Initial Roadblocks
Most therapies for treating anterior-segment and ocular-surface diseases have traditionally involved the use of drops to deliver the drug. Topical drug delivery has significant advantages over other methods: It minimizes systemic exposure and systemic side effects; delivers drug precisely to the tissue of need; and reduces the potential for drug interactions. Despite these advantages, though, regulators have been reluctant to allow for concomitant contact lens use and topical delivery of drugs. One factor in these decisions is the perceived effects of preservatives such as benzalkonium chloride on the ocular surface, and the risk that if BAK-containing solutions are sequestered between the lens and the eye they might cause greater damage to cornea and conjunctiva. In addition, Phase II and Phase III studies of most topical agents are conducted on patients where contact lens use is typically excluded, leading to similar exclusions on package inserts. The approval process for lenses is separate, as they are classified as devices by the Food and Drug Administration, and concurrent use of topical medications is similarly excluded from this regulatory pathway.
Using a philosophy of, “If you can’t beat them, join them,” a number of studies have explored combining contact lenses with medications more commonly found in topical formulations as an alternative means of delivering drugs to the eye. The goal is to minimize the complications of lens use for these patients, while at the same time affording them a one-step method of addressing both medication needs and refraction correction. Providing therapeutic options to these patients is no small feat: There are an estimated 35 million contact lens users in the United States, and at least 125 million worldwide.1
 |
Drug-loading Strategies
Several different approaches have been investigated for combining drugs and soft contact lenses.2,3 These range from complex strategies such as the use of colloidal nanoparticles or molecular imprinting methods, to more straightforward methods such as including a drug in lens storage solutions. Each of these methods provides distinct advantages, so each may ideally be applicable for specific indications.
Colloidal nanoparticles are sub-micron-sized particles either coated with or encapsulating a drug’s molecules. Two common forms are composed of either lipid spheres (liposomes) or colloidal gold or silver;4-6 these are then dispersed into the contact lens matrix. Once the lenses are placed on the eye, the drug diffuses out of the contact lens matrix and away from the nanoparticle to enter the tear film. This packaging of drug in the colloidal particulate creates a two-step process that slows the diffusion of drug out of the lens and allows for a more sustained delivery of the drug to the corneal surface.
The idea of providing a nanoparticle vehicle for a drug within the matrix of the contact lens has been simplified in a methodology termed molecular imprinting.7-9 This process involves modification of the contact lens material to create a template for the drug molecule within the hydrogel complex. Examples of this are lenses synthesized in the presence of varying concentrations of the non-steroidal anti-inflammatory, diclofenac. By modifying the ratio of a key hydrogel component to drug, the capacity of the lens for drug and the drug release kinetics from the contact lenses can be systematically varied.10 Imprinted contact lenses allow for a significantly higher drug load than lenses loaded by simple diffusion in a drug solution, and so may be particularly useful in situations in which a higher dose of the drug is required for therapeutic efficacy.8,9
One of the earliest and simplest methods of preparing drug-lens combinations is to simply equilibrate the lens in a physiological solution containing the drug. The hydrophilic matrix of soft contact lenses, which ranges from 30 to 80 percent water, can absorb the drug and then release it by simple diffusion when inserted into the eye. This process was demonstrated successfully in an early study using lenses loaded with pilocarpine as a treatment for acute closed-angle glaucoma;11 after equilibration with 1% pilocarpine, a two-hour contact lens treatment reduced intraocular pressure by a mean of 46 percent, from 55.6 to 30 mm Hg. While the simplicity of this method of loading drug is expedient, it’s limited by the rapid pharmacokinetics of diffusion, yielding a dose of, and exposure to, the therapeutic agent that is similar to topical application. A study comparing uptake and release of commonly used ocular pharmaceuticals in several soft contact formulations showed that the maximal lens uptake was drug-specific; for example, similar lenses could absorb 7 to 8 mg of cromolyn sodium but only 0.07 mg of dexamethasone sodium phosphate.12 In addition, the rates of uptake and release were different with each drug. For most tested compounds, both uptake and release were rapid and complete in under an hour; the exception to this was the antihistamine ketotifen, which eluted more gradually over approximately five hours.
Loading strategies are likely to be influenced by the specific indications involved. While a drug-lens combination for glaucoma would benefit from the prolonged release provided by nanoparticles or imprinting, treatments for dry eye or allergy could benefit from diffusion-loaded lenses. Regardless of the loading strategy that’s used, all drug-lens combinations face a common set of logistical hurdles before they reach the market.
Further Challenges
Despite what you might have read about the novelty of Google’s lenses that measure blood pressure or glucose, efforts to bring multitasking to the contact lens realm have been around for decades, and are more nuanced than it might seem. In the case of the Google lenses, we know that the barrier function of the conjunctival vasculature can impact the exchange of solutes between tear film and general circulation, so hyperemia may alter measures of hyperglycemia. For combinations of lenses and therapeutics, there are still several hurdles to clear on the way to creating a viable drug-delivery device beyond just regulatory and safety issues. These include pharmacokinetic issues, lens material issues and drug stability concerns.
|
Another major concern is the potential loss of lens transparency, critical to visual function, in the course of the loading or storage process. This is a potential issue when surfactants are included in the formulation process of incorporating the drug in the lenses.13 Opacity of the lens can be avoided by opting for drug-loaded nanoparticles, which can be preferentially loaded towards the periphery of the lens to minimize the impact on visual function. Loading contact lenses with drugs also can potentially diminish the oxygen permeability of the lens. This is of particular concern in long-term use, as it could lead to corneal edema and epithelial damage.3
In some cases, therapeutic benefit may be limited by the drug capacity of the lens, and this will be a function of the equilibrium solubility of the drug. This is of particular concern when working with hydrophobic drugs, as the hydrophilic matrix of soft contact lenses is not a receptive environment for hydrophobic drug molecules.14 Attempts have been made to address this issue by encapsulating the hydrophobic molecules in either liposomes or nanoparticles. The use of molecular imprinting may also improve the delivery of hydrophobic drugs, but in such cases release of the drug is likely to be slowed, yielding a longer duration of exposure but a lower peak drug concentration in the tear film.
The Future of Combinations
The growing number of drug-lens combinations in clinical and pre-clinical development suggests that we are on the verge of a real breakthrough. For example, in pre-clinical studies with latanoprost-loaded lenses, encapsulation of the drug in a biodegradable film provides therapeutic levels of drug in the aqueous humor for at least a month.15 This type of lens could be beneficial even for patients who don’t require vision correction, because of the issues of compliance associated with the use of IOP-lowering agents.
Several drug-lens combinations have reached the clinic, including a ketotifen-lens combination (Vistakon/Johnson&Johnson) for patients with allergic conjunctivitis (see, for example, NCT 00432757), and a combination product for lens wearers with dry eye containing the plant polysaccharide lubricant alginic acid (NCT 01918410). These combination products have the potential to provide allergy or dry-eye patients with a valuable option for vision correction, and would address two major causes of contact lens dropouts.
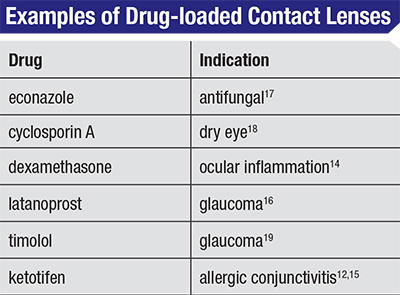
Other examples of drug-lens combinations in development are listed in the table on p. 57. Each provides a distinct advantage over current therapy, and allows for combining drug treatment with vision correction if needed. Simplifying treatments, especially when it means reducing the number of steps our patients must execute to receive appropriate therapy, is almost always a good thing. While the process of combining contact lenses with drugs has taken more time than we could have imagined, the end results—better compliance, better therapeutic outcomes and better vision—will be worth the effort. REVIEW
Dr. Abelson is a clinical professor of ophthalmology at Harvard Medical School. Dr. McLaughlin is a medical writer at Ora Inc.
1. http://www.aao.org/newsroom/upload/Eye-Health-Statistics-April-2011.pdf Accessed 12/30/2014
2. Guzman-Aranguez A, Colligris B, Pintor J. Contact lenses: Promising devices for ocular drug delivery. J Ocul Pharm Ther 2013;29:2:189-99.
3. Gulsen D, Chauhan A. Ophthalmic drug delivery through contact lenses. Invest Ophthalmol Vis Sci 2004;45:7:2342-7.
4. Sung AY, Kim TH. Ophthalmic application of poly (HEMA)s containing nanoparticles and dimethylacrylamide. J. Nanosci. Nanotechnol 2012;12:7:5210-5.
5. Gulsen D, Li C-C, Chauhan A. Dispersion of DMPC Liposomes in Contact Lenses for Ophthalmic Drug Delivery. Curr Eye Res 2005;30:12:1071-80.
6. Garhwal R, Shady SF, Ellis EJ, et al. Sustained ocular delivery of ciprofloxacin using nanospheres and conventional contact lens materials. Invest Ophthalmol Vis Sci 2012;53:3:1341-52.
7. White CJ, Byrne ME. Molecularly imprinted therapeutic contact lenses. Expert Opin Drug Delivery 2010; 7:6:765-80.
8. Alvarez-Lorenzo C, Yañez F, Barreiro-Iglesias R, Concheiro A. Imprinted soft contact lenses as norfloxacin delivery systems. J Controlled Release 2006;113:3:236-44.
9. Hiratani H, Alvarez-Lorenzo C. Timolol uptake and release by imprinted soft contact lenses made of N, N-diethylacrylamide and methacrylic acid. J of Controlled Release 2002; 83:2:223-30.
10. Tieppo A, Pate KM, Byrne ME. In vitro controlled release of an anti-inflammatory from daily disposable therapeutic contact lenses under physiological ocular tear flow. Eur J Pharm Biopharm 2012;81:1:170-7.
11. Hillman JS. Management of acute glaucoma with pilocarpine-soaked hydrophilic lens. Brit J Ophthalmol 1974;58:7:674-9.
12. Karlgard CC, Wong NS, Jones LW, Moresoli C. In vitro uptake and release studies of ocular pharmaceutical agents by silicon-containing and p-HEMA hydrogel contact lens materials. International Journal of Pharmaceutics 257:2003:141–151.
13. Kapoor Y, Thomas JC, Tan G, John VT, Chauhan A. Surfactant-laden soft contact lenses for extended delivery of ophthalmic drugs. Biomaterials 2009; 30:5:867-78.
14. Kim J, Peng CC, Chauhan A. Extended release of dexamethasone from silicone-hydrogel contact lenses containing vitamin E. J of Controlled Release 2010;148:1:110-6.
15. Soluri A, Hui A, Jones L. Delivery of ketotifen fumarate by commercial contact lens materials. Optom Vis Sci 2012;89:8:1140-9.
16. Ciolino JB, Stefanescu CF, Ross AE, Salvador-Culla B, Cortez P, Ford EM, et al. In vivo performance of a drug-eluting contact lens to treat glaucoma for a month. Biomaterials 2014;35:1:432-9.
17. Ciolino JB, Hudson SP, Mobbs AN, et al. A prototype antifungal contact lens. Investigative ophthalmology & visual science 2011; 52:9:6286-91.
18. Peng CC, Chauhan A. Extended cyclosporine delivery by silicone-hydrogel contact lenses. Journal of Controlled Release. 2011;154:3:267-74.
19. Jung HJ, Abou-Jaoude M, Carbia BE, Plummer C, Chauhan A. Glaucoma therapy by extended release of timolol from nanoparticle loaded silicone-hydrogel contact lenses. Journal of Controlled Release 2013;165 :1:82-9.




