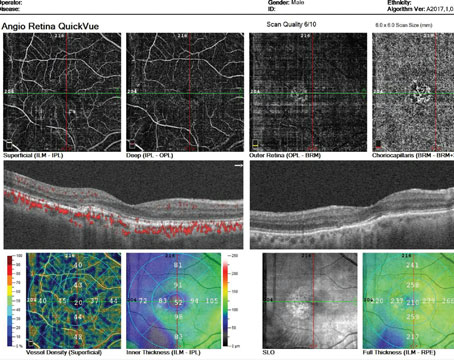
Many new anti-VEGF agents are in various stages of development, and the hope is that these new drugs will provide better durability and have better drying effects than the currently available options. Current anti-VEGF monotherapies for wet age-related macular degeneration are burdensome because they require frequent office visits for eye injections.
“Unfortunately, the benefits of new agents don’t seem to translate directly into better vision. Durability of a drug is critical, but it’s still too early to know how it’s going to be translated into real-life use,” says Karl Csaky, MD, PhD, who is in practice in Dallas.
In this article, experts discuss how a new addition to their armamentarium, as well as some agents in the pipeline, may fit into their therapeutic strategy.
Beovu
The Food and Drug Administration recently approved Beovu (brolucizumab, Novartis) injection for the treatment of wet AMD.1 The approval was based on findings from the Phase III HAWK and HARRIER clinical trials, in which Beovu demonstrated non-inferiority versus aflibercept in mean change in best-corrected visual acuity (BCVA) at one year, but with a potentially less-frequent treatment schedule.
HAWK and HARRIER were similar in design (double-masked, multicenter, active-controlled, randomized).2 They included 1,817 patients with untreated, active choroidal neovascularization due to AMD in the study eye. Patients were randomized to receive intravitreal brolucizumab 3 mg (HAWK only), brolucizumab 6 mg, or aflibercept 2 mg. After loading with three monthly injections, brolucizumab-treated eyes received an injection every 12 weeks and were interval-adjusted to every eight weeks if disease activity was present. Eyes in the aflibercept group received treatment every eight weeks.
At week 48, both brolucizumab groups demonstrated noninferiority to aflibercept in BCVA change from baseline. In the HAWK study, patients receiving 3 mg of brolucizumab gained 6.1 letters, those receiving 6 mg of brolucizumab gained 6.6 letters, and those receiving aflibercept gained 6.8 letters. In the HARRIER study, those receiving 6 mg of brolucizumab gained 6.9 letters and those receiving aflibercept gained 7.6 letters. More than half of the eyes treated with 6 mg of brolucizumab were maintained on 12-week dosing through Week 48 (56 percent in the HAWK study and 51 percent in the HARRIER study). At week 16, after identical treatment exposure, fewer eyes treated with 6 mg of brolucizumab had disease activity compared to eyes treated with aflibercept in both studies (24 percent vs 34.5 percent in HAWK and 22.7 percent vs 32.2 percent in HARRIER).
“I now have patients just starting to come back a month after their second injection, and the hope is that Beovu will be a longer-lasting injection,” says New York ophthalmologist K. Bailey Freund, MD. “It’s a smaller molecule that has high affinity for VEGF so the molar concentration of the drug in the syringe with the same volume is considerably higher than the other three agents that are commonly used. It is expected that some patients will be able to be dosed less frequently.”
According to Wills Eye Hospital’s Carl Regillo, MD, ophthalmologists are excited about the availability of Beovu. “It just got its J-code, so that’s going to pave the way for increasing utilization in practice,” he says. “But, time will tell. The TALON study, which is a Phase IIIb study that has been launched in Europe, is going to give us a good idea of how well it performs in terms of durability compared to Eylea because the drugs are head-to-head and dosed in a treat-and-extend fashion that is identical in both arms. We won’t know the answer for a couple of years, but at least it’s in testing.”
Dr. Freund notes that physicians are trying to strike a balance between safety and efficacy with these drugs. “Obviously, there is a benefit to less frequent injections in terms of treatment burden and cost, but there isn’t the long-term safety data that the others have,” he says. “One issue is that the FDA label for Beovu specifies that this drug is to be used monthly for three doses and then every eight to 12 weeks thereafter. The label reflects the protocol, so the expectation is that the payers might not allow for more frequent dosing than every eight weeks after the first three doses. If an eye can’t be kept dry enough and stable with eight-week dosing, this would be a problem as far as reimbursement. You might have to switch the patient back to one of the other agents with which you can treat every month. My understanding is that Novartis is currently doing studies that would show that there are certain cases where monthly dosing is beneficial and safe, and there would eventually be a label change. But at the present time, that option doesn’t exist.”
At press time, the American Society of Retina Specialists issued a statement that could impact Beovu’s usage. The ASRS stated that, in addition to reports of mild-moderate intraocular inflammation following the administration of Beovu, it’s received reports of 14 cases of vasculitis, “of which 11 were designated as occlusive retinal vasculitis by the reporting provider.” It adds that the etiology of the events is unclear, and long-term outcomes and treatment strategies remains undefined. It does recommend deferring anti-VEGF injection in patients that have any signs of intraocular inflammation.
In response, Novartis issued a statement, part of which notes, “Novartis’ review of this information is ongoing and classification of these ASRS-reported cases by Novartis has not been established, but we are aware of recently reported adverse events following treatment with Beovu. Novartis stands behind the safety and efficacy of Beovu. In addition to our own internal assessment, we have engaged an external safety review committee to further evaluate these post-marketing cases.”
Abicipar
Allergan’s Abicipar is currently being studied in patients with wet AMD. Two-year data from the CEDAR and SEQUOIA clinical studies have shown that four injections of abicipar resulted in the maintenance of visual gains comparable to monthly ranibizumab.3
CEDAR and SEQUOIA are identical global Phase III studies designed to assess the efficacy and safety of abicipar eight-week and 12-week treatment regimens compared with monthly ranibizumab in treatment-naïve patients with wet AMD. Through week 104, patients received abicipar 2 mg every eight or 12 weeks or ranibizumab 0.5 mg every four weeks. At week 104 in the pooled Phase III data, 93 percent of patients in the eight-week abicipar group, 90 percent of patients in the 12-week abicipar group and 94 percent of patients in the four-week ranibizumab group achieved stable vision.
Mean changes in BCVA during year two were similar when compared to year one across all treatment arms.
The incidence of adverse events was comparable between the treatment groups at the end of year two. The rate of new cases of intraocular inflammation in year two was similar for all groups (1.9 percent for patients who received abicipar for both eight and 12 weeks and 1 percent for patients who received ranibizumab).
“Abicipar looks like it’s more durable to some degree than the anti-VEGFs we have been using in practice, and it will potentially be FDA-approved sometime later this year,” observes Dr. Regillo. “There were some issues in Phase III testing with relatively high rates of intraocular inflammation with abicipar, but there’s since been some reformulation, and a recent Phase IIb (MAPLE) study shows lower rates of intraocular inflammation with the latest formulation.”
Faricimab
Genentech’s faricimab is a bispecific antibody designed for the treatment of retinal conditions, simultaneously binding to and neutralizing Angiopoietin-2 (Ang-2) and VEGF-A which, its maker says, may lead to improved and sustained efficacy at longer treatment intervals.
In late 2018, Genentech reported positive results from the Phase II STAIRWAY study for drug faricimab, which evaluated the extended durability of the agent in the treatment of wet AMD.4 At 52 weeks, faricimab patients dosed either every 16 weeks or every 12 weeks demonstrated sustained vision outcomes comparable to ranibizumab dosed every four weeks. Additionally, the company has now completed patient enrollment in the Phase III YOSEMITE and RHINE clinical trials.4 The YOSEMITE and RHINE studies will assess the safety and efficacy of faricimab for the treatment of DME compared to aflibercept. The primary endpoint of each study is the change in BCVA at a year.
“Faricimab is intriguing in the sense that you’re targeting both VEGF and Ang-2, and there’s reason to believe that’s synergistic and will provide vessel stabilization,” notes Dr. Csaky. “When we use anti-VEGF agents, we are primarily changing the permeability aspects of the disease, and that’s why we think we have to continue to inject for long periods of time. As the drug wears off after a time, we’re not necessarily modifying the underlying abnormal blood vessels.
“Faricimab’s Phase II data indicated that there was increased durability compared with an anti-VEGF alone,” he note. “Approximately 70 percent of patients who were given faricimab had no sign of CNV activity at 12 weeks. It’s intriguing that faricimab may induce vessel stabilization and have even more chance for durability, but that remains to be proven.”
The Port Delivery System
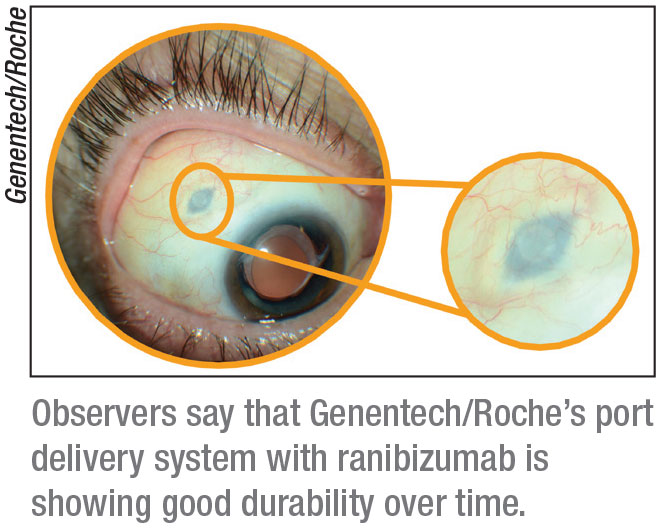 |
The Port Delivery System with ranibizumab is a refillable eye implant designed to continuously release a customized formulation of ranibizumab over a period of months, with the goal of avoiding repeat visits for multiple injections over time. Genentech/Roche have completed patient enrollment in the Phase III Archway clinical trial investigating the PDS in wet AMD.
“It has shown tremendous durability in Phase II testing,” says Dr.
Regillo. “It requires a trip to the operating room to insert, and it’s a device that stays in the eye wall. However, it’s been shown to provide a duration of effect for over a year in most patients. It’s the first long-acting delivery platform. With emerging injectable anti-VEGFs in clinical trials, we might be able to extend the durability by two to four weeks or so, but this PDS is truly long-acting—it slowly releases the drug over many months. In Phase III testing, we’re automatically refilling it every six months, whether they need it or not, but its potential is really quite a bit longer than that.”
Conbercept
Dr. Freund says conbercept (Chengdu Kanghong Biotech) is a molecule very similar to aflibercept. “It was developed in China and, like aflibercept, is a recombinant fusion protein that binds to VEGF-A, VEGF-B, and placental growth factor,” he notes. It’s approved in China.
A study found that conbercept dosing of three initial monthly administrations followed by quarterly treatments was effective in AMD.5
In this prospective, double-masked, multicenter, sham-controlled, Phase III randomized trial, patients with choroidal neovascularization secondary to AMD were enrolled and randomized to the conbercept group or the sham control group. The treatment group received intravitreal injections of conbercept 0.5 mg monthly for the first three months, then quarterly until month 12. The sham group received three monthly sham injections and then three monthly injections of conbercept 0.5 mg, followed by quarterly administrations until month 12.
A total of 114 patients from nine sites in China completed the 12-month study. At the three-month primary endpoint, the mean changes in BCVA from baseline were +9.20 letters in the conbercept group and +2.02 letters in the sham group, respectively. At 12 months, the mean changes from baseline in BCVA letter score were +9.98 letters in the conbercept group and +8.81 letters in the sham group. The most common ocular adverse events were associated with intravitreal injections, such as conjunctival hemorrhage and increased IOP. REVIEW
Dr. Csaky has a financial interest in Allergan, Applied Genetic Technologies Corporation, Gyroscope, Novartis, Ocular Therapeutix, Ribomics and Roche/Genentech.
Dr. Freund has a financial interest in Novartis, Allergan, Bayer and Genentech/Roche.
Dr. Regillo receives research grant support from Allergan, Chengdu Kanghong, Genentech, Novartis and Regeneron, and is a consultant for Allergan, Chengdu Kanghong, Genentech and Novartis.
1. Novartis press release. https://www.novartis.com/news/media-releases/novartis-receives-fda-approval-beovu-offering-wet-amd-patients-vision-gains-and-greater-fluid-reductions-vs-aflibercept.
2. Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: Phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127:1:72-84.
3. https://www.allergan.com/News/Details/2019/10/Allergan%20and%20Molecular%20Partners%20Present%20LateBreaking%20Data%20from%20Phase%203%20Studies%20of%20Investigational%20Ab
4. https://www.gene.com/media/press-releases/14762/2018-10-27/new-stairway-study-data-shows-potential-
5. Liu K, Song Y, Xu G, et al. Conbercept for treatment of neovascular age-related macular degeneration: Results of the randomized Phase III PHOENIX study. Am J Ophthalmol 2019;197:156-167.