Adequate visualization is paramount to any successful vitreoretinal surgery. Although visualization may be impacted by various anatomic factors, the viewing system that the surgeon uses intraoperatively plays a significant role in achieving adequate visualization and, ultimately, successful surgical outcomes. The initial viewing systems that were used early in the development of pars plana vitrectomy provided a very limited angle of visualization. Today, however, we have a variety of viewing system options that provide excellent wide-angle visualization. This article will review these systems, which range from contact and non-contact wide-angle viewing systems to heads-up displays for 3-D vitrectomy.
Contact vs. Non-contact
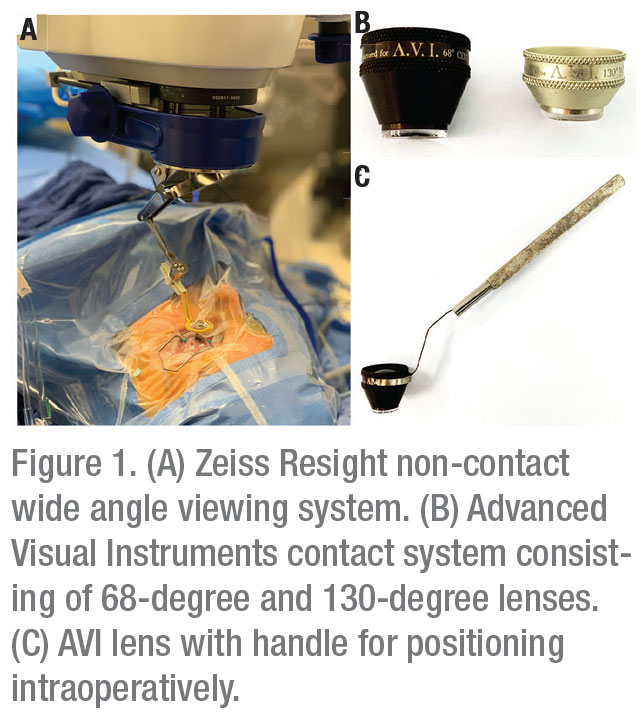 |
Wide-angle viewing systems are routinely used in vitreoretinal surgery, and both contact and non-contact systems have benefits and limitations.
The popular Advanced Visual Instruments (AVI) contact system was developed in 1989; it consists of two lenses that provide excellent clarity for both macular pathology and the periphery, extending the field of view to 130 degrees (Figure 1B and 1C)1 The clarity provided by contact systems is the result of their ability to compensate for corneal aberrations. However, scleral depression can be more challenging with a contact lens in place. These systems are associated with an increased rate of corneal abrasions, and a capable assistant is required to hold the lens in place.2,3 Modified self-stabilizing lenses have been developed to reduce, but not completely eliminate, the need for a skilled assistant to hold the lens intraoperatively.4,5
Alternatively, non-contact systems eliminate the need for an assistant to hold the lens, but provide a slightly smaller viewing angle and reduced clarity in comparison to contact systems.3 Previously, viewing system selection was based on surgeon training and preference, as there was minimal data available comparing anatomical and functional outcomes of contact versus non-contact viewing systems. In 2012, an abstract presented at the Association for Research in Vision and Ophthalmology compared outcomes of 33 eyes that underwent pars plana vitrectomy with either a non-contact viewing system (the Optical Fiber Free Intravitreal Surgery System, or OFFISS) or a contact lens system, for a variety of retinal pathologies including retinal detachment, macular hole, lens dislocation, epiretinal membrane, vitreous hemorrhage, intraocular foreign body removal and silicone oil removal.6 Despite the small sample size, this report found no statistically significant difference in operative time, postoperative visual outcomes, postoperative complications such as hypotony, intraocular pressure elevation, choroidal detachment or hyphema. However, there was more vitreous hemorrhage reported in the non-contact group.6
We recently compared anatomical and visual outcomes of patients undergoing non-complex retinal detachment repair with either a contact or non-contact viewing system as part of the Primary Retinal Detachment Outcomes Study.7 This multicenter, interventional, retrospective, comparative study is the largest comparative study of contact versus non-contact viewing systems published to date.7 In this study, we analyzed 2,256 eyes, of which 1,893 were repaired using a non-contact system and 363 were repaired using a contact-based system.7 We found no statistically significant difference in single surgery anatomic success at three months postoperatively, or at the final follow-up visit (with an average follow-up of 337 days for the non-contact group and 229 days for the contact group).7 Postoperative visual acuity was better in the contact group (logMAR 0.345 [20/44 Snellen]) compared to the non-contact group (logMAR 0.475 [20/60 Snellen]).7
There were multiple factors that could explain the difference in final visual acuity: better preoperative visual acuity in the contact group (logMAR 0.998; Snellen equivalent 20/199 in the contact group versus logMAR 1.134; Snellen equivalent 20/272 in the non-contact group); surgeons using the contact system were highly experienced (84 percent of surgeons in the contact group had over 15 years of experience); and longer follow-up in the non-contact group, which may have resulted in the formation of more mature cataracts.7 However, when controlling for confounding variables in a multivariate analysis, viewing system was no longer statistically significant.7 Therefore, given the excellent outcomes of both viewing systems, we recommended surgeons continue to operate with their preferred system (contact or non-contact).
 |
Non-contact Systems
As detailed in the sidebar on page 68, the development of the first non-contact viewing system—the BIOM—in 1987 provided an alternative to contact-based viewing systems and eliminated the need for an assistant to hold the lens in place intraoperatively.8 Since the development of the BIOM, multiple other non-contact wide-angle viewing systems have been introduced (also mentioned in the sidebar).8 In 2012, a report compared five different non-contact wide angle viewing systems: BIOM (Oculus); Merlin (Volk Optical); OFFISS (Topcon); Resight (Carl Zeiss Meditec); and the Peyman-Wessels-Landers semi-wide-angle viewing system (Ocular Instruments).3
Of the five systems evaluated, the OFFISS obtained the widest angle of view (95 degrees in fluid and 125 degrees in air).3 The report found the Resight system to be the least affected by pupil size and to have the least amount of image distortion when viewing the peripheral retina (Figure 1A). Also, the report concluded the Resight provided the clearest view of the posterior pole.
 |
Heads-up 3-D Viewing
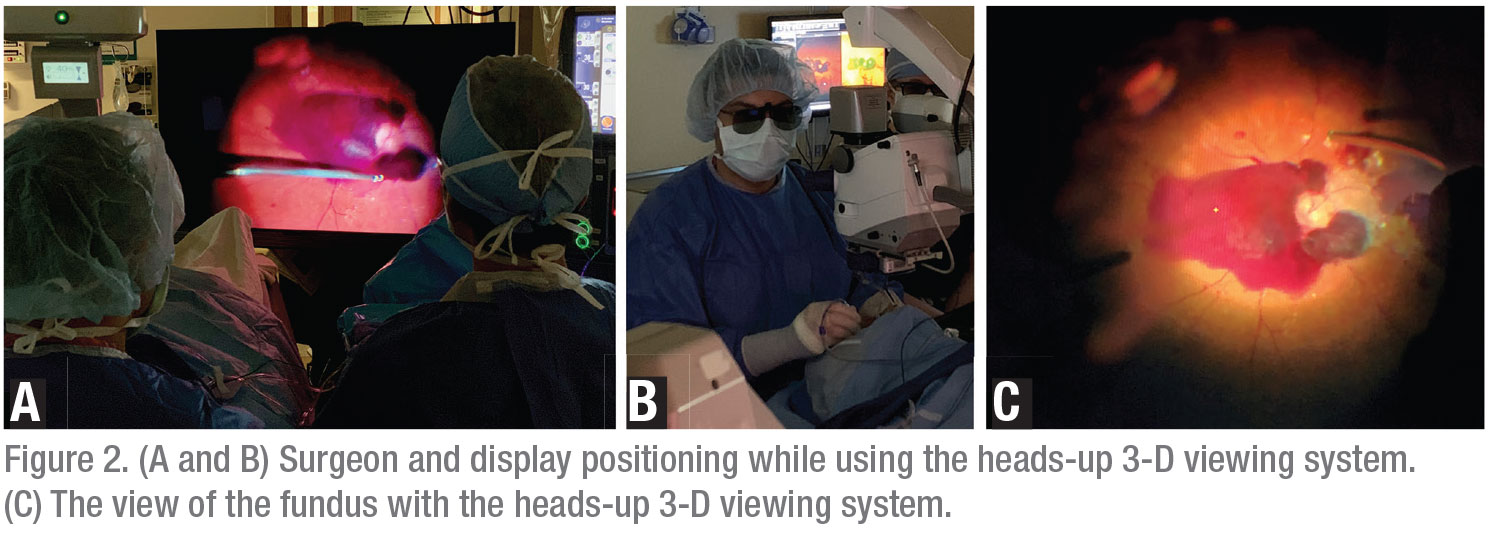
Heads-up 3D viewing systems were initially used in anterior segment surgery and later adapted for use in vitreoretinal surgery.9 Systems include the Ngenuity (Alcon), the Sony HD Medical Display and the Zeiss Artevo 800. In the case of Ngenuity, the system usually consists of a large display (around 46-inch) placed 1.5 meters from the surgeon and doesn’t require viewing through the microscope eyepieces (Figures 2A and 2B).9
An initial report on a heads-up 3-D system in 2016 evaluated speed, resolution, ease of operation and ergonomics for 20 participants performing three different tasks using the system as well as a traditional microscope.9 The participants found the heads-up viewing system to have better ergonomics than traditional vitrectomy viewing systems.9 In addition, the majority of participants found the image to be sharper and believed tasks could be performed equally fast or faster than with traditional vitrectomy viewing systems (Figure 2C). One limitation noted was decreased resolution compared to traditional viewing systems, but the participants compensated by increasing magnification. This initial report also assessed the outcomes of 43 cases of vitrectomy with ILM peeling for macular holes performed by one surgeon using the heads-up 3-D system. These cases had excellent anatomic outcomes, with a 97.7-percent closure rate and only one case requiring reoperation. An additional benefit reported was the ability to decrease light source intensity which, theoretically, could reduce the risk of phototoxicity.9 The cost of acquiring the heads up 3D viewing system, however, may be a barrier to large-scale adoption. For example, the Ngenuity system produced by Alcon costs approximately $75,000. While there are likely no additional costs thereafter for continued use of the system, a practice must weigh whether this upfront cost justifies the additional benefits of the system versus operating with a traditional microscope. Another limitation noted in earlier 3-D viewing systems was an image delay however, this has decreased as the technology has improved.
More recently, a 2017 report compared traditional viewing systems versus heads-up 3-D displays for cases of retinal detachment.10 These cases consisted of both primary, simple retinal detachments and complex, recurrent detachments. The report consisted of seven cases repaired with a 3-D system and 15 with a traditional viewing system. There was no statistically significant difference in terms of anatomic success at 30 days (p=0.74) and no major postoperative complications.There was no statistically significant difference in surgical time between the two groups (55 ±35 for the 3-D group and 62 ±28 for the control group, p=0.07). Mean endoillumination power was significantly lower in the 3-D group (10 percent versus 45 percent in the control group, p<0.0001). The researchers noted, however, that surgical time is influenced by the difficulty of each singular case, and that the homogeneity between approaches can’t be guaranteed by a non-randomized retrospective comparison. Therefore, differences in operating time between the two systems should be “interpreted with caution.”
A larger study of 326 eyes compared the outcomes of a traditional viewing system (Resight) versus a 3-D heads-up viewing system (Ngenuity) for cases of idiopathic epiretinal membrane, vitreous hemorrhage with and without tractional retinal detachment, macular hole, rhegmatogenous retinal detachment (primary and PVR-related), pathologic myopic foveoschisis, silicone oil removal and vitreous opacities.11 This report found no statistically significant difference in postoperative visual acuity, anatomic outcomes or postoperative complications between the two groups.11
These reports didn’t identify increased complications or significant differences in outcomes when comparing a heads-up 3-D viewing system to traditional microscope viewing systems for a wide range of surgical indications in vitreoretinal surgery. In addition, the 3-D viewing systems may offer long-term ergonomic benefit.
In conclusion, since the development of pars plana vitrectomy in the 1970s, viewing systems have dramatically improved, resulting in excellent visualization in vitreoretinal surgery. Initial viewing systems, such as the modified Goldmann contact lens used by Machemer, provided limited visualization of the posterior pole, but subsequent innovations have increased the field of view and maintained excellent clarity. Non-contact wide-angle viewing systems have now become the standard in vitreoretinal surgery, allowing for excellent surgical results. In addition, the recent development of heads-up 3-D viewing systems can also lead to excellent vitreoretinal surgical outcomes and better ergonomics.
With each advance in viewing systems, visualization in vitreoretinal surgery continues to improve, and these advances ultimately lead to more precise surgeries and better patient outcomes. REVIEW
Dr. Gobuty Tieger is an ophthalmology resident at Massachusetts Eye and Ear, Harvard Medical School. Dr. Eliott is the director of the Retina Service at Massachusetts Eye and Ear’s department of ophthalmology.
Dr. Eliott is a consultant for Alcon, Alimera, Allergan, Dutch Ophthalmic, Genentech (DE), RegenxBio, and is on the scientific advisory board for Pykus Therapeutics. He’s a stockholder in Aldeyra Therapeutics.
Corresponding author:
Dean Eliott, MD (Dean_Eliott@meei.harvard.edu), Director, Retina Service, Department of Ophthalmology, Massachusetts Eye and Ear, 243 Charles Street, Boston, MA 02114. Phone: (617) 573-3736; fax: (617) 573-3698.
1. Chang S. Gonin Presentation - Wacker Prize Lecture. http://www.avi-panoramic.com/WackerLecturePrize.html, 1992.
2. Virata SR, Kylstra JA, Singh HT. Corneal epithelial defects following vitrectomy surgery using hand-held, sew-on, and noncontact viewing lenses. Retina 1999;19:4:287-290.
3. Oshima Y. Choices of wide-angle viewing systems for modern vitreoretinal surgery. Retina Today, September 2012.
4. Shah VA, Chalam KV. Self-stabilizing wide-angle contact lens for vitreous surgery. Retina 2003;23:5:667-669.
5. Murthy RK, Brar VS and Chalam KV. Evaluation of ultra wide angle “ora-ora” high refractive index self-stabilizing contact lens for vitreous surgery. Retina 2010; 30:9:1551-1553.
6. Liu P WW, Wu H, Tsai M. The comparison of sutureless vitrectomy (25g+ or 23g) with Alcon Constellation Vision System plus non-contact wild field lens system (OFFISS Lens System), and traditional 20g vitrectomy with contact lens system in management of vitreoretinal disease: A comparative pilot study. Invest Ophthalmol Vis Sci 2012;53:3756.
7. Tieger M, Rodriguez M, Wang J et al. Impact of contact versus non-contact wide angle viewing systems on outcomes of primary retinal detachment repair (PRO Study Report Number 5). Br J Ophthalmol. Accepted for publication April 17, 2020.
8. Spitznas M. A binocular indirect ophthalmomicroscope (BIOM) for non-contact wide-angle vitreous surgery. Graefes Arch Clin Exp Ophthalmol 1987;225:1:13-15.
9. Eckardt C, Paulo EB. Heads-up surgery for vitreoretinal procedures: An experimental and clinical study. Retina 2016;36:1:137-147.
10. Coppola M, La Spina C, Rabiolo A, Querques G, Bandello F. Heads-up 3D vision system for retinal detachment surgery. Int J Retina Vitreous 2017;3:46.
11. Zhang T, Tang W and Xu G. Comparative analysis of three-dimensional heads-up vitrectomy and traditional microscopic vitrectomy for vitreoretinal diseases. Curr Eye Res 2019;44:10:1080-1086.



