not as good. It's still better than a monofocal lens, however."
Dr. Davison says that the lens could be approved by the end of the year.
"I've been setting aside candidates for it, such as young patients with traumatic cataracts, early cataract patients and patients over 50 who are interested in refractive surgery, because it may be approved late this year," he says.
Dr. Carones is a paid consultant for Alcon's refractive surgery division, but not for his work with the ReStor lens.
Dr. Davison is an investigator in Alcon's U.S. trial of the ReStor lens.
Dr. Packard doesn't receive fees from Alcon for his work with ReStor.



