 |
| Catalys surgeons can alter the femto fragmentation cube sizes, using smaller cubes to achieve greater softening. |
Catalys
The first aspect of the AMO Catalys laser that surgeons will put into action is called the Liquid Optics patient interface. This is the part of the system that fits on the patient’s eye and allows the laser to dock and deliver its energy accurately.
The Liquid Optics interface is a small reservoir that’s placed onto the patient’s eye and filled with balanced salt solution. The laser is then brought down onto this liquid/cornea interface. The company says the liquid helps smooth out corneal imperfections that could affect the beam. “It’s an elegant way to dock with a femtosecond laser,” says Doug Koch, MD, chair of the ophthalmology department at the Baylor College of Medicine in Houston. “It’s easy to place on the eye and it’s not threatening to the patient; it’s just a small suction ring being placed rather than a large gantry coming down. Also, it raises the intraocular pressure minimally, raising it by 5 mmHg, 10 mmHg at the most.”
Once the machine is docked, the system helps the surgeon visualize and plan the treatment using a 3-D optical coherence tomographer and an Integral Guidance system. The OCT images such structures as the anterior and posterior cornea, the iris and the pupil. “It does a great job figuring out the tilt and position of the lens,” Dr. Koch avers. It will adjust the photodisruption pattern based on any tilt, helping to avoid complications from disrupting the wrong structures. “The advantage is that you can feel confident going to a deeper level with the laser,” Dr. Koch adds. “You can get a consistent, deep softening of the lens. It consistently goes to a good depth and leaves very little residual lens material. In particular, you’re very rarely left with any residual epinucleus, because a portion of the epinucleus is softened. This eliminates the sometimes frustrating step of having to remove the epinucleus after you’ve removed the lens itself.”
Surendra Basti, MD, associate professor of ophthalmology at Northwestern’s Feinberg School of Medicine, says the instantaneous nature of the OCT is a boon, as well. “The most recent version of the software has yielded a clear improvement in managing cataracts,” he says. “It shows a real-time OCT image. Let’s say, for instance, that the OCT was taken and you then make some adjustments. When you’re about to make the incision, you can quickly check a real-time image. So, if the patient’s eye has moved in the interim between your initial imaging and now, it will show you and allow you to make an adjustment.”
As with other femtosecond cataract technology, Catalys surgeons have been discovering which patterns work best for fragmenting the nucleus. The device has different options to choose from to help them select the pattern that’s right for them. “You can segment it into four, six or eight pieces,” explains Dr. Koch. “You also have a lot of flexibility with regard to softening: You can do no softening, you can do large cubes, medium cubes or small cubes. If you create quadrants, you can determine the amount of lens that you want to soften, leaving some unsoftened area right in the center along the vertical and horizontal splitting line. The advantage to such a pattern is that leaving a little lens material can make it easier to aspirate and pull that quadrant to the center; it’s harder to pull it to the center if you’re pulling directly on the cubes.”
Dr. Basti says not many lens-softening cubes are necessary in less-dense lenses. “In a lens such as a 1+ nuclear sclerotic cataract, what you really need is some weakening of the lens so you can break it into pieces,” he says. “If the cataract is a 3+, though, you need weakening of the lens as well as softening, so you’d do well to break it into cubes so the total amount of phaco energy is lower. For a dense nucleus, I use 350-µm cubes.”
Overall, Dr. Koch says the strides the Catalys has taken since its inception are noticeable. “The flow of the Catalys is very quick now,” he says. “My typical suction times are now around two minutes. This includes placing the ring with the bed outside the laser, moving it in, docking and performing the treatment.”
LENSAR
The past year saw a sizeable upgrade to the LENSAR system in terms of a new software bundle, called Streamline, that increases the system’s functionality.
“Streamline is enough of a change that it required Food and Drug Administration studies to achieve clearance,” says Largo, Fla., surgeon Robert Weinstock, who performed some of the studies for the FDA clearance trial. Streamline involves five new features, two of which are built into the LENSAR, and three that require the i-Optics Cassini Corneal Shape Analyzer device.
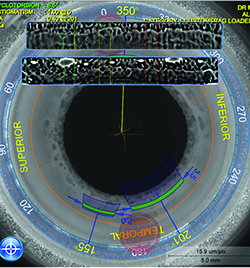 |
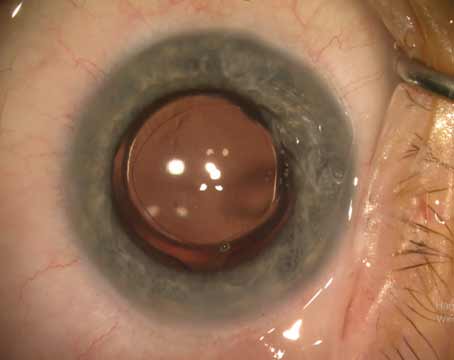
| LENSAR’s Streamline iris registration can compensate for cyclorotation during the incision-planning phase of the procedure. |
The other Streamline features center on the fact that the laser can now interface with the Cassini Analyzer. “The interface involves taking preop corneal measurements, pupillometry and slit-lamp photos with the Cassini, then feeding them wirelessly to the laser,” explains Dr. Weinstock. “This accomplishes several things: One, it can automate limbal relaxing incisions and corneal wound placement with automated nomograms that the surgeon can customize. For instance, if the patient has 1 D of cylinder at 90 degrees on the Cassini, and the system is programmed to understand that the surgeon has a surgically induced astigmatism of 0.3 D at 180, there will be an automatic nomogram adjustment that will be waiting there for the surgeon when he gets behind the laser and enters the patient’s name.
“On top of that,” adds Dr. Weinstock, “since the Cassini takes an infrared image, iris registration software was created. Using the Cassini’s undilated image of the iris and the image provided by the laser once it’s docked, the laser registers the eye based on the iris architecture. Software compensates for head tilt, cyclotorsion and movement that might occur during docking. This allows you to find the ‘true North’ and the true primary meridians, which can then be reference points for your LRIs and corneal wounds. This feature can potentially improve the accuracy, predictability and outcomes of astigmatic correction.” Also, if the surgeon owns a TrueVision 3D system, the LENSAR/Streamline can interface with that, too.
Dr. Weinstock says his femtosecond laser has become key to managing certain patients. “It’s become so integral to managing eyes with pseudoexfoliation and/or Fuchs’ dystrophy and dense cataracts that, if such a patient came in and couldn’t afford the astigmatism correction part of laser cataract surgery, I’d go ahead and do the laser and eat the cost,” he says. “I know it will lead to a better outcome because that sick eye is going to heal much better and have less risk for complications both intraop and postop.”
LenSx
The LenSx laser (Alcon) has replaced the solid applanation plate it originally used with a patient interface known as SoftFit. It also uses a high-res OCT for imaging of the anterior chamber and to help program the locations of corneal incisions.
“The older, original interface resulted in a certain percentage of corneas having striae due to compressional corneal folds,” explains LenSx user Ming Wang, MD. “These striae inhibited the laser energy that passed through them, resulting in capsulorhexes with certain parts not being cut or having a tissue tag. This was an issue because, even if you tried completing the capsulotomy with Utrata forceps, some of these tags would have a small radial component that could increase the risk of a radial tear-out. The compressional striae can also impede the OCT too, creating a shadow on the image.
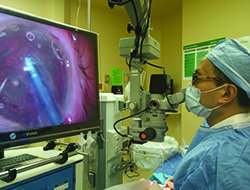 |
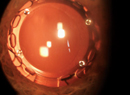
| Ming Wang, MD, uses a “phaco-less” removal of lens fragments after having pre-chopped them with the LenSx femtosecond laser. |
The OCT’s resolution has improved since the LenSx was first introduced, as well. “In terms of resolution, it’s tremendously improved,” says Dr. Wang. “You want a good signal-to-noise ratio when you’re reaching the backside of the lens and trying to gauge how deep your laser should go with respect to the distance to the posterior capsule. A high-resolution OCT improves this visualization, especially through a dense lens.
“There are other software improvements also,” Dr. Wang adds. “This includes auto-recognition/auto-centration. Once the suction ring’s on, you want to reduce the time it takes to set the laser parameters and the actual photodisruption. There’s a big difference between doing all the steps in 20 to 30 seconds versus 40 or 50, and with an increased time come significant increases in the risk of suction loss. With auto-recognition software, you hit the button and the system immediately centers the capsulorhexis on the pupillary center. It also helps center the nuclear fragmentation and the laser AK. The auto-recognition of the anterior capsule has improved, too. You can input 300 µm anterior to the anterior capsule and 250 µm posterior to it and it will automatically recognize the capsule and set that zone of laser treatment automatically.”
For astigmatism management, the LenSx can receive registration data from the Verion system. “Rather than eyeballing it under the microscope or using a Mendez ring/protractor device to determine the degrees,” Dr. Wang says, “the surgeon can perform the Verion digital imaging preop. That data is sent to the LenSx laser and the system can place the LAK incision exactly where the preop image dictates. The beauty of it is, once the eye is registered, the LenSx can do the LAK right away. With manual marking, you can have an error, but this automatically places the LAK incisions based on the digital markings. It helps avoid error.”
Victus
Bausch + Lomb’s Victus femtosecond laser features a live-action, swept-source OCT and docking technology that helps reduce the risk of eye tilt and distortion. The Victus is the only femtosecond cataract laser that can also make a LASIK flap (the LenSx is approved for them but doesn’t currently do them).
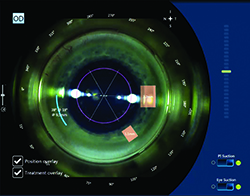 |
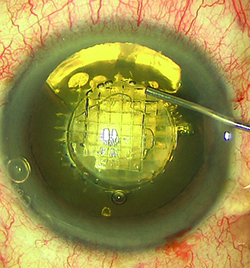
| Originally created for corneal applications, surgeons say the Bausch + Lomb Victus laser is proficient at making corneal incisions, such as entry wounds, paracenteses and limbal relaxing incisions (above). |
For the docking portion of the procedure, the Victus actually has a “wet-to-dry” approach. It uses a curved interface with a thin layer of fluid for internal eye operations (such as the capsulotomy) and a dry, direct-contact interface for corneal applications (such as corneal incisions). It also has pressure sensors to help the surgeon know when a good dock is achieved. “Having a fluid interface allows the system to more accurately detect, measure and treat structures for the anterior capsulotomy and the femto fragmentation,” Dr. Whitman says. “The pressure sensors are helpful; as you bring the eye up into the patient interface using your hand control, the sensors begin at the bottom of the screen in red, meaning the docking is not yet satisfactory, to yellow and then green. It’s colormetric and easy to see.
“When the internal treatments are done,” Dr. Whitman continues, “we raise the bed, push out the fluid, and then it’s dry again. We can then use the dry interface for the entry and arcuate incisions. It’s important to recall that the system was originally created for use on the cornea, so it’s proficient at treating it.”
The new Victus software also provides a variety of patterns for femto-fragmentation. “For myself, I prefer to do six radials for softer nuclei, say 2+ or less, and when there are 3+ and harder lenses, I like to create three to four circles within a 4-mm radius, and then six radials mixed with that so I have an interesting chop pattern,” says Dr. Whitman. “Whenever I hear arguments about whether this technology is worthwhile, I just say, ‘Do a dense cataract with this and you’ll be sold,’ because it’s made my dense cataracts much easier to handle.” REVIEW
Drs. Koch and Basti are consultants to AMO. Dr. Weinstock is a consultant to LENSAR and Dr. Whitman is a consultant to Bausch + Lomb. Dr. Wang has no financial interest in any of the products discussed.