and Juan V. Espinoza, MD
Since the days of radial keratotomy, retinal complications have been reported after refractive surgery. Surgical advances in myopic correction since then, from automated lamellar keratoplasty to clear-lens extraction and LASIK, have all led to reports of postoperative retinal complications. Such reports include: optic neuropathy,1 flap displacement,2 retinal tears,3 retinal detachments,4 corneo-scleral perforations,5 retinal hemorrhages,5 macular hemorrhages,6 macular holes7 and choroidal neovascular membranes.5
This article will review retinal complications that may occur after refractive surgery, with an emphasis on LASIK.
Retinal Detachments, Retinal Breaks
Several studies have reported individual cases of retinal detachment after LASIK, including a bilateral retinal detachment associated with a giant retinal tear8 and a case of rhegmatogenous retinal detachment.9 Incidence rates for retinal detachment in myopic eyes after LASIK have been reported at 0.25 percent (and a mean best-corrected visual acuity of 20/45 after retinal surgery in one series)5 and 0.22 percent.10 A recent report involved four eyes that had early RRD within three months of LASIK for correction of high myopia.11
Although no cause-effect relationship between LASIK and retinal detachment can be inferred from the studies, the authors' cases suggest that LASIK may be associated with retinal detachment, particularly in highly myopic eyes.
We reviewed the medical records and obtained follow-up information on all patients in our files with RRD after LASIK for the correction of myopia at five institutions.12 A total of 38,823 LASIK procedures (eyes) were performed during the five-year study period. Patients underwent surgical correction of myopia ranging from -0.75 to -29 D (mean: -6 D). Patients were followed for a mean of 48 months after LASIK (range: six to 60 months). 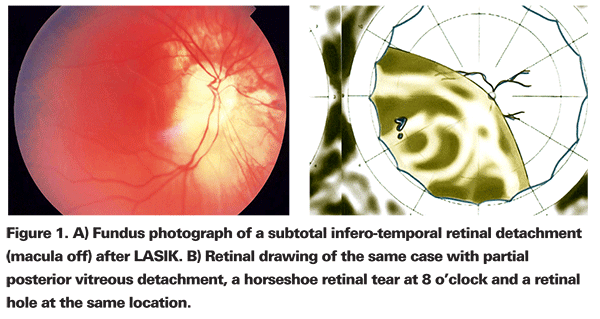
Five vitreoretinal surgeons and 33 eyes (27 patients) that developed RRD after myopic LASIK participated in the study (See Figure 1). Final visual acuity was defined as the BCVA at last follow-up examination, ranging from three to 34 months (mean: 14 months) after vitreo-retinal surgery to repair RRD after LASIK.
In our series, 9 percent (3/33) of eyes that developed a RRD had some kind of enhancement after LASIK. No patient had a history of any other ocular surgery after LASIK. The frequency of RRD determined in our study is 0.08 percent (33/38,823). RRDs occurred between 12 days and 60 months (mean: 16.3 months) after LASIK. Eyes that developed a RRD had from -1.5 to -16 D of myopia (mean: -8.75 D) before LASIK.
Retinal detachments were managed with vitrectomy, cryoretinopexy, scleral buckling, argon laser retinopexy and pneumatic retinopexy techniques. Vitreoretinal surgery to repair RRD after LASIK was performed at a mean of 56 days (range: one day to 18 months) after the onset of visual symptoms. The mean follow-up after retinal surgery was 14 months (range: three to 34 months) and 38.7 percent of the 31 eyes (two patients refused surgery) had a final BCVA of 20/40 or better. Final VA was better than 20/200 in 77.4 percent of eyes. Poor VA (20/200 or worse) occurred in 22.6 percent of eyes. 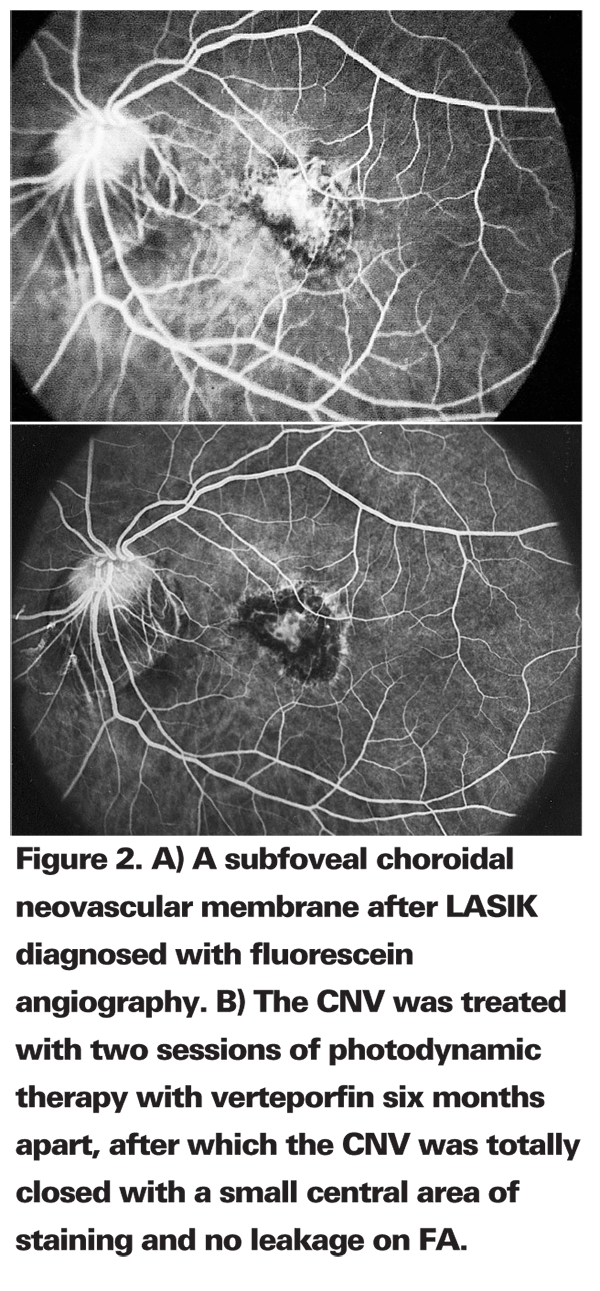
Reasons for poor VA included: the development of proliferative vitreo-retinopathy (n=5); epiretinal membrane (n=1); chronicity of RRD (n=1); new breaks (n=1); displaced corneal flap (n=1); and cataract (n=1). Final VA after RRD surgery improved two lines or more in 51.6 percent (16/31) of eyes. The anatomic success at final follow-up with one surgery was 87.1 percent (27/31). The anatomic success at final follow-up including reoperations was 90.3 percent (28/31). Information regarding VA after LASIK and before the development of RRD was available in 24 eyes; 45.8 percent (11/24) of eyes lost two or more lines of VA after vitreoretinal surgery.
In another recent study, the incidence of retinal disease after refractive surgery (including LASIK) was observed in 9,239 consecutive eyes (5,099 patients). Retinal detachment occurred at a mean 24.6 ±20.4 months after LASIK in 11 eyes (0.36 percent).13
Hemorrhage, Lacquer Cracks, CNV
Macular hemorrhage may occur after LASIK, even in the absence of previously identified risk factors, such as high myopia, pre-existing CNV, lacquer cracks and sudden changes in IOP associated with microkeratome-assisted flap creation.14
In one case of bilateral macular hemorrhage after LASIK, the patient's uncorrected visual acuity was in the 20/50 range at one day postop, and by 17 days after surgery his visual acuity had declined to the 20/200 range. Fundus examination showed multifocal subretinal macular and posterior pole hemorrhages. Fluorescein angiography showed some macular lesions compatible with lacquer cracks.15
The first reported case of unilateral macular hemorrhage following uncomplicated, bilateral, simultaneous LASIK with femtosecond laser flap creation was in a patient without macular pathology.16 The intraocular pressure is only raised to approximately 35 to 40 mmHg during laser flap creation.
We described the first CNV membrane after LASIK.3,17 Others have reported an incidence of 0.1 percent CNV after LASIK and one case after photorefractive keratectomy (1/5,936)18,19 as well as one case of CNV after LASIK in a patient with low myopia.20
While the incidence seems to be very low, the appearance and treatment of CNV was followed by a significant decrease of visual acuity. Two recent reports discuss the characteristics and potential mechanisms of a macular lacquer crack (one with subsequent development of subfoveal CNV) in myopic patients corrected by LASIK.21,22 Lacquer cracks often lead to poor visual outcomes because of CNV and macular atrophy in pathologic myopia. The risk of lacquer cracks in highly myopic patients corrected by LASIK, though uncommon, must be considered.
Another group analyzed the results obtained with photodynamic therapy to treat subfoveal and juxtafoveal CNV in patients with high myopia corrected by LASIK (See Figure 2) or by implanting a phakic intraocular lens.23 They analyzed the results from 14 highly myopic eyes corrected by LASIK (seven cases) or by PIOL implantation (seven cases), which later developed CNV and were treated by PDT with verteporfin. They found that the mean BCVA after refractive surgery was 0.45, SD 0.17 D (range, 0.2 to 0.8), with residual spherical equivalent (RSE) -0.5, SD 1.8 D (range, 1 to 5.5 D). After CNV appearance, BCVA was 0.10, SD 0.19 D (range, 0.025 to 0.7). CNV was treated in all cases by PDT (mean, 2, SD 0.8 treatments). After CNV closure, the mean BCVA improved up to 0.22, SD 0.18 (range, 0.1 to 0.63) (RSE -1.4, SD 1.4 D, range, 0.5 to -4 D). Differences in RSE after refractive surgery and after PDT, and differences between BCVA after CNV appearance and final were not statistically significant (p=0.82 and p=0.06, respectively). 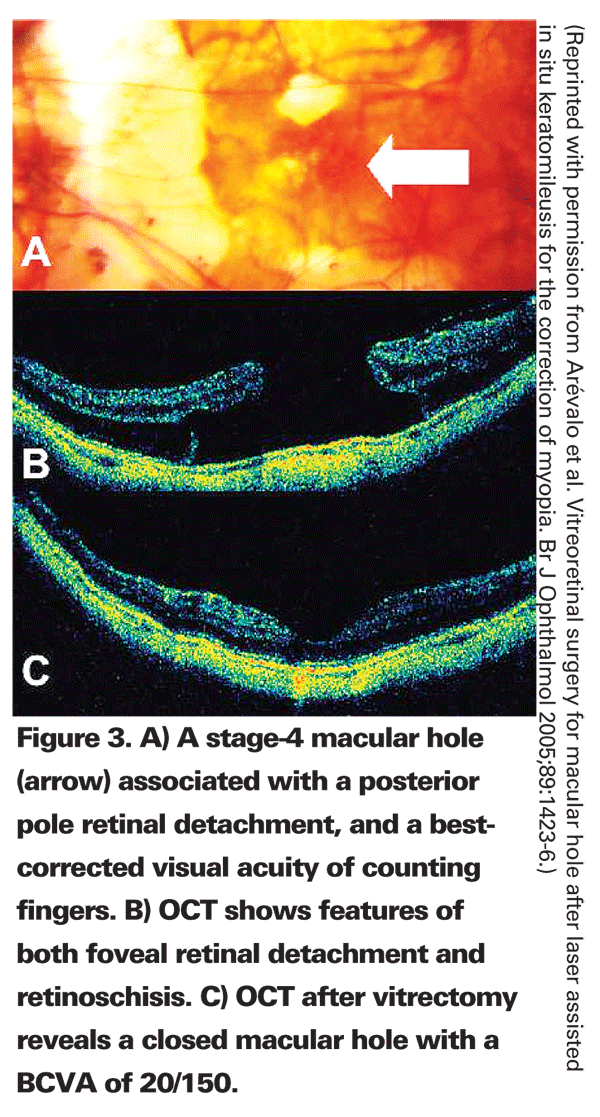
The authors considered PDT to be effective in achieving closure of CNV in myopic patients after refractive surgery without inducing changes in spherical equivalent.
CNV is related to myopia itself and its incidence varies from 4 to 11 percent in patients with high myopia. In addition, lacquer cracks have been found to be associated with CNV in up to 82 percent of cases with myopia.22 Theoretically, when a break in Bruch's membrane occurs, it allows the progression of the neovascular complex under the retina.
The increase in intraocular pressure to levels over 60 mmHg during suction with the microkeratome suction ring up to 4 mm posterior to the limbus may exert traction and compression posteriorly. In addition, we have to consider that the excimer laser is responsible for a shock wave that is transmitted to the eye. These mechanisms may open the gap in Bruch's membrane even more. We believe that in patients with high myopia and lacquer cracks, LASIK should be considered contraindicated, and some other method of refractive surgery should be offered.
Macular Hole
Macular hole may develop in myopic eyes after LASIK or PRK and vitreoretinal interface changes may play a role. In addition to a report of three eyes of three myopic patients that developed a macular hole in one eye after bilateral LASIK or PRK,7 and another single-case report,24 our group recently reported 20 eyes (19 patients) that developed a macular hole in one eye after bilateral LASIK for the correction of ametropia (See Figure 3).25 The macular hole formed between one and 83 months after LASIK (mean: 12.1 months). Eighteen percent of patients were female; all eyes were myopic. PVD was not present before and was documented after LASIK in 55 percent of eyes. A vitrectomy closed the macular hole in all 14 eyes that underwent surgical management. These 20 eyes reflect an incidence of 0.02 percent (20/83,938), and represent the largest series of macular hole after LASIK to date.
Cap Displacement During Vitrectomy
Post-LASIK displacement of a corneal flap is a serious complication. Possibilities include losing the cap, epithelial ingrowth, interface particles and striae in the flap (trauma to the flap may affect the final refractive status). Corneal flap displacement has been described after corneal epithelial debridement during a scleral buckling procedure and vitrectomy.
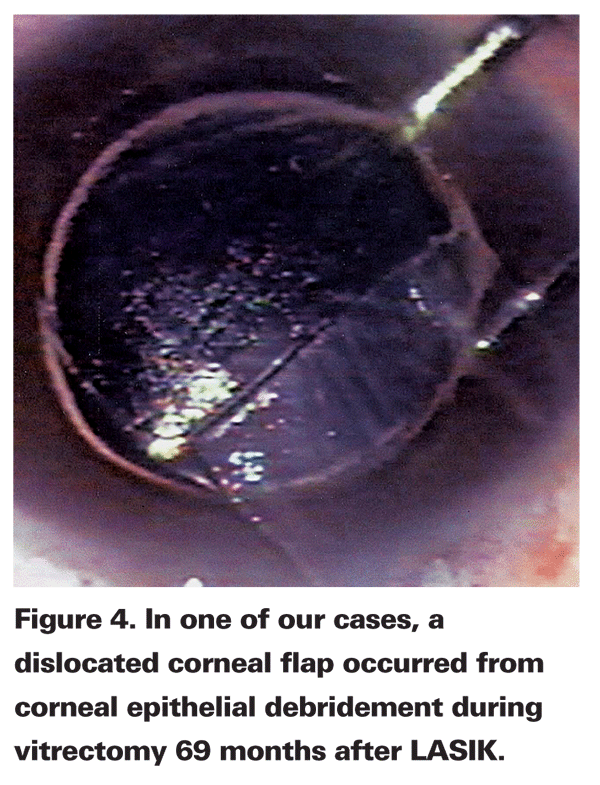
In one of our cases, a dislocated corneal flap occurred from corneal epithelial debridement during vitrectomy 69 months after LASIK. A similar case had been previously reported.26 The case underwent vitreous surgery only four months after LASIK (See Figure 4).
Recommendations for vitreoretinal surgeons when treating an eye with a history of LASIK include avoiding debridement of the corneal epithelium. However, if it is necessary, start corneal debridement nasally and advance temporally (most cases have a nasal hinge). If a displaced corneal flap occurs, initial management includes repositioning of the flap, followed by patching and topical steroids. Refractory cases may require suture fixation. A bandage contact lens may be useful if striae develop. If striae persist, it is an indication to elevate and reposition the flap.
Final Considerations
Based on published data, we cannot determine whether prophylactic treatment is indicated. At this time it is not possible to scientifically determine whether peripheral retinal lesions should be treated differently from standard practice just because a patient is to undergo refractive surgery. Most practitioners suggest that patients scheduled for refractive surgery be carefully examined with indirect ophthalmoscopy and scleral depression under pupillary dilatation to detect any myopic peripheral lesion that requires treatment before refractive surgery is performed. One could argue that this is prudent in myopes whether they undergo refractive surgery or not; given the potential of the procedure to exacerbate pre-existing pathology, it might be wise to treat such pathology more aggressively.
No cause-effect relationship between refractive surgery and retinal complications can be proved from previous studies. However, it is very important to inform patients that refractive surgery only corrects the refractive aspect of myopia, and that myopic eyes have the potential for serious complications.
Drs. Arévalo and Espinoza practice at the Clínica Oftalmologica Centro Caracas, where Dr. Arévalo is the director as well as the director of the Retina and Vitreous Service. Contact him at arevalojf2020@gmail.com.
1. Bushley DM, Parmley VC, Paglen P. Visual field defect associated with laser in situ keratomileusis. Am J Ophthalmol 2000;129:668-671.
2. Lee AG. LASIK-induced optic neuropathy. Ophthalmology 2002;109:817;author reply 817.
3. Lin SC, Tseng SH. Prophylactic laser photocoagulation for retinal breaks before laser in situ keratomileusis. J Refract Surg 2003;19:661-665.
4. Ruiz-Moreno JM, Perez-Santoja JJ, Alio JL. Retinal detachment in myopic eyes after laser in situ keratomileusis. Am J Ophthalmol 1999;128:588-594.
5. Arévalo JF, Ramírez E, Suarez E, et al. Incidence of vitreo-retinal pathologic conditions 24 months after laser-assisted in situ keratomileusis (LASIK). Ophthalmology 2000;107:258-262.
6. Zaldivar R, Davidorf JM, Oscherow S. Laser in situ keratomileusis for myopia from -5.50 to -11.50 diopters with astigmatism. J Refract Surg 1998;14:19-25.
7. Chan CK,
8. Ozdamar A, Aras C, Sener B, et al. Bilateral retinal detachment associated with giant retinal tear after laser-assisted in situ keratomileusis. Retina 1998;18: 176-177.
9. Stulting RD, Carr JD, Thompson KP, et al. Complications of laser in situ keratomileusis for the correction of myopia. Ophthalmology 1999;106:13-20.
10. Aras C, Ozdamar A, Karacorlu M, et al. Retinal detachment following laser in situ keratomileusis. Ophthalmic Surg Lasers 2000;31:121-125.
11. Farah ME, Hofling-Lima
12. Arévalo JF, Ramírez E, Suarez E et al. Retinal detachment in myopic eyes after laser in situ keratomileusis. J Refract Surg 2002;18:708-714.
13. Ruiz-Moreno JM, Alio JL. Incidence of retinal disease following refractive surgery in 9,239 eyes. J Refract Surg 2003;19:534-547.
14. Kim HM, Jung HR. Laser assisted in situ keratomileusis for high myopia. Ophthalmic Surg Lasers 1996; 27:508-511.
15. Luna JD, Reviglio VE, Juarez CP. Bilateral macular hemorrhage after laser in situ keratomileusis. Graefe's Arch Clin Exp Ophthalmol 1999;237:611-613.
16. Principe AH, Lin DY, Small KW, et al. Macular hemorrhage after laser in situ keratomileusis (LASIK) with femtosecond laser flap creation. Am J Ophthalmol 2004;138:657-659.
17. Arévalo JF. Posterior segment complications after laser-assisted in situ keratomileusis. Curr Opin Ophthalmol 2008;19:177-84.
18. Ruiz-Moreno JM, Artola A, Ayala MJ, et al. Choroidal neovascularization in myopic eyes after photorefractive keratectomy. J Cataract Refract Surg 2000;26:1492-1495.
19. Ruiz-Moreno JM, Perez-Santonja JJ, Alio JL. Choroidal neovascularization in myopic eyes after laser-assisted in situ keratomileusis. Retina 2001;21:115-120.
20. Saeed M, Poon Wallace, Goyal S, et al. Choroidal neovascularization after laser in situ keratomileusis in a patient with low myopia. J Cataract Refract Surg 2004;30:2632-2635.
21. Maturi RK, Kitchens JW, Spitzberg DH, et al. Choroidal neovascularization after LASIK. J Refract Surg 2003;19:463-464.
22. Ruiz-Moreno JM, Montero J, Alio JL. Lacquer crack formation after LASIK. Ophthalmology 2003; 110:1669-1671.
23. Amat-Peral P, Lugo F, Montero JA, de-la-Vega C, Arévalo JF, Alió Y Sanz JL. [Photodynamic therapy with verteporfin in choroidal neovascularization after refractive surgery]. Arch Soc Esp Oftalmol 2008;83: 539-44.
24. Ruiz-Moreno JM, Artola A, Perez-Santonja JJ, et al. Macular hole in a myopic eye after laser in situ keratomileusis. J Refract Surg 2002;18:746-749.
25. Arévalo JF, Mendoza AJ, Velez-Vasquez W, et al. Full-Thickness Macular Hole after LASIK for the Correction of Myopia. Ophthalmology 2005;112:1207-1212.
26. Chaudhry NA, Smiddy WE. Displacement of corneal cap during vitrectomy in a post-LASIK eye. Retina 1998;18:554-555.