|
Many applications of laboratory-built human retinal tissues can be envisioned, including using them to test drugs and study degenerative diseases of the retina such as retinitis pigmentosa. One day, it may also be possible replace multiple layers of the retina in order to help patients with more widespread retinal damage.
“We don’t know how far this technology will take us, but the fact that we are able to grow a rudimentary retina structure from a patient’s blood cells is encouraging, not only because it confirms our earlier work using human skin cells, but also because blood as a starting source is convenient to obtain,” says
David Gamm, MD, pediatric ophthalmologist and senior author of the study. “This is a solid step forward.”
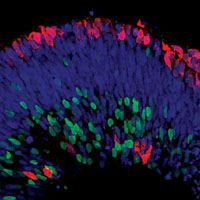
In 2011, Dr. Gamm’s lab at the UW Waisman Center created structures from the most primitive stage of retinal development using embryonic stem cells and stem cells derived from human skin. While those structures generated the major types of retinal cells, including photoreceptors, they lacked the organization found in more mature retina.
This time, the team, led by Dr. Gamm, assistant professor of ophthalmology and visual sciences in the UW School of Medicine and Public Health, and postdoctoral researcher and lead author Joseph Phillips, PhD, used their method to grow retina-like tissue from iPS cells derived from human blood gathered via standard blood draw techniques.
In their study, about 16 percent of the initial retinal structures developed distinct layers. The outermost layer primarily contained photoreceptors, whereas the middle and inner layers harbored intermediary retinal neurons and ganglion cells, respectively. This particular arrangement of cells is reminiscent of what is found in the back of the eye. Further, work by Dr. Phillips showed that these retinal cells were capable of making synapses, a prerequisite for them to communicate with one another.
The results were published in the
March 12, 2012 online issue of Investigative Ophthalmology & Visual Science.
Retinopathy A Marker for Cognitive Loss
Women 65 or older who have even mild retinopathy are more likely to have cognitive decline and related vascular changes in the brain, according to a study led by scientists at the University of California, San Francisco.
The findings suggest that a relatively simple eye screening could serve as a marker for cognitive changes related to vascular disease, allowing for early diagnosis and treatment, potentially reducing the progression of cognitive impairment to dementia.
A diagnosis could indicate early stages of Type II diabetes or hypertension, before they are clinically detectable. Early diagnosis could allow for lifestyle or drug interventions when they might be most effective.
“Lots of people who are pre-diabetic or pre-hypertensive develop retinopathy,” said the lead author of the study,
Mary Haan, DPH, MPH, UCSF professor of epidemiology and biostatistics. “Early intervention might reduce the progression to full- onset diabetes or hypertension.”
The results, reported in the
March 14, 2012, online issue of Neurology, were based on data from the Women’s Health Initiative Memory Study and the Site Examination study, two ancillary studies of the Women’s Health Initiative Clinical Trial of Hormone Therapy.
In the study, the team followed 511 women with an average starting age of 69 for 10 years. Each year, the women took a cognition test focused on short-term memory and thinking processes. In the fourth year, they received an exam to assess eye health. In the eighth year, they received a brain scan.
Of the full group of women, 39 women, or 7.6 percent, were diagnosed with retinopathy. On average, these women scored worse on the cognition test than the other women. They had more difficulty, for instance, recalling a list of several words five minutes after hearing them.
The women with retinopathy also had more damage to the blood vessels of the brain. They had 47 percent more ischemic lesions in the vasculature overall and 68 percent more lesions in the parietal lobe. The lesions, associated with vascular disease and sometimes stroke, are believed to be caused by high blood pressure. They also had more thickening of the white matter tracks that transmit signals in the brain, which also appear to be caused by high blood pressure.
Notably, the women did not have more brain atrophy, which is associated with Alzheimer’s disease. This result indicates that retinopathy is a marker of neurovascular disease rather than Alzheimer’s disease, according to Dr. Haan.
Swedish Study: Stem Cells to Epithelial Cells
Scientists at the Sahlgrenska Academy at the University of Gothenburg, Sweden, have for the first time successfully cultivated stem cells on human corneas, which may in the long term remove the need for donators.
Approximately 500 corneal transplantations are carried out each year in Sweden, and about 100,000 in the world. But the procedure requires a donated cornea, and there is a severe shortage of donated material, particularly where religious or political views hinder the use of donated material.
Scientists at the Sahlgrenska Academy have taken the first step towards replacing donated corneas with corneas cultivated from stem cells. Scientists
Charles Hanson, PhD, and Ulf Stenevi, MD, have used defective corneas obtained from the ophthalmology clinic at Sahlgrenska University Hospital in Mölndal.
Their study was published in Acta Ophthalmologica, and shows how human stem cells can be caused to develop into epithelial cells after 16 days’ culture in the laboratory and a further six days’ culture on a cornea.
“Similar experiments have been carried out on animals, but this is the first time that stem cells have been grown on damaged human corneas. It means that we have taken the first step towards being able to use stem cells to treat damaged corneas,” says Dr. Hanson. “If we can establish a routine method for this, the availability of material for patients who need a new cornea will be essentially unlimited. Both the surgical procedures and the aftercare will also become much more simple,” says Dr. Stenevi.
X-ray Stops Glaucoma in Mouse Model
Researchers at the Jackson Laboratory, an independent, biomedical research institution, have demonstrated that a single, targeted x-ray treatment of an individual eye in young, glaucoma-prone mice provided that eye with apparently life-long and typically complete protection from glaucoma. The research
was published March 19 in the Journal of Clinical Investigation.
Gareth Howell, PhD,
Simon John, PhD, and colleagues also used sophisticated genomics methods to uncover some of the very first pathways to change during glaucoma in these mice. The first pathway they detected to change suggests a critical mechanism that could be responsible for the earliest damage that glaucoma inflicts on the optic nerve.
About 10 years ago, Dr. John’s lab made the surprising discovery that just a single dose of whole body irradiation along with bone marrow transfer conferred an unprecedented protection against glaucoma: 96 percent of treated eyes had no glaucoma a year later compared to only 20 percent of control eyes. Although this result was unexpected, there is some evidence that radiation may protect from human glaucoma. Epidemiologists who followed Japanese survivors of the atomic bombings in World War II had noticed that exposure to radiation increased the incidence of thyroid and other cancers but seemed to provide protection against glaucoma.
The new demonstration that irradiation of just a single eye is protective, and at lower doses than they previously published, raises the possibility of using highly controlled localized radiation of just the eye to prevent human glaucoma.
Further research in other animal models to assess protection as well as safety and efficacy is necessary before attempting human treatments. But, as the authors conclude in their paper, “given both the robust and long-term efficacy of a single dose of X-ray radiation in preventing cellular entry into the optic nerve and retina, it will be important to further evaluate the use of X-rays for preventing glaucoma.”
The study showed that in response to early tissue stresses, monocytes, a class of immune cells, enter the optic nerve and retina in glaucoma. These monocytes express damaging molecules that appear critical for nerve damage in glaucoma. The entry of these cells is controlled in part by endothelial cells that line blood vessels. Radiation treatment appears to change how these endothelial cells respond to the early tissue stresses and affect the entry of the monocytes into the optic nerve and retina.
“While more work is needed to fully understand how the radiation confers long-term protection,” Dr. Howell says, “radiation appears to hinder the adhesion and migration of monocytes into the areas of the eye prone to nerve damage.” This finding strongly implicates the entry of cells into the eye as a key component of the nerve damage that leads to blindness. It also suggests vision may be maintained in eyes with high intraocular pressure by treatments that block the entry of monocytes into the eye. REVIEW