The study group, comprising 32 patients in a consecutive, single-surgeon series with dislocated foldable IOLs, was seen from July 1996 through July 2003. The mean age of the patients was 73 years (range, 51 to 87 years), and the group was made up of 16 men and 16 women (15 right eyes and 17 left eyes). The mean duration from dislocation to treatment was 165 days (range, 0 to 1,000 days). The mean interval from implantation to dislocation was 105 days (range, 0 to 1,000 days).
Thirty-two eyes underwent dislocated IOL repair including repositioning without scleral sutures (17 eyes), repositioning using scleral suture (8), and IOL exchange (7). The IOL types included silicone plate haptic IOLs (15 eyes), three-piece acrylic IOLs (12), and others (5). The visual acuity results and complication rates were similar for all IOL types and surgical techniques. Though the study concluded that the preferred approach in the management of dislocated foldable IOLs is sulcus fixation without sutures, IOL exchange techniques are considered a worthwhile alternative.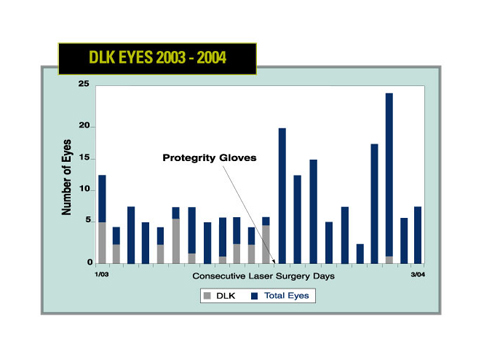
(Retina 2005; 25:576-580)
Smiddy W E.
Surgical Glove–Associated DLK
Researchers in Oregon determined that silicone oil contamination of surgical gloves is a significant risk factor for the development of epidemic diffuse lamellar keratitis.
The retrospective study analyzed an epidemic of diffuse lamellar keratitis in a single surgical center over a three-year period from January 2000 to September 2003. During that time the center noted an increased incidence of epidemic DLK that was unresponsive to changes in instrumentation or technique. Attempts to eliminate the epidemic included switching from detergent instrument cleaners to enzymatic cleaners, and discontinuation of Betadine preps. Despite these modifications, there was no appreciable or dramatic change in the occurrence of DLK.
After becoming aware of the possible association of surgical glove contamination as an etiology of DLK in September 2003, altering the brand of surgical gloves used during surgery resulted in a cessation of DLK. Optical microscopy, scanning electron microscopy-energy dispersive spectroscopy, and Fourier transform infrared spectroscopy were performed on both brands of surgical gloves to allow for comparisons and determine possible surface contaminants responsible for the DLK outbreaks. The analysis of both glove brands revealed extensive silicone oil contamination on the internal and external surfaces of the DLK-associated gloves and insignificant amounts of silicone oil on the external surface of the DLK-free gloves. As a result of these findings, definitive animal studies are planned for the future.
(Cornea 2005;24:699-704)
Hoffman R, Fine IH, Packer M, Reynolds T, Van Bebber C.
Intravitreal Triamcinolone For BRVO CME
Intravitreal triamcinolone may be useful in treating cystoid macular edema due to branch retinal vein occlusion as either a primary or supplemental treatment modality, according to researchers in St. Louis.
A computerized review of the medical records was performed to identify patients with a primary diagnosis of branch retinal vein occlusion, with no prior laser therapy, who had been treated with IVT. The medical records of a total of six eyes of six patients (four females, two males) ranging in age from 53 years to 87 years (mean, 66 years) were reviewed for the study. The time between BRVO and treatment with IVT ranged from two months to 4.7 months. Pretreatment visual acuity ranged from 20/40 to 6/200. The length of follow-up ranged from 107 days to 175 days. Final visual acuity ranged from 20/40 to 3/200.
Three of the six eyes demonstrated improvement in vision. The three patients who did not show improvement were given a second injection. At the final follow-up visit all six eyes demonstrated improved vision from baseline. Five of six (83.3 percent) eyes showed an improvement of >2 lines of vision; one patient had a postop rise in intraocular pressure and required a trabeculectomy. With IVT offering potential treatment of CME due to BRVO, the study suggests that a randomized trial to clarify this role is appropriate.
(Retina 2005; 25:551-555)
Lee H, Shah G.
Improved LASIK Safety
After reviewing case notes retrospectively from 1,000 consecutive LASIK procedures performed by one surgeon between January 2000 and January 2004 at Moorfields Eye Hospital, London, researchers concluded that the risk of visual loss is significantly reduced in contemporary LASIK when compared with premillennial results.
Participants in the study were taken from 1,982 cases with six-month review data in postmillennial Food and Drug Administration trials of LASIK for myopia and astigmatism and 5,203 patients with six-month review data in similar premillennial FDA trials. Of the 1,000 LASIK procedures, there were 899 myopic treatments (spherical equivalent <-10.5 D; average -4.2 D; standard deviations = 1.9 D; astigmatism <4.75 D), 87 hyperopic treatments (spherical equivalent <+4.25 D; average +2.4 D; SD = 0.9 D; astigmatism <3.5 D), and 14 treatments for mixed astigmatism (<4.5 D).
In the clinical series, no case with a preoperative best spectacle corrected visual acuity of 20/20 or better had a final BSCVA of 20/30 at final review (97.5 percent confidence interval, 0 percent to 0.37 percent); four eyes had a BSCVA <20/25 (0.41 percent; 95 percent confidence interval, 0.11 percent to 1.02 percent).
In FDA studies recruiting pre-2000, 1.4 percent of patients lost >2 lines of BSCVA versus 0.6 percent in postmillennial studies (p=0.005); 0.45 percent of patients in premillennial studies lost BSCVA to <20/40 compared with no patients in postmillennial studies (p=0.001); and 1.68 percent of patients in premillennial studies with a postop BSCVA >20/20 had a postop BSCVA <20/25 compared with 0.16 percent in postmillennial studies (p=0.001). With the data demonstrating that there is low risk of serious visual loss associated with contemporary LASIK, researchers suggest that it may be appropriate to modify risk figures for LASIK based on earlier large-scale clinical series.
(Ophthalmology 2005;1375-1380)
Watson S, Bunce C, Allan B.
Travoprost Combo for POAG Or OHT
A fixed combination of travoprost/timolol appears to produce marked reduction of intraocular pressure in a once-daily dosing regimen, researchers concluded based on the results of a multicenter study.
The prospective, randomized, double-masked, parallel group, active-controlled clinical trial included adult patients (n=403) of either gender with open-angle glaucoma or ocular hypertension in at least one eye. To qualify for the trial, the patient's IOP had to be between 22 and 36 mmHg in the same eye at two consecutive eligibility visits.
Results of the study showed mean IOP ranged from 16.2 to 17.4 mmHg with the combination travoprost/timolol, compared with 15.4 to 16.8 mmHg in the concomitant travoprost plus timolol group, from baselines of 23.1 to 25.6 mmHg and 22.9 to 25 mmHg, respectively. The fixed combination of travoprost/timolol lowered IOP by 7 to 9 mm, similar to the IOP reductions noted with concomitant therapy.
The study concluded that the travoprost/timolol fixed combination drop may also provide the benefits of better adherence, reduced cost compared with concomitant therapy, and improved side-effect profile.
(Am J Ophthalmol 2005;140:242-249)
Schuman J, Katz G, Lewis R, Henry C, Mallick S, Wells D, Sullivan EK, Landry T, Bergamini M, Robertson S.




