VRmagic GmbH (Mannheim, Germany) unveiled its virtual-reality slit lamp to the United States in October 2018 at the American Academy of Ophthalmology meeting in Chicago. The company has integrated its virtual-reality software—allowing users to conduct routine examinations and diagnose rare conditions—into the hardware of a Haag Streit BQ 900 slit lamp (Köniz, Switzerland). Christopher S. Sales, MD, MPH, assistant professor of ophthalmology, cornea & refractive service at Weill Cornell Medical College in New York City, was impressed. “That slit lamp is ridiculous!” he says. “I swear it’s as if there’s a patient in front of you; it blinks and everything.” Dr. Sales has devised a simpler simulator of his own: an artificial iris diaphragm that helps surgeons learn to shallow the anterior chamber to facilitate graft manipulation in DMEK. A brief description of both devices follows, framed by consideration of how ophthalmology might imbue simulation training with an emotional component to help doctors learn how to keep their cool in the face of stress.
VRmagic Eyesi Slit Lamp
In addition to learning how to handle the slit lamp itself, users have the opportunity to expedite their learning by gaining lifelike exposure to pathologies they might not otherwise see during the course of medical study or residency, says Dr. Markus Schill, CEO of VRmagic. “Your learning speed is typically governed by the flow of patients,” he says.“One of the biggest advantages of having cases provided through a simulator is that you can sit down and experience a case independent of the patient flow. We have conditions that should present in a regular exam, but we also have very rare things that one might not necessarily see in regular practice.”
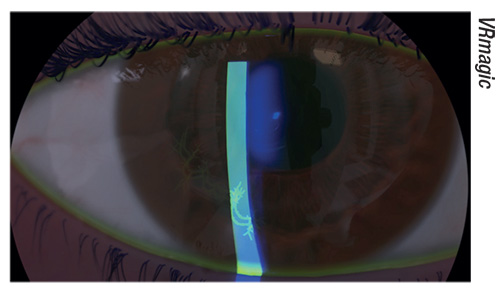 |
| The Eyesi Slit Lamp simulator will feature both routine and unusual cases. Shown here: herpes. |
VRmagic also manufactures indirect and direct ophthalmoscope simulators. Of the indirect ophthalmoscope, Dr. Schill says the software offers a menu of rare pathologies for users to diagnose in addition to routine cases. “We also have very rare cases, which we call global health cases, which are in a package showing ocular manifestations of pathologies we would not regularly see in a typical Western hospital, like syphilis or Zika. This case curriculum will be the same in the slit lamp. Right now, we only have 15 cases, but that’s obviously going to increase,” he says.
The Eyesi Slit Lamp simulator also provides feedback on individual performance and progress. “It’s a multi-user system and everybody has individual logins,” says Dr. Schill. “The training history is saved, and it can draw plots showing skill improvement.”
He says the slit lamp simulator is slated for release in summer 2019. “It’s a staged release: In summer 2019 it will have anterior segment; one year later in summer 2020, we’ll add the retina exam, and in the summer of 2021, we will add gonioscopy.”
An Iris Diaphragm for DMEK
“It’s not anything fancy,” says Dr. Sales of his creation. “It’s just putting a piece of rubber on top of the pedestal of an artificial anterior chamber.” Using a latex glove, one 3-mL syringe and a donor DMEK tissue punched from a corneoscleral cap, an 18-mm latex diaphragm is trephined from the glove and placed onto the artificial anterior chamber pedestal. The corneoscleral cap is mounted over the latex diaphragm, which acts as an “iris” when the space between the latex diaphragm and the mounted cornea is filled with saline to represent a pressurized anterior chamber; the posterior segment of the model is pressurized by air from a syringe.
“The fundamental reason the eye behaves as it does during any kind of anterior segment surgery is that the iris and the lens create a separation between the back of the eye and the front of the eye,” he says. “They’re essentially two separate systems. If you let pressure out of the front of the eye, the pressure in the back of the eye remains comparatively higher, so it pushes everything forward. What makes the anterior chamber ‘shallow’ is the forward movement of the iris-lens diaphragm, not the backward bowing of the cornea. Likewise, if you increase the pressure in the front of the eye, it’ll push the iris backward, which will deepen the anterior chamber.
“When you pressurize the posterior segment and then let fluid out of the front of the artificial anterior chamber, you can bring that latex diaphragm forward, which effectively makes the anterior chamber shallow, and at the same time keeps the cornea in the taut convex configuration needed to see what’s going on with the DMEK graft,” Dr. Sales explains. “Without the latex diaphragm, it’s impossible to shallow the artificial anterior chamber. All that happens is the cornea collapses into a wrinkly concave configuration, which is actually quite similar to what happens if you try to shallow a human aphakic eye that has undergone vitrectomy.
 |
| Close-up view of a donor corneoscleral cap mounted onto Dr. Sales’ device. |
“Ironically, when you’re really doing DMEK surgery, it’s easy to shallow the anterior chamber: You just let fluid out of the front of the eye by burping one of the incisions,” he says. “That’s not the part that seems to trip surgeons up when learning DMEK. In my experience, the challenge seems to be recognizing that the anterior chamber needs to be far more shallow than you would otherwise make it for other anterior- segment surgeries.
“Before this model was introduced, the difficult part in the wet lab was not only recognizing when the anterior chamber was shallow enough but also the very act of making it shallow in the first place,” he explains. “Cadaveric human eyes behave a lot like aphakic vitrectomized human eyes and are notoriously touchy to work with. Most of the time when you try to shallow the anterior chamber in a cadaveric human eye, all you get is a wrinkly concave cornea. This is why it used to be rare for surgeons to finish a DMEK wet lab with a clear picture of what a ‘shallow anterior chamber’ really looks like and how DMEK tissue behaves in that setting.
“I love it when I’m in a wet lab with surgeons and the chamber is just a little too deep,” Dr. Sales continues. “All I do is walk over and put a little bit of pressure into the back of the artificial anterior chamber, which brings the diaphragm up and shallows the chamber a little bit. Then I tell them to do the same thing that they were doing, and ask them how much easier it becomes. It shows surgeons that the ‘DMEK Dance’ is more about thinking than it is about dexterity. Most people can figure out how to tap on a cornea in the span of a few minutes. It’s the recognition of the conditions that make that tapping effective that matters, and it’s the simulator that allows you to show them.”
| Surgical CME Videos Enter the 3-D Era |
| For the first time—thanks largely to the work of Richard Mackool, MD, medical director of The Mackool Eye Institute and Laser Center and senior attending surgeon at the Mt. Sinai New York Eye and Ear Infirmary and New York University Medical Center—modern video technology will soon allow members of U.S. ophthalmology residency programs to view ultra-high-definition 3-D CME-approved surgical videos. The videos will be observable using 3-D viewing equipment that will be offered at no cost to all 120 qualified residency departments through an Academic Independent Medical Education grant from Alcon. “Enabled users will be able to log into a new website known as MackoolOnline3Dcme.com, which is a monitored, continuing education program, and either view the ultra-high-definition videos online or rapidly download them for later viewing offline,” Dr. Mackool explains. “The user can then watch the videos using special LED high-definition 3-D video glasses containing a small HD monitor in each eyepiece. The view through the goggles is identical in every way to the surgeon’s view. The goggles also have earbuds, so the viewer can listen to the narration while watching the video. “Two viewing glasses will be provided to each program so that, for example, a faculty member and a resident can watch simultaneously,” Dr. Mackool continues. “The system is entirely portable, like a briefcase. The cost of providing large-screen monitors to every residency program would have been prohibitive, but ultra-high-resolution video glasses work perfectly, and are both portable and cost-efficient. The high-definition cameras used to make the videos are manufactured by Sony, and an in-kind grant from The Mackool Eye Institute Research Foundation enabled the creation of the 3-D website.” Dr. Mackool says that, to the best of his knowledge, no other medical specialty has ever done anything like this before. The CME program will be launched in early 2019. Every qualified U.S. residency program will receive an invitation to participate, and there are no subscriber or other fees. (Dr. Mackool consults for Alcon, but has no financial interest in this 3-D service.) —Christopher Kent |
Teaching Emotional Control
“Virtual-reality simulation systems are nothing short of amazing, but they’re very sterile,” Dr. Sales observes, adding that unmanaged emotional pressure can hamper even the most technically skilled hands. “With a VRmagic simulator, for example, you can go through a complication run for a ruptured posterior capsule: It’s great because you can learn to do A, then B, then C. But trainees still need to get a feel for what it’s like to have that cortisol response, when they start to have a tremor and feel nervous with that vitrector in the eye, so they can learn how to overcome it.”
Dr. Sales cites Noa Kageyama, a professor of performance psychology at Juilliard, and his blog, the Bulletproof Musician (https://bulletproofmusician.com). “We’ve studied this at our institution in conjunction with Juilliard,” he says. “Juilliard has a specific program where they teach fantastic concert musicians who haven’t had the training, or lack the internal wherewithal, to get a hold of their psyches. When they practice at home, it’s easy for them to hit a home run; but when they sit down for an audition with their peers or they get onstage, despite their ability, they can’t always get their heads together sufficiently to do their best when it counts.”
Dr. Sales designed a study with Drs. Noa Kageyama and Ana Alzaga (Weill Cornell Medical College department of Ophthalmology) and submitted a poster to the Association of University Professors of Ophthalmology describing a small pilot investigation. “In our study, we found that by exposing residents to some stress during practice on a virtual-reality simulator, they didn’t have as much of a delta between how they did in training and how they did under real stress,” he explains. “We basically made our residents do simulated procedures in front of their peers and also a guest audience. We ultimately brought in the chairman, but we didn’t tell them that he was coming. All of a sudden, the stakes were raised because we were scoring them in front of the chairman. It’s not exactly what happens in the operating room, but it was one way to artificially introduce stress and give them a chance to learn how to manage it in their own way.”
Simulators are valuable training aids precisely because mistakes have no grave consequences. But one of the very things that makes them great may also be a limitation, notes Dr. Sales. “You’re going to have the Michael Jordans of the world, and you’re going to have the nervous nellies of the world. Can you get the nervous nellies to be at least average in their ability to get their heads together when it counts? Part of that comes with experience, but I think it’s also a question of how we train,” he concludes. REVIEW
Dr. Sales has a financial interest in his device through Cornell University, which has licensed the technology to Network Medical.